How Cysteine-Depleting 4T1 Tumor Models Revealed Synergistic Chemotherapy, Ferroptosis, and Immunotherapy
Introduction
Paclitaxel, a first-line chemotherapeutic agent, faces significant clinical challenges due to its extremely low water solubility and severe systemic toxicity. Although various formulations have been developed, low bioavailability and toxic side effects remain major obstacles to achieving a satisfactory therapeutic window. While combination therapies show promise in advanced cancers, how to simultaneously enhance drug delivery efficiency and anti-tumor activity through simple molecular design requires further exploration.
For breast cancer research utilizing triple-modality combination therapy, our validated 4T1 and B16-OVA cell lines provide robust models for evaluating complex therapeutic strategies.
Order Now>>
4T1 : 4T1 Cell Line / 4T1-OVA Cell Line / 4T1-Luciferase Cell Line
B16: B16 Cell Line / B16 -OVA Cell Line / B16 -Luciferase Cell Line
Innovative Prodrug Design: Cysteine-Depleting Paclitaxel Conjugate
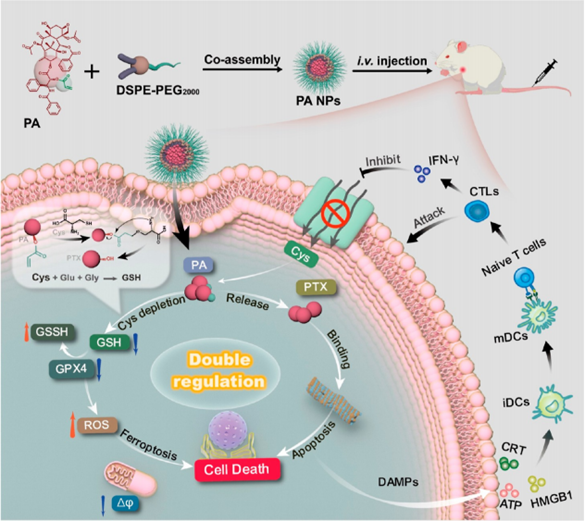
Traditional prodrug designs have primarily focused on improving drug solubility or achieving responsive release, often overlooking the potential for prodrugs to serve as functional modules in therapeutic regulation. This study introduces a novel approach where paclitaxel was conjugated with acrylic acid through a one-step esterification reaction, creating prodrug PA with cysteine-depletion capability. The control product PC, conjugated with hexanoic acid, was also synthesized for comparison. High-performance liquid chromatography analysis demonstrated that when PA was co-incubated with cysteine, the PA peak gradually decreased while the PTX (paclitaxel) peak increased correspondingly, achieving over 90% PTX release within 6 hours. No significant change was observed with PC under identical conditions.
Electrospray mass spectrometry detected characteristic peaks for PA-Cys intermediate and cyclic amide products, confirming the reaction mechanism: the alkene group of PA reacts with the thiol group of cysteine to form a thioether intermediate, followed by intramolecular cyclization that releases PTX and cyclic amide. Notably, PA exhibited specificity toward cysteine with minimal reactivity toward other amino acids. To form stable nanoparticles, PA was self-assembled with DSPE-PEG2000 using thin-film hydration, producing PA NPs with an average diameter of approximately 142 nm. Transmission electron microscopy revealed uniform spherical morphology, and the nanoparticles demonstrated excellent stability in 5% glucose and 0.9% NaCl solutions with no significant size change after one week of storage at 4°C.
Targeted Delivery and Dual-Modal Cytotoxicity
Cellular uptake experiments using BDP-labeled PA NPs demonstrated time- and energy-dependent internalization by 4T1 cells with strong co-localization with lysosomes. Researchers first confirmed that tumor cells (4T1, B16-OVA) contained higher intracellular cysteine levels compared to normal cells (L929, NIH-3T3). MTT assays revealed that PA NPs exhibited significantly superior cytotoxicity against 4T1 cells compared to PC NPs and conventional Taxol, reducing cell viability to only 11.8% at 20 μM PTX-equivalent concentration. The cytotoxic effect was dose-dependently reversed by exogenous cysteine supplementation, confirming dependence on cysteine depletion.
For precise evaluation of prodrug nanoparticles and combination therapies, our comprehensively characterized 4T1 breast cancer cell line serves as an ideal in vitro model. Request a quote now to access our standardized cell culture protocols and detailed characterization documentation.
Ferroptosis Induction through Cysteine-GSH-GPX4 Axis Disruption
Mechanistic investigations revealed that PA NPs induced ferroptosis—a distinct form of programmed cell death characterized by iron-dependent lipid peroxidation. Following PA NP treatment, 4T1 cells exhibited significantly reduced intracellular GSH content, downregulated GPX4 expression, elevated ROS levels, and increased malondialdehyde (MDA), a lipid peroxidation product. Importantly, ferroptosis inhibitor Lip-1 partially reversed the cytotoxicity, confirming ferroptosis involvement. JC-1 staining demonstrated significant mitochondrial membrane potential decline and structural damage, while microtubule immunofluorescence revealed noticeable microtubule aggregation indicating successful intracellular PTX release. These findings confirm that PA NPs achieve dual anti-tumor effects through PTX chemotherapy and ferroptosis induction via cysteine depletion.
Immunogenic Cell Death and Systemic Immune Activation
Having validated the in vitro anti-tumor mechanisms, researchers assessed the immune activation capability and in vivo anti-tumor efficacy of PA NPs. Immunofluorescence staining demonstrated that PA NP treatment increased calreticulin membrane exposure, HMGB1 release, and ATP secretion from 4T1 cells, confirming successful induction of immunogenic cell death (ICD). When supernatants from treated cells were co-incubated with DC2.4 dendritic cells, flow cytometry revealed significantly enhanced DC maturation in the PA NP group. Importantly, activated T cells secreted IFN-γ, which further inhibited cystine uptake, creating a feedback loop that enhanced ferroptosis efficacy when combined with PA NPs.
Superior In Vivo Efficacy and Immune Microenvironment Remodeling
In 4T1 tumor-bearing mouse models, IR780-labeled PA NPs showed excellent accumulation at tumor sites, peaking at 10 hours and maintaining for 72 hours. Treatment experiments demonstrated that the PA NP group exhibited the best tumor suppression, with tumor volume and weight significantly lower than Taxol and PC NP groups, even surpassing clinical formulation Abraxane. Tumor tissue sections stained with H&E showed maximal nuclear fragmentation and cytoplasmic reduction in the PA NP group, while GPX4 immunofluorescence revealed the lowest expression. Tumor-draining lymph nodes showed DC maturation rates reaching 49.1%, and tumors exhibited significantly increased infiltration of CD4+ and CD8+ T cells alongside enhanced T cell proliferation in the spleen.
ELISA analysis detected elevated levels of TNF-α, IL-6, and IFN-γ in tumor tissues. PA NPs also remodeled the immunosuppressive microenvironment by promoting M1 macrophage polarization and reducing myeloid-derived suppressor cell (MDSC) numbers, with CD8 immunofluorescence confirming increased T cell infiltration. Safety evaluation showed stable body weight in the PA NP group with normal blood parameters and major organ histology, while the Taxol group exhibited weight loss, confirming the excellent biological safety profile of PA NPs.
Conclusion
This study successfully constructed paclitaxel prodrug PA with cysteine-depletion functionality through simple one-step esterification, enabling self-assembly into stable PA NPs. These nanoparticles specifically respond to intracellular cysteine to release PTX while simultaneously inducing ferroptosis through cysteine depletion and GSH downregulation. The released PTX executes conventional chemotherapy and induces immunogenic cell death, while the activated immune system further enhances ferroptosis efficacy through IFN-γ feedback. This therapeutic platform achieved triple synergistic effects—chemotherapy, immunotherapy, and ferroptosis—in breast cancer models with lower systemic toxicity than clinical paclitaxel formulations.
For advanced breast cancer research combining chemotherapy with emerging therapeutic modalities, our thoroughly authenticated 4T1 cell models offer consistent performance for evaluating complex therapeutic strategies. Place your order and personalized technical support for your specific research objectives.
Order Now>>
4T1 : 4T1 Cell Line / 4T1-OVA Cell Line / 4T1-Luciferase Cell Line
B16: B16 Cell Line / B16 -OVA Cell Line / B16 -Luciferase Cell Line
Significance
This research demonstrates how innovative prodrug molecular design can integrate ferroptosis induction into classical chemotherapeutic agents, achieving synergistic integration of multimodal therapies. This "three-birds-with-one-stone" simple strategy provides new insights for developing efficient, low-toxicity combination treatments and highlights the important value of functionalizing "non-pharmacophore groups" in prodrug design. The approach represents a paradigm shift in addressing the limitations of traditional chemotherapy through rational molecular engineering.
References
https://pubs.acs.org/doi/10.1021/acsnano.4c06399

