A549-luc: Illuminating How Chemotherapy-Induced Senescence Drives Treatment Resistance in Lung Cancer
Introduction
Lung cancer remains the leading cause of cancer-related mortality worldwide, claiming over 1.8 million lives annually. Non-small cell lung cancer (NSCLC), which accounts for approximately 85% of all lung cancers, is routinely treated with platinum-based chemotherapy as a foundational regimen. Despite initial responses, many patients eventually develop treatment resistance, and the biological mechanisms that drive this resistance remain incompletely understood. One underexplored mechanism involves therapy-induced cellular senescence—a durable proliferative arrest triggered by chemotherapy—and the pro-tumorigenic secretome (SASP) that senescent cells release into the tumor microenvironment (TME).
Power your lung cancer research with our A549 cell line— Contact us to discuss your research needs and receive expert technical support.
Contact us now: A549 Cell Line / A549 Luciferase Cell Line
Background: The Double-Edged Sword of Chemotherapy
Cisplatin and other platinum-based drugs exert their cytotoxic effects by cross-linking DNA, triggering permanent cell cycle arrest in a fraction of treated tumor cells. These senescent cells upregulate a senescence-associated secretory phenotype (SASP)—a complex mixture of cytokines, chemokines, growth factors, and proteases that can substantially remodel the TME. Although SASP was initially regarded as tumor-suppressive, accumulating evidence suggests it can paradoxically stimulate the proliferation and survival of neighboring, non-senescent cancer cells, thereby driving treatment resistance and relapse. Before this study, the specific SASP components responsible for this paracrine pro-tumorigenic effect in NSCLC, and the downstream signaling pathways they activate, had not been systematically characterized.
Study Design: A549-luc as the Central Experimental Model
The research team at Cambridge University employed A549-luc cells—a human lung adenocarcinoma cell line stably expressing firefly luciferase—as their principal NSCLC model. The luciferase reporter enabled real-time, non-invasive bioluminescent monitoring of tumor burden in live animals, a capability that proved crucial for longitudinal assessment of tumor growth and treatment responses in vivo. Senescence was induced by exposing A549 cells to cisplatin (15 μM for 7 days), docetaxel (200 nM), or palbociclib (a CDK4/6 inhibitor). Conditioned medium (CM) was collected from these senescent cells and applied to non-senescent A549-luc cells to model paracrine SASP effects. An orthotopic lung model used tail-vein injection of L1475(luc) mouse lung cancer cells for bioluminescence imaging of lung colonization.
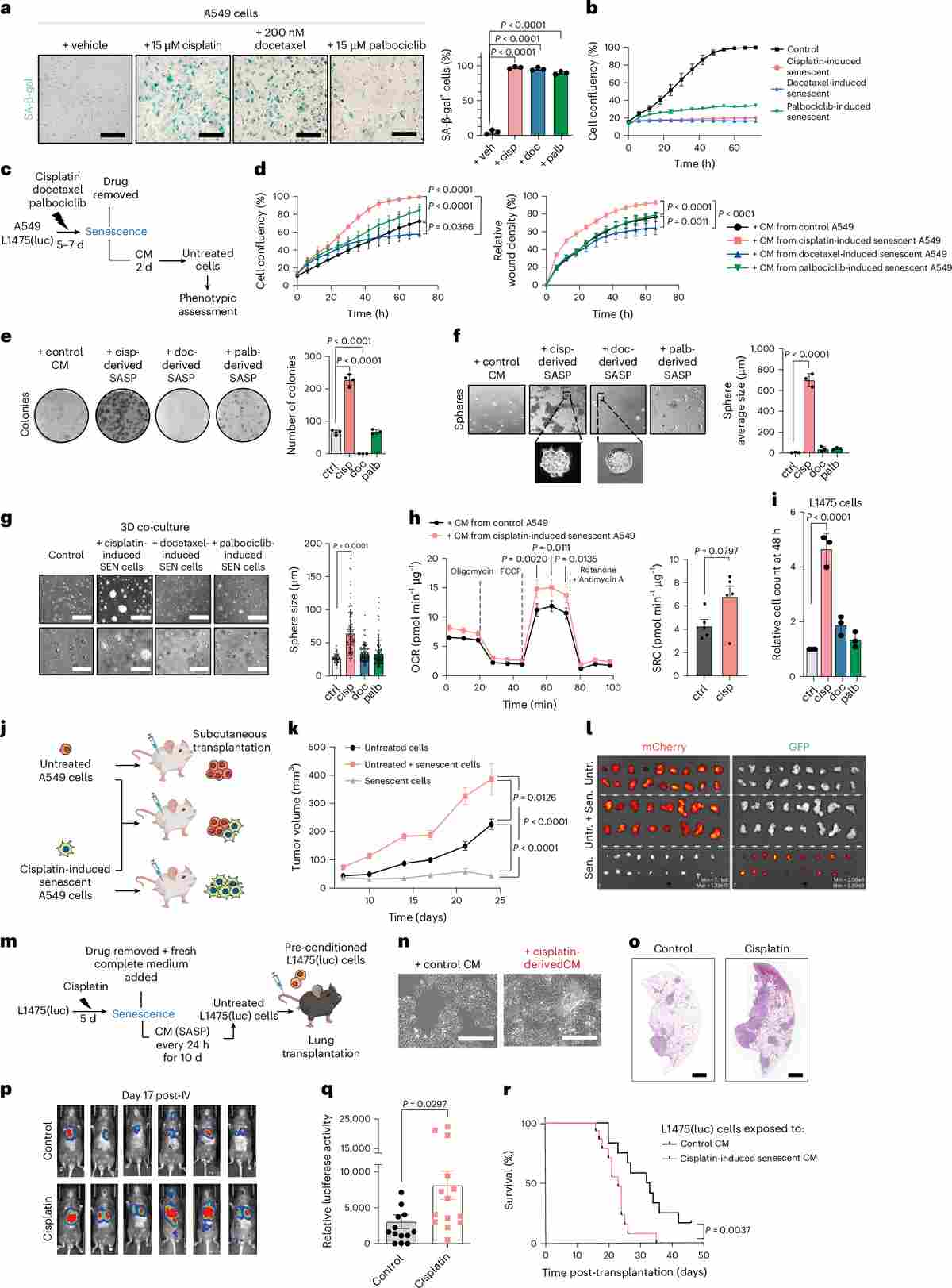
Figure 1. Cisplatin-induced senescent A549 and L1475(luc) cells and their secretomes promote increased tumor growth in xenograft and orthotopic models of NSCLC. (a–b) SA-β-gal staining and confluence curves confirm senescence induction. (c–h) Conditioned medium from senescent A549 cells enhances proliferation, migration, colony formation, spheroid growth, and metabolic capacity in non-senescent counterparts. (i–r) In vivo xenograft and orthotopic lung colonization models demonstrate significantly elevated tumor burden and shortened survival in mice receiving senescent-cell-conditioned A549-luc cells.
Key Finding 1: TGFβ-Dominant SASP Drives Malignant Phenotypes
Transcriptomic (RNA-seq) and proteomic profiling of cisplatin-induced senescent A549 cells revealed a SASP signature markedly enriched in TGFβ ligands (TGFβ1, TGFβ2, TGFβ3) compared with senescence induced by docetaxel or palbociclib. A microenvironment microarray (MEMA) platform was used to systematically screen 80 SASP-associated ligands for their ability to enhance A549-luc proliferation, as measured by EdU incorporation. TGFβ1 and TGFβ2 emerged as the top-ranked drivers of enhanced proliferation, confirmed by ELISA quantification of their secreted levels in conditioned medium.
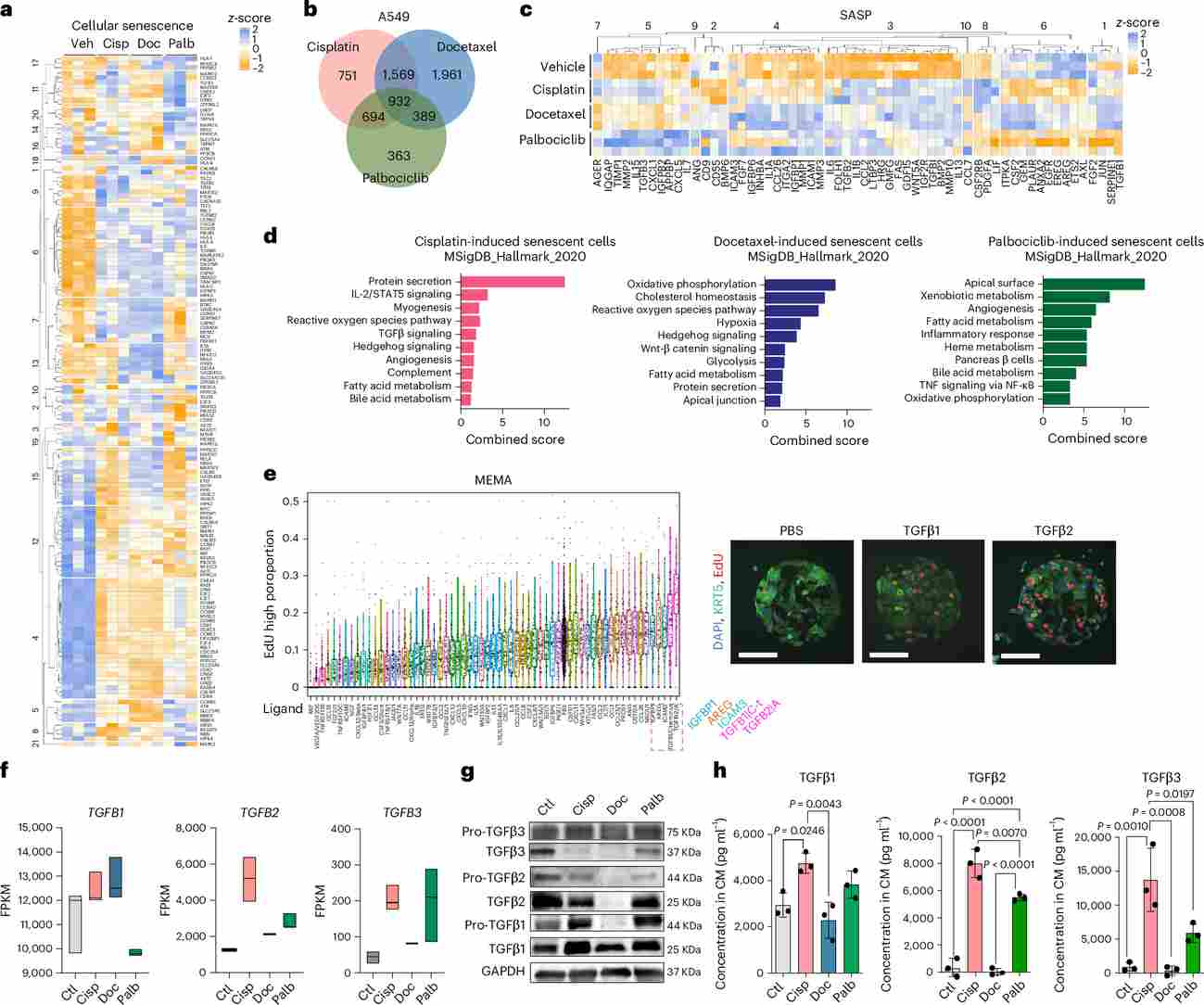
Figure 2. Transcriptomic and proteomics analyses reveal different transcriptional and SASP signatures of chemotherapy-induced senescent cells and highlight TGFβ ligands as potential candidate drivers of malignant traits in cisplatin-derived SASP. (a) Heatmap of senescence-pathway gene z-scores. (b) Venn diagram of upregulated genes across three senescence inducers. (c–d) SASP gene expression heatmap and MSigDB Hallmark pathway enrichment. (e) MEMA screen identifying TGFβ1/β2 as top proliferation-promoting ligands. (f–h) FPKM, Western blot, and ELISA data confirming elevated TGFβ in cisplatin-derived SASP.
Key Finding 2: TGFBR1–AKT/mTOR Axis Mediates Resistance
To delineate the signaling mechanism, phospho-kinase arrays and Western blot analyses were performed on non-senescent A549-luc cells exposed to cisplatin-SASP conditioned medium. AKT and its downstream effectors p70S6K and 4E-BP1 were consistently hyperphosphorylated within 30 minutes of SASP exposure, indicative of mTOR pathway activation. shRNA-mediated knockdown of TGFBR1 (the primary type I TGFβ receptor) fully abrogated SASP-driven proliferation, confirming TGFBR1 as the apical receptor. Furthermore, treatment with galunisertib (LY2157299), a clinical TGFBR1 kinase inhibitor, dose-dependently suppressed AKT/mTOR phosphorylation and reversed SASP-promoted colony formation and spheroid growth.
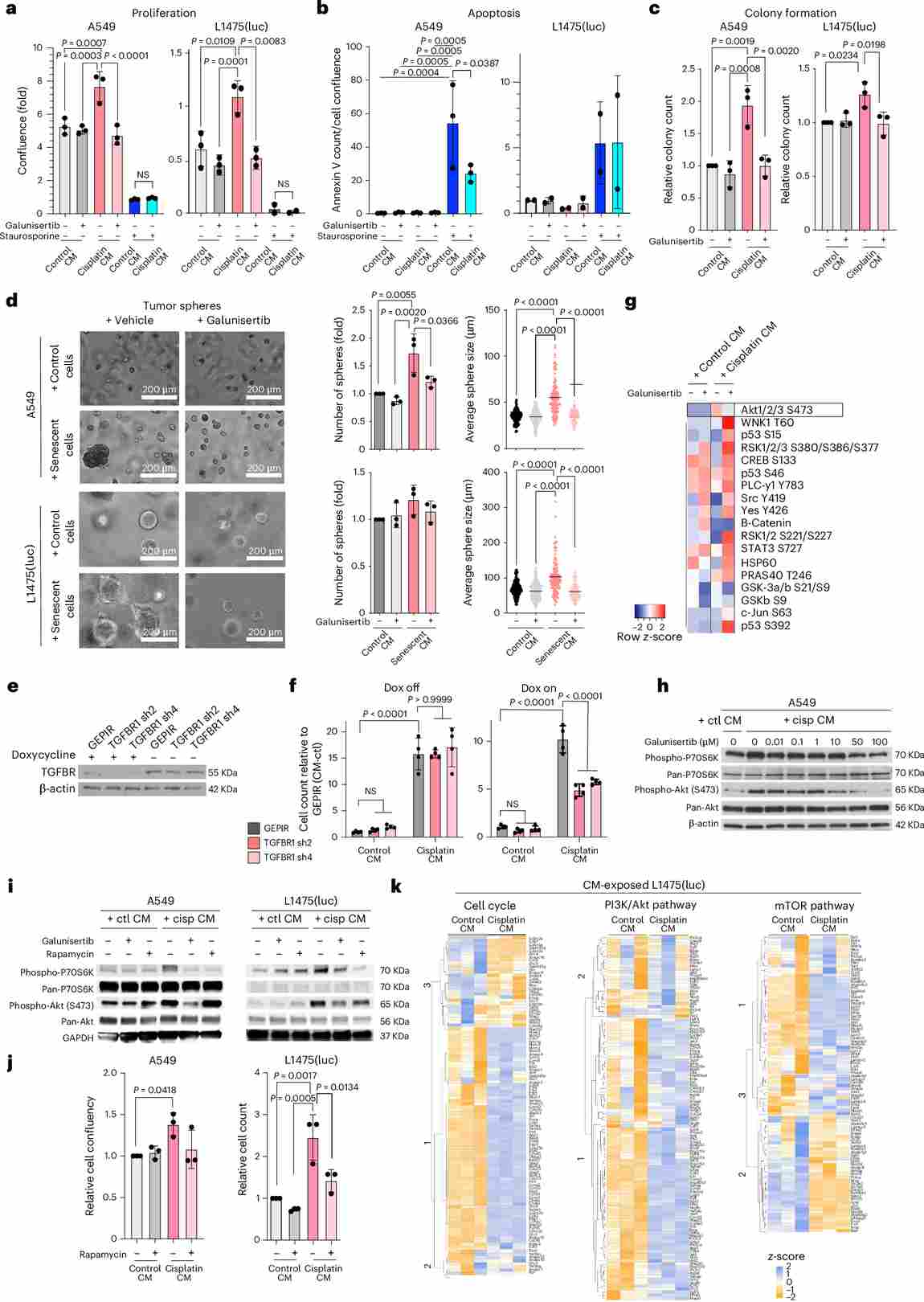
Figure 3. TGFBR1-driven activation of AKT/mTOR pathway orchestrates the induction of malignant traits upon exposure to cisplatin-derived SASP. (a–d) Galunisertib reverses SASP-driven proliferation, apoptosis resistance, colony formation, and spheroid growth in A549 and L1475(luc) cells. (e–f) TGFBR1 shRNA knockdown abrogates SASP-induced proliferation. (g–k) Phospho-kinase array, Western blot, and transcriptomic analyses identify AKT/mTOR as the key downstream effector pathway.
In Vivo Validation: Bioluminescence Imaging with A549-luc
The bioluminescent capability of A549-luc cells was pivotal for real-time in vivo assessment. In the subcutaneous xenograft model, co-injection of non-senescent A549-luc cells with cisplatin-induced senescent A549 cells resulted in significantly larger tumors at day 30 compared to controls. Galunisertib treatment reduced co-injection tumor weight by approximately 48%, and ABT-737 (a senolytic) further decreased tumor burden by selectively eliminating senescent cells. In the orthotopic lung colonization model, bioluminescence imaging at day 10 revealed markedly elevated luciferase signal intensity in mice whose L1475(luc) cells had been pre-conditioned with cisplatin-SASP medium. Kaplan–Meier survival analysis demonstrated significantly shortened survival in SASP-exposed groups (median ~21 days vs ~30 days in controls, p < 0.001).
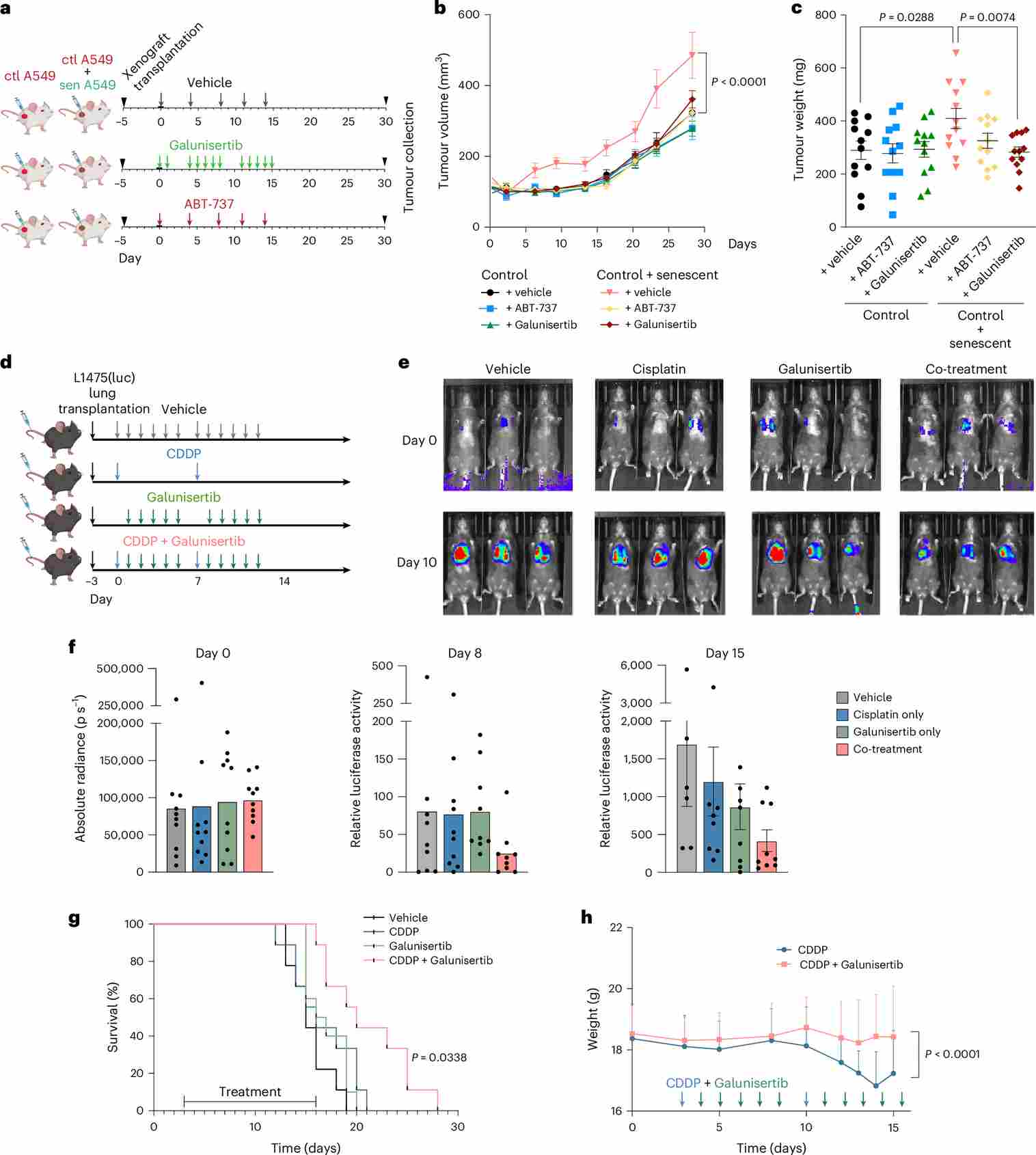
Figure 4. TGFBR1 inhibition effectively prevents enhanced proliferation derived from the exposure of cisplatin-induced senescent SASP in xenograft and orthotopic models of lung adenocarcinoma. (a) Experimental design schematic. (b–c) Tumor volume and weight over time showing galunisertib-mediated reduction. (d) Orthotopic model layout with tail-vein injection of L1475(luc) cells. (e–f) Representative bioluminescence images and quantification of luciferase activity at days 2 and 10. (g–h) Kaplan–Meier survival curves and body weight changes across treatment groups.
Genetically Engineered Mouse Model: KrasG12V-Driven Lung Cancer
To evaluate the combination regimen in a clinically relevant immunocompetent model, the team employed KrasG12V/+;p53frt/frt genetically engineered mice (GEMM). Lung tumors were induced by adenoviral FLP recombinase delivery and confirmed by CT imaging 8 weeks post-induction. Animals then received cisplatin, galunisertib, both, or vehicle over a defined treatment window. Strikingly, combined cisplatin–galunisertib treatment produced the greatest reduction in tumor volume (median fold-change 0.62 vs 2.1 in cisplatin-only group), significantly prolonged survival, and reduced pAKT and p70S6K immunohistochemical staining in residual tumors, consistent with in vitro mechanistic findings. This GEMM model adds important translational weight to the combination strategy.
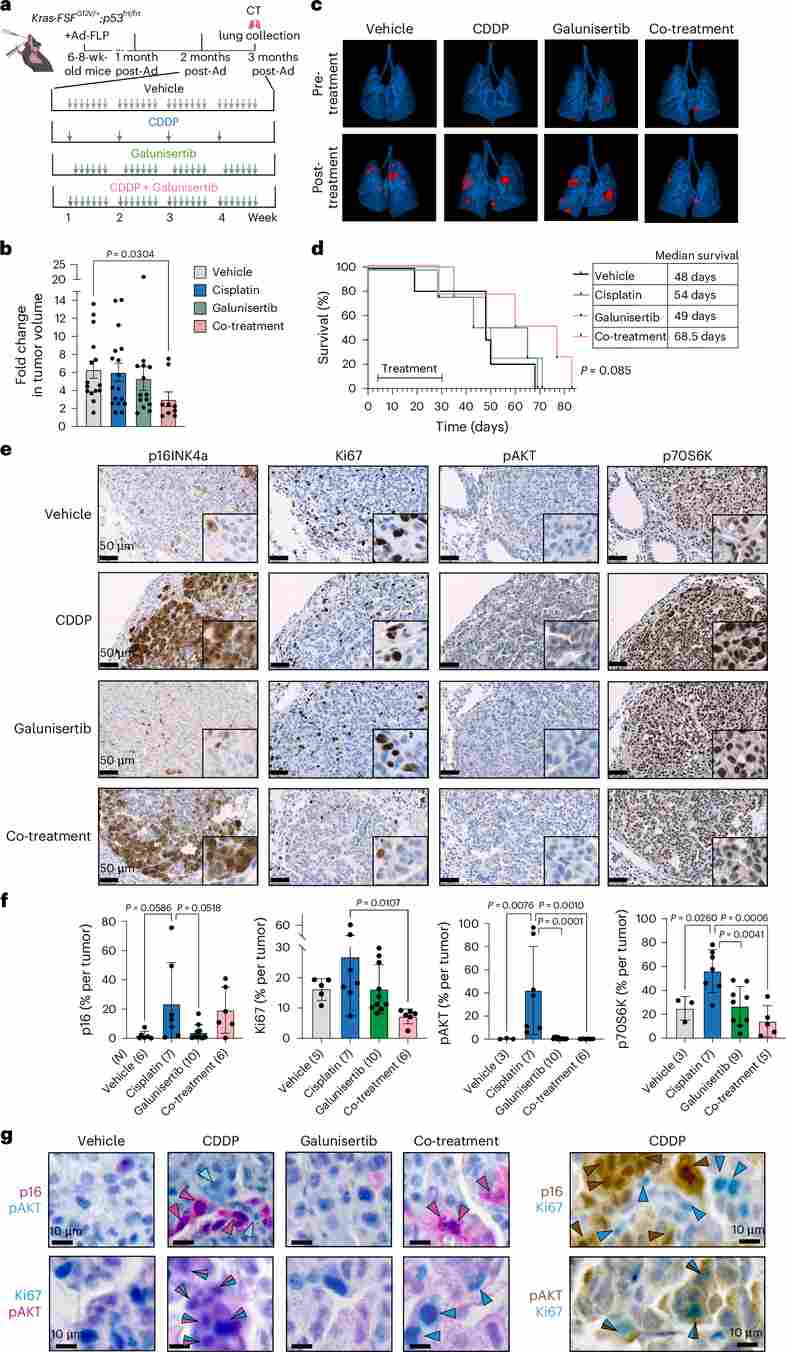
Figure 5. Cisplatin and galunisertib concomitant treatment significantly reduces tumor burden and enhances survival in a KrasG12V-oncogene-driven model of lung cancer. (a) Experimental layout schematic for KrasG12V/+;p53frt/frt GEMM. (b) CT-measured tumor volume fold-change after treatment. (c) 3D lung CT reconstructions before and after treatment (tumors in red). (d) Kaplan–Meier survival curves. (e–f) Representative IHC images and quantification of p16, Ki67, pAKT, and p70S6K. (g) Dual IHC co-staining showing spatial correlation between p16+ senescent cells and pAKT+ proliferating neighbors.
Clinical Translatability: Evidence from NSCLC Patient Samples
To contextualize the preclinical findings, the research team analyzed matched pre- and post-treatment tumor tissue from NSCLC patients who received neoadjuvant platinum-based chemotherapy. Post-treatment specimens showed a significant increase in p21+, p16+, and SenTraGor+ cells (markers of therapy-induced senescence) compared to treatment-naive biopsies. Critically, areas with high p21 density were spatially associated with elevated phospho-AKT and phospho-P70S6K staining in neighboring non-senescent cells—recapitulating the paracrine SASP-driven signaling observed in vitro and in animal models. Co-staining of consecutive tissue sections also revealed elevated TGFβ1 expression in p21-rich zones, connecting the clinical observation to the TGFβ-SASP mechanism.
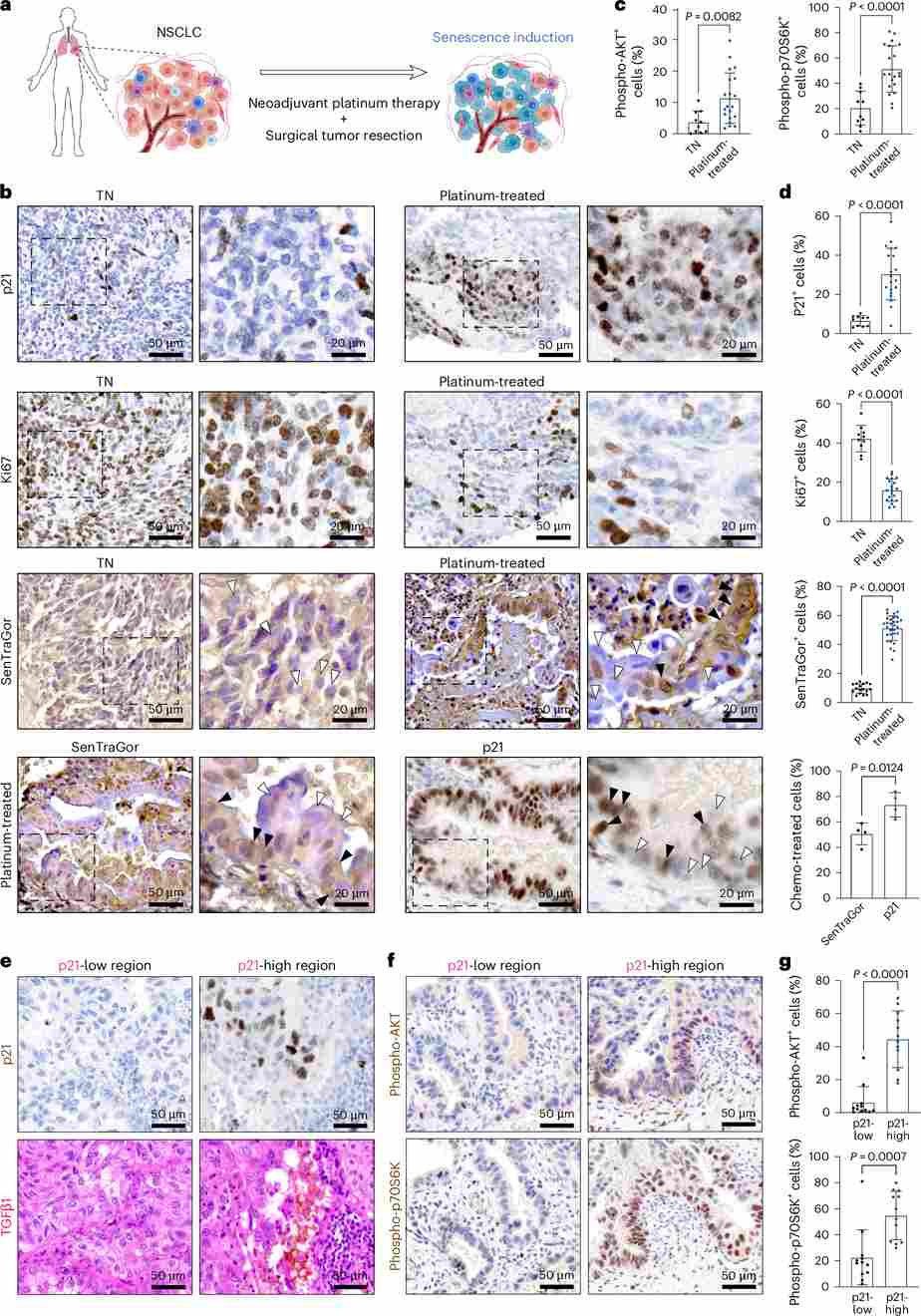
Figure 6. Neoadjuvant platinum-based therapy induces senescence in patients with NSCLC, and this correlates with increased phospho-AKT and phospho-P70S6K signaling in surrounding cells. (a) Study schematic for human NSCLC samples. (b) Representative IHC images of Ki67, p21, and SenTraGor (GL13) staining. (c–d) Quantification of pAKT+, pP70S6K+, Ki67+, p21+, and GL13+ cells in treatment-naive vs. platinum-treated specimens. (e–f) Co-staining of p21 with TGFβ1, pAKT, and pP70S6K in consecutive sections. (g) Spatial stratification by p21-low vs. p21-high zones reveals elevated pAKT/pP70S6K in senescence-rich regions.
Ovarian Cancer Evidence: Transcriptomic Analysis
The study further extended its findings to high-grade serous ovarian cancer (HGSOC). Using the CTCR-OV01 clinical trial microarray dataset (GEO: GSE15622), gene set enrichment analysis (GSEA) was performed comparing pre-treatment and post-treatment tumor biopsies from chemotherapy-treated ovarian cancer patients. Post-treatment samples were significantly enriched in senescence, TGFβ, and AKT/mTOR signaling gene sets, mirroring the transcriptomic signature observed in cisplatin-treated A549 cells. This cross-cancer validation demonstrates that TGFβ-driven senescent secretome signaling is not a lung-cancer-specific phenomenon but a broad mechanism of platinum-resistance across gynecological and thoracic malignancies.
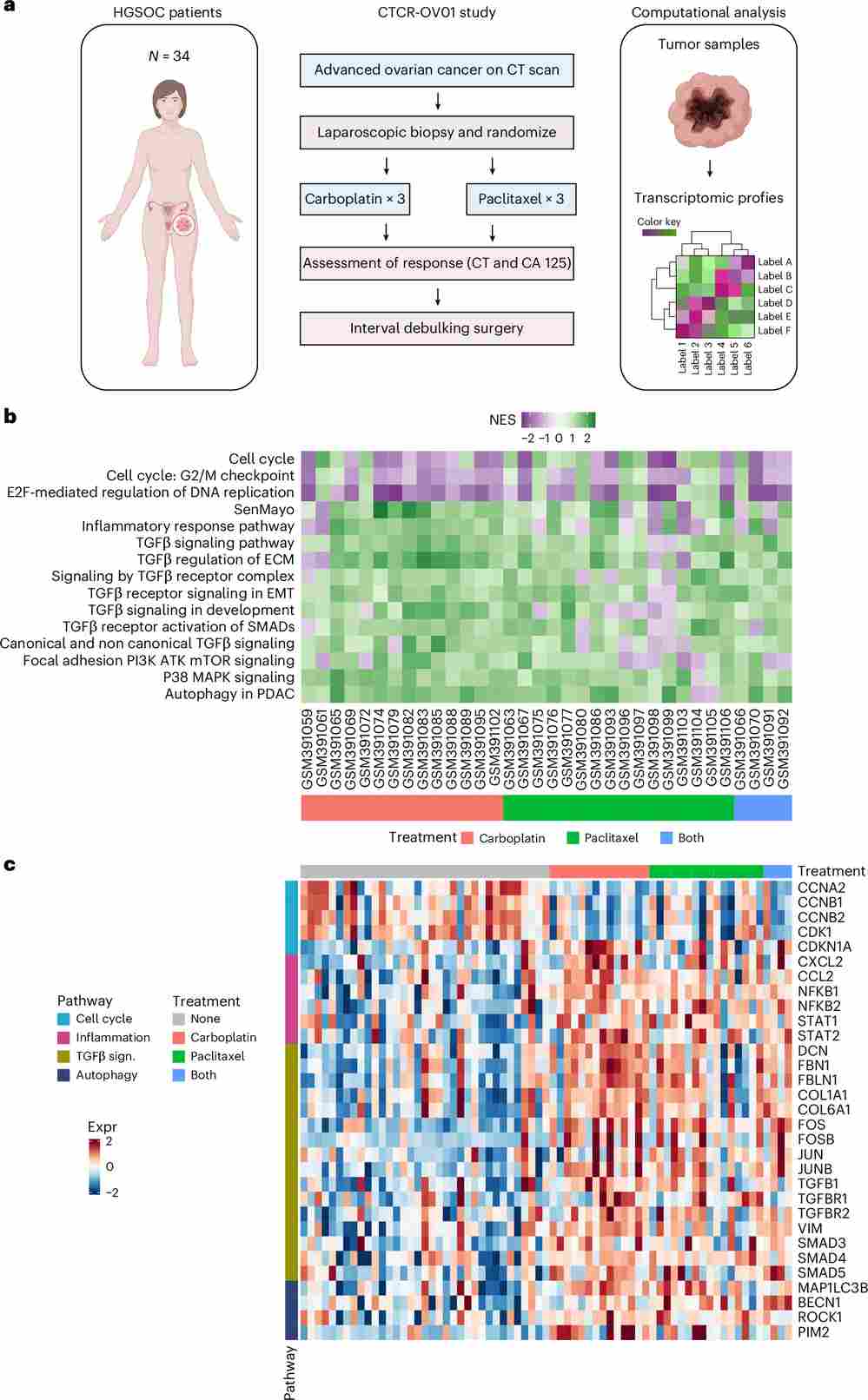
Figure 7. GSEA of microarray datasets from patients with ovarian cancer after chemotherapy treatment. (a) Schematic of the CTCR-OV01 study design. (b) Heatmap of normalized enrichment scores (NES) for 34 evaluable patients, showing enrichment in senescence and TGFβ pathways post-treatment. (c) Heatmap of gene expression z-scores for selected signaling pathway genes, stratified by pre- vs. post-treatment status and demonstrating consistent upregulation of TGFβ and mTOR signatures after platinum-based chemotherapy.
HGSOC Patient Cohort: IHC Validation of Senescence Markers
Complementing the transcriptomic data, a tissue microarray (TMA) comprising samples from HGSOC patients treated by interval primary surgery (IPS) or neoadjuvant chemotherapy (NACT) was subjected to multi-marker IHC analysis. NACT-treated specimens showed significantly higher p21, p16, pS6RP, and pAKT staining relative to IPS controls, confirming that platinum-based chemotherapy induces senescence and activates downstream survival signaling in ovarian cancer as well. Notably, pS6RP positivity was further associated with suboptimal surgical debulking, suggesting that SASP-driven mTOR activation may contribute to residual disease and unfavorable surgical outcomes. These findings collectively support galunisertib’s potential as a broad-spectrum senolytic adjunct across platinum-treated solid tumors.
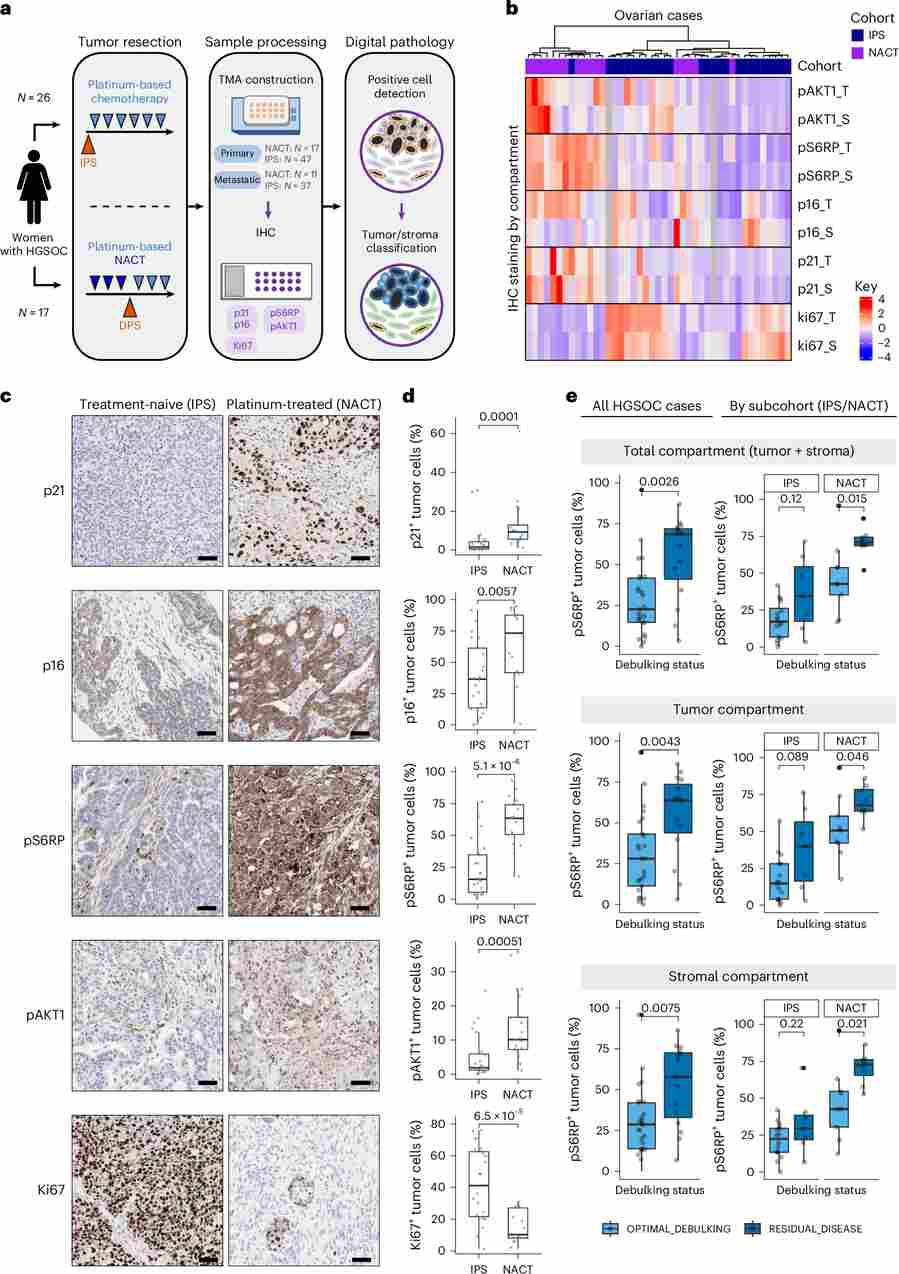
Figure 8. Senescence induction in patients with HGSOC after neoadjuvant treatment with platinum-based chemotherapy. (a) Study design schematic for HGSOC TMA cohort. (b) IHC staining heatmap by marker and region (S = stroma, T = tumor). (c) Representative IHC images of p21, p16, pS6RP, pAKT1, and Ki67 for IPS and NACT cohorts. (d) Quantification of IHC-positive cells in tumor regions. (e) pS6RP positivity stratified by surgical debulking outcome (optimal vs. residual disease) in IPS and NACT subgroups.
Ready to replicate or build upon this research? Our A549 cell line is available with guaranteed mycoplasma-free certification and passage-number documentation.
Request a quote now: A549 Cell Line / A549 Luciferase Cell Line
Implications for Combination Therapy
The study’s translational significance is substantial. Galunisertib (LY2157299) is an orally bioavailable TGFBR1 kinase inhibitor that has already completed Phase II clinical trials in hepatocellular carcinoma and pancreatic cancer, with an established safety profile. The current study provides a compelling mechanistic rationale for combining galunisertib with first-line platinum-based chemotherapy in NSCLC and HGSOC. By blocking TGFBR1 during or immediately after cisplatin treatment, the combination prevents surviving non-senescent cancer cells from co-opting the TGFβ-SASP signal for proliferative advantage, thereby closing a key escape route. Moreover, the identification of senolytics (e.g., ABT-737, navitoclax) as viable adjuncts to platinum therapy adds further options to dismantle the senescence-dependent resistance niche.
Conclusion
This Cambridge University study, published in Nature Aging (2026), establishes a mechanistic framework in which platinum-based chemotherapy creates a TGFβ-enriched senescent secretome that co-opts TGFBR1–AKT/mTOR signaling in residual lung and ovarian cancer cells to drive treatment resistance. A549-luc cells, by virtue of their bioluminescent tracking capability, were indispensable for demonstrating real-time tumor progression and therapeutic response in vivo. The study not only illuminates a critical vulnerability in chemotherapy-treated cancers but also positions galunisertib as a clinically actionable co-treatment partner. As combination immunotherapy and targeted therapy protocols evolve, integrating TGFBR1 inhibition into platinum-based regimens represents a promising strategy to improve durable response rates and patient survival outcomes.
References
Maciel-Baron, L.A., Reinius, B., et al. (2026). Treatment resistance to platinum-based chemotherapy in lung and ovarian cancer is driven by a targetable TGFβ senescent secretome. Nature Aging, 6(2), 368–392. https://doi.org/10.1038/s43587-025-01054-2

