Engineering tLyp-1-Modified Exosomes to Silence oncogenic circRNA in A549 Lung Cancer Models
Introduction
Lung adenocarcinoma (LUAD) is the most prevalent subtype of lung cancer, with metastasis being the primary cause of mortality and a 5-year survival rate below 20%. Circular RNAs (circRNAs) have emerged as critical regulators of tumor metastasis due to their high stability and tissue specificity. CircRAPGEF5 is overexpressed in LUAD and promotes metastasis; however, its precise mechanism and effective targeted intervention strategies have remained unclear. Exosomes offer advantages as nucleic acid delivery vehicles—low immunogenicity and high biocompatibility—but unmodified exosomes suffer from poor targeting, limiting clinical translation. The tLyp-1 peptide can specifically bind to neuropilin-1 (NRP1)/NRP2, which are highly expressed in LUAD cells, enabling tumor-targeted delivery of therapeutic cargo.
Studying exosome-based nucleic acid delivery in non-small cell lung cancer? Our validated wild-type A549 cell line is A powerful tool for LUAD research.
Contact us now: A549 Cell Line / A549 Luciferase Cell Line
Construction and Characterization of tLyp-1-EXO-circRAPGEF5 ASO
The research team engineered a targeted exosome delivery system by exploiting the selective overexpression of NRP1 and NRP2 on LUAD cell surfaces. Exosomes displaying streptavidin (mSA) were generated by transiently transfecting HEK293T cells, then purified by differential centrifugation and 0.22 μm filtration. The tLyp-1 peptide was conjugated to the exosome surface via streptavidin–biotin chemistry, and modification efficiency was verified by nanoscale flow cytometry. circRAPGEF5 ASO labeled with Cy3 was loaded into modified exosomes by electroporation (250 V, 125 μF), achieving a loading efficiency of approximately 60%.
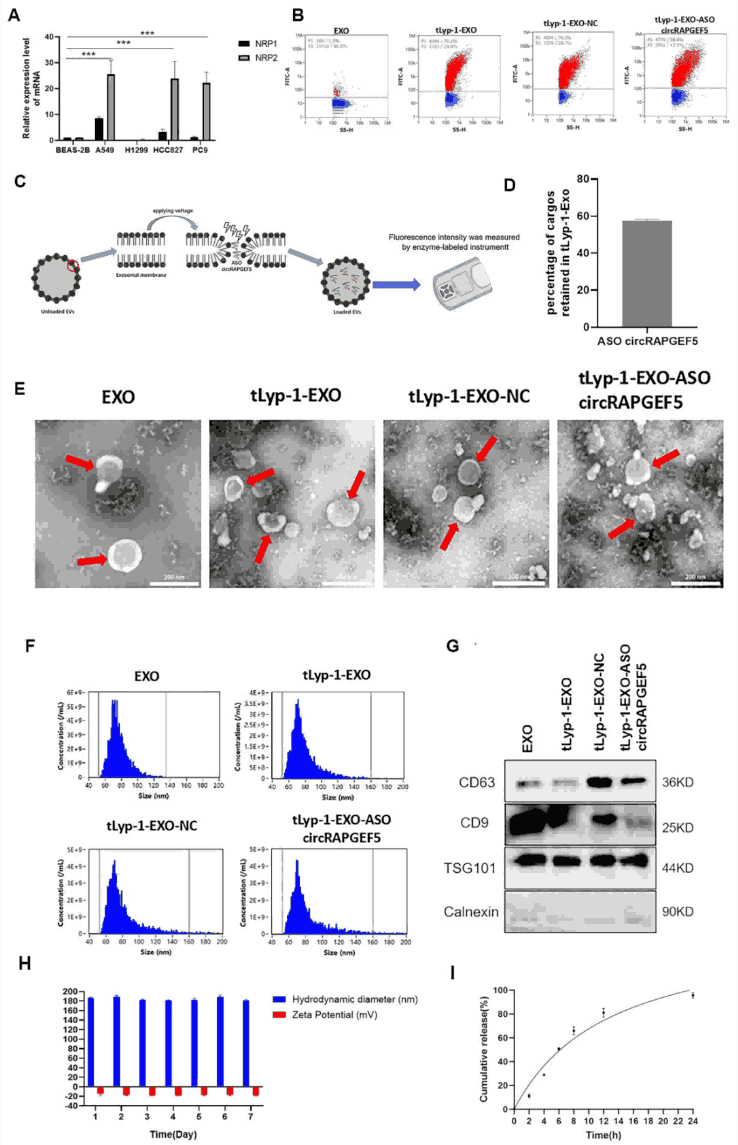
Figure 1. Construction and characterization of tLyp-1-EXO-circRAPGEF5 ASO. (A) NRP1/NRP2 overexpression in LUAD cells (A549, HCC827, etc.) verified by Western blot. (B) Nanoscale flow cytometry confirming tLyp-1 peptide conjugation on exosome surface. (C-D) Cy3-labeled circRAPGEF5 ASO successfully loaded, ~60% loading efficiency. (E) TEM showing intact bilayer cup-shaped morphology post-modification. (F) NTA showing size distribution of 52–160 nm with no significant change after modification. (G) Western blot confirming CD63/CD9/TSG101 expression and absence of Calnexin. (H) Stable particle size (181–189.1 nm) in PBS over 7 days. (I) ~95% ASO release within 24 hours, confirming delivery potential.
Structural integrity was confirmed by TEM showing the characteristic bilayer membrane morphology, while NTA indicated a size distribution of 52–160 nm with negligible changes following peptide modification and ASO loading. Western blot analysis verified the expression of canonical exosome markers (CD63, CD9, TSG101) and the absence of the endoplasmic reticulum marker Calnexin, confirming high purity. Stability assessment demonstrated consistent particle size (181–189.1 nm) in PBS over 7 days, and in vitro release kinetics revealed approximately 95% ASO release within 24 hours, establishing the therapeutic delivery potential of this platform.
In Vitro and In Vivo Targeting Efficiency and ASO Delivery
To validate targeting specificity, DiO-labeled tLyp-1-modified exosomes were co-cultured with A549 and HCC827 cells. Laser scanning confocal microscopy revealed significantly higher cellular uptake of tLyp-1-EXO compared to unmodified exosomes, reaching peak internalization at 12 hours. Cy3-labeled ASO was confirmed to be successfully delivered to the intracellular compartment, and RT-qPCR demonstrated significant downregulation of circRAPGEF5 expression in cells treated with tLyp-1-EXO-circRAPGEF5 ASO.
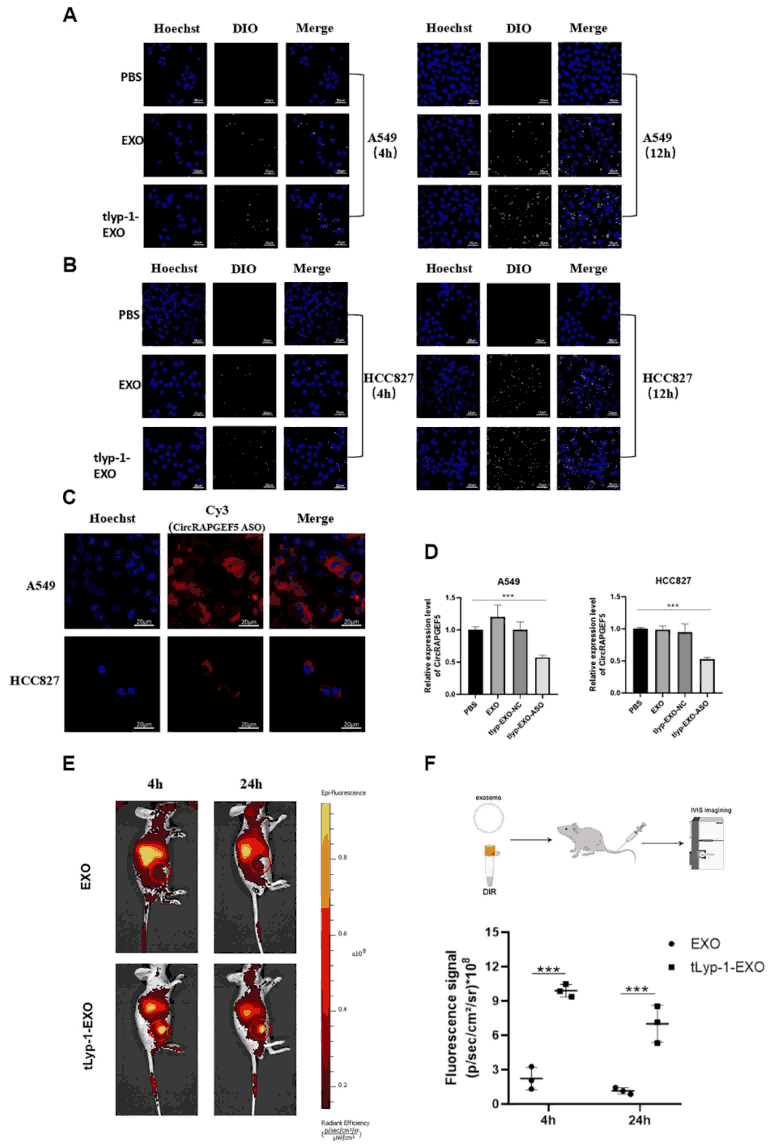
Figure 2. In vitro and in vivo targeting efficiency and ASO delivery. (A-B) Confocal microscopy showing enhanced uptake of DiO-labeled tLyp-1-EXO by A549/HCC827 cells, peaking at 12 h. (C) Intracellular delivery of Cy3-labeled ASO confirmed by confocal imaging. (D) RT-qPCR showing circRAPGEF5 downregulation following tLyp-1-EXO-ASO treatment. (E-F) IVIS imaging showing superior tumor accumulation and retention of DiR-labeled tLyp-1-EXO versus unmodified exosomes after tail vein injection.
In vivo biodistribution was assessed using DiR-labeled exosomes in A549-Luc tumor-bearing nude mice. IVIS live imaging demonstrated significantly greater accumulation and prolonged retention of tLyp-1-modified exosomes at tumor sites compared to unmodified counterparts following tail vein injection, establishing the in vivo targeting capability of this engineered system.
In Vitro Anti-Tumor Effects of tLyp-1-EXO-circRAPGEF5 ASO
The functional impact of tLyp-1-EXO-circRAPGEF5 ASO on LUAD cells was systematically evaluated across multiple oncogenic behaviors. CCK-8 proliferation assays showed significant inhibition of A549 and HCC827 cell growth compared to controls. Colony formation assays confirmed a marked reduction in clonogenic capacity. Transwell migration and invasion assays demonstrated substantially decreased motility and invasiveness, while Western blot analysis revealed coordinated downregulation of EMT markers N-cadherin and Vimentin—proteins associated with mesenchymal transition and metastatic potential. Collectively, these results establish that tLyp-1-EXO-circRAPGEF5 ASO effectively inhibits proliferation, migration, invasion, and EMT in LUAD cells.
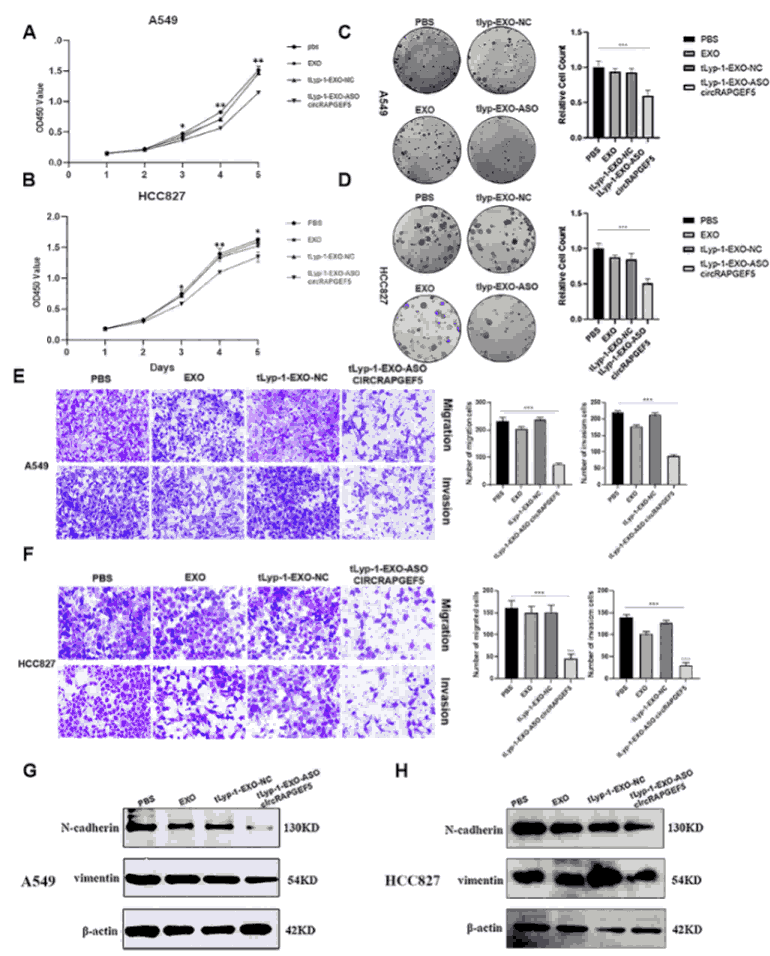
Figure 3. In vitro anti-tumor effects. (A-B) CCK-8 assays showing inhibited A549/HCC827 proliferation. (C-D) Colony formation assays confirming reduced clonogenic capacity. (E-F) Transwell assays showing suppressed migration and invasion. (G-H) Western blot showing downregulation of EMT markers N-cadherin and Vimentin.
For reliable anti-metastasis assays in lung cancer drug development, our fully authenticated A549 cells provide consistent baseline performance across proliferation, migration, and invasion readouts.
Request a quote now: A549 Cell Line / A549 Luciferase Cell Line
CircRAPGEF5 Acts as a miR-570-3p Sponge to Promote LUAD Progression
Bioinformatic prediction identified a potential binding site between circRAPGEF5 and miR-570-3p, which was validated by dual-luciferase reporter assays confirming direct interaction. MiR-570-3p expression was found to be significantly lower in LUAD cells compared to normal bronchial epithelial cells. Forced overexpression of circRAPGEF5 downregulated miR-570-3p, while its knockdown led to upregulation, confirming the sponge relationship. Functional experiments showed that miR-570-3p mimics suppressed LUAD cell proliferation, migration, and invasion, whereas miR-570-3p inhibitors promoted these oncogenic behaviors.
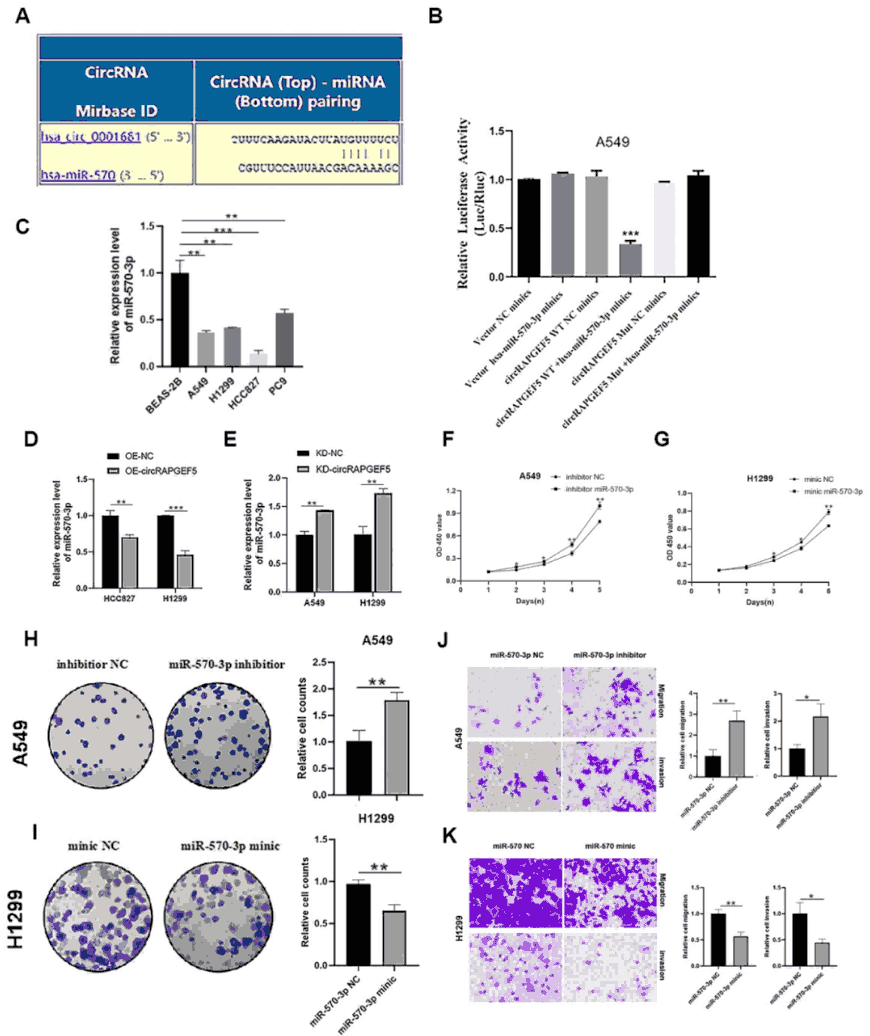
Figure 4. CircRAPGEF5 sponges miR-570-3p in LUAD. (A) Bioinformatic prediction of circRAPGEF5–miR-570-3p binding site. (B) Dual-luciferase reporter assay confirming direct binding. (C) Lower miR-570-3p expression in LUAD versus normal cells. (D-E) CircRAPGEF5 overexpression downregulates and knockdown upregulates miR-570-3p. (F-K) miR-570-3p mimics suppress, and inhibitors promote, LUAD cell proliferation, migration, and invasion.
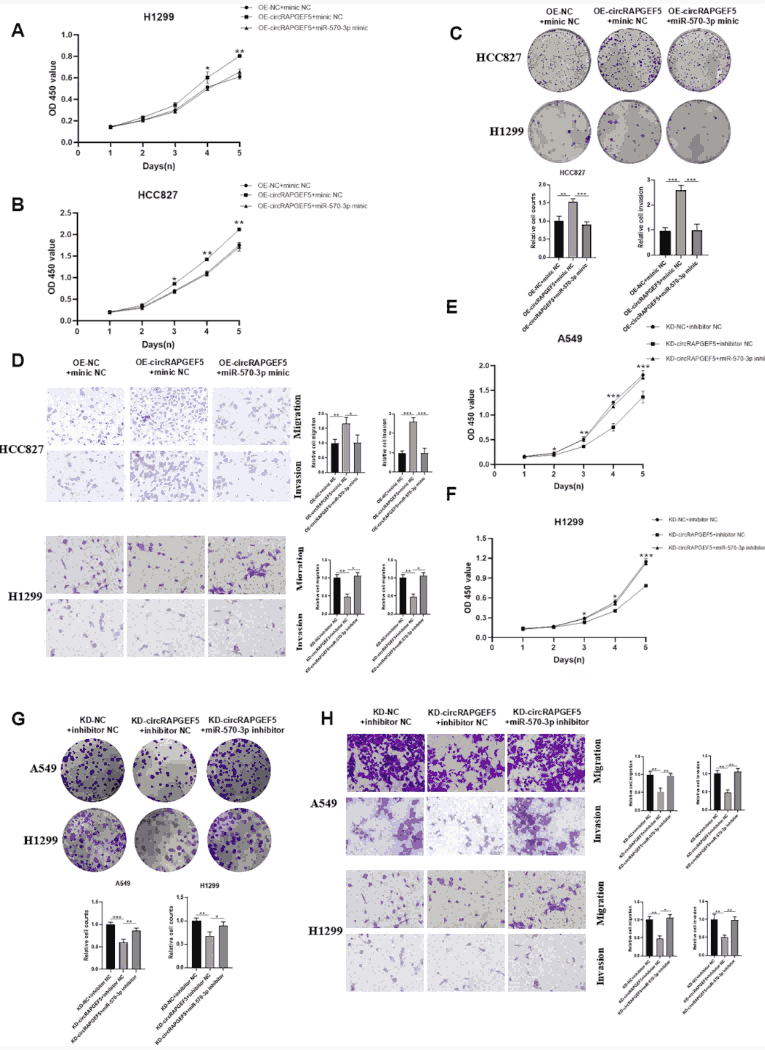
Figure 5. Rescue experiments confirming ceRNA mechanism. (A-D) miR-570-3p mimics reverse the pro-tumorigenic effects of circRAPGEF5 overexpression. (E-H) miR-570-3p inhibitors reverse the tumor-suppressive effects of circRAPGEF5 knockdown, confirming that circRAPGEF5 drives LUAD progression by sequestering miR-570-3p.
miR-570-3p Directly Regulates SPP1 as a Downstream Effector
To identify the downstream target mediating the circRAPGEF5/miR-570-3p axis, researchers performed dual-luciferase reporter assays confirming that miR-570-3p directly binds the 3'UTR of SPP1. miR-570-3p mimics downregulated SPP1 expression, while inhibitors upregulated it. SPP1 overexpression promoted LUAD cell proliferation, colony formation, migration, and invasion. Furthermore, circRAPGEF5 overexpression upregulated SPP1 and knockdown suppressed it, and SPP1 overexpression could rescue the tumor-suppressive phenotype induced by circRAPGEF5 knockdown, confirming that SPP1 is a bona fide target of this regulatory axis.
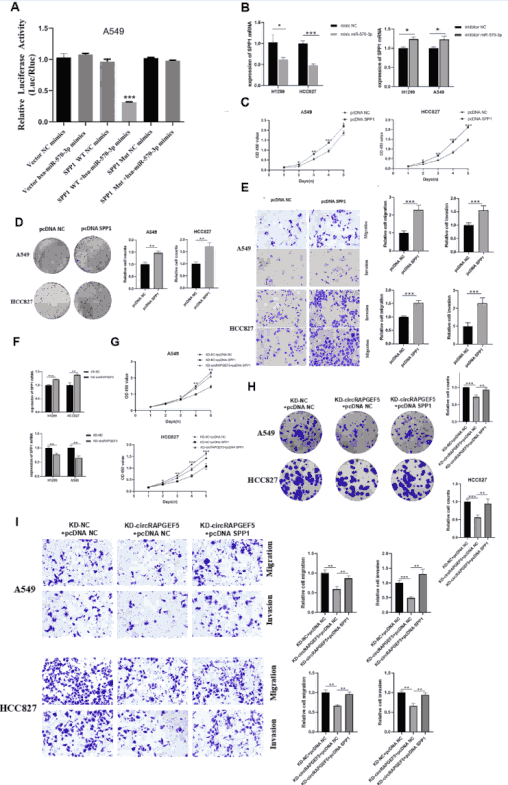
Figure 6. miR-570-3p directly targets SPP1. (A) Dual-luciferase reporter confirming miR-570-3p binding to SPP1 3'UTR. (B) miR-570-3p mimics downregulate and inhibitors upregulate SPP1. (C-E) SPP1 overexpression promotes LUAD proliferation, colony formation, migration, and invasion. (F) CircRAPGEF5 regulates SPP1 expression. (G-I) SPP1 overexpression reverses the tumor-suppressive effects of circRAPGEF5 knockdown.
Validation of the circRAPGEF5/miR-570-3p/SPP1 Axis
To verify the integrity of the complete signaling cascade, researchers performed systematic rescue experiments. miR-570-3p mimics reversed the SPP1 upregulation induced by circRAPGEF5 overexpression, while miR-570-3p inhibitors reversed the SPP1 downregulation caused by circRAPGEF5 knockdown. Critically, treatment with tLyp-1-EXO-circRAPGEF5 ASO resulted in upregulation of miR-570-3p and downregulation of SPP1 in LUAD cells, confirming that the therapeutic construct exerts its anti-tumor effects through the circRAPGEF5/miR-570-3p/SPP1 regulatory axis.
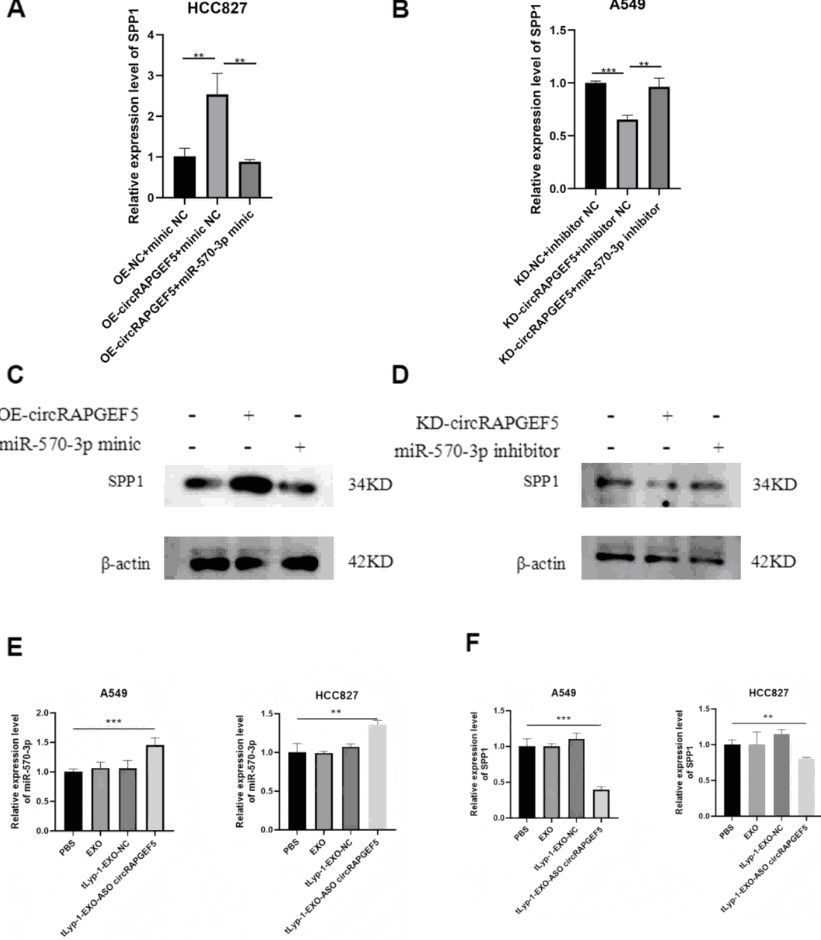
Figure 7. Pathway integrity validation. (A-C) miR-570-3p mimics reverse circRAPGEF5-overexpression-induced SPP1 upregulation. (B-D) miR-570-3p inhibitors reverse circRAPGEF5-knockdown-induced SPP1 downregulation. (E-F) tLyp-1-EXO-circRAPGEF5 ASO treatment upregulates miR-570-3p and downregulates SPP1 in LUAD cells, confirming on-target pathway modulation.
In Vivo Anti-Tumor Efficacy and Biosafety Assessment
In vivo therapeutic efficacy was evaluated in nude mice bearing tail-vein-injected A549-Luc lung metastasis models. Animals were randomized into PBS, tLyp-1-EXO-NC, and tLyp-1-EXO-circRAPGEF5 ASO groups, receiving six intravenous administrations every 4 days. IVIS bioluminescence imaging demonstrated significantly reduced tumor signal intensity in the treatment group. Post-sacrifice analysis confirmed markedly fewer lung metastatic nodules and less histological lung damage by H&E staining in the treatment group compared to controls.
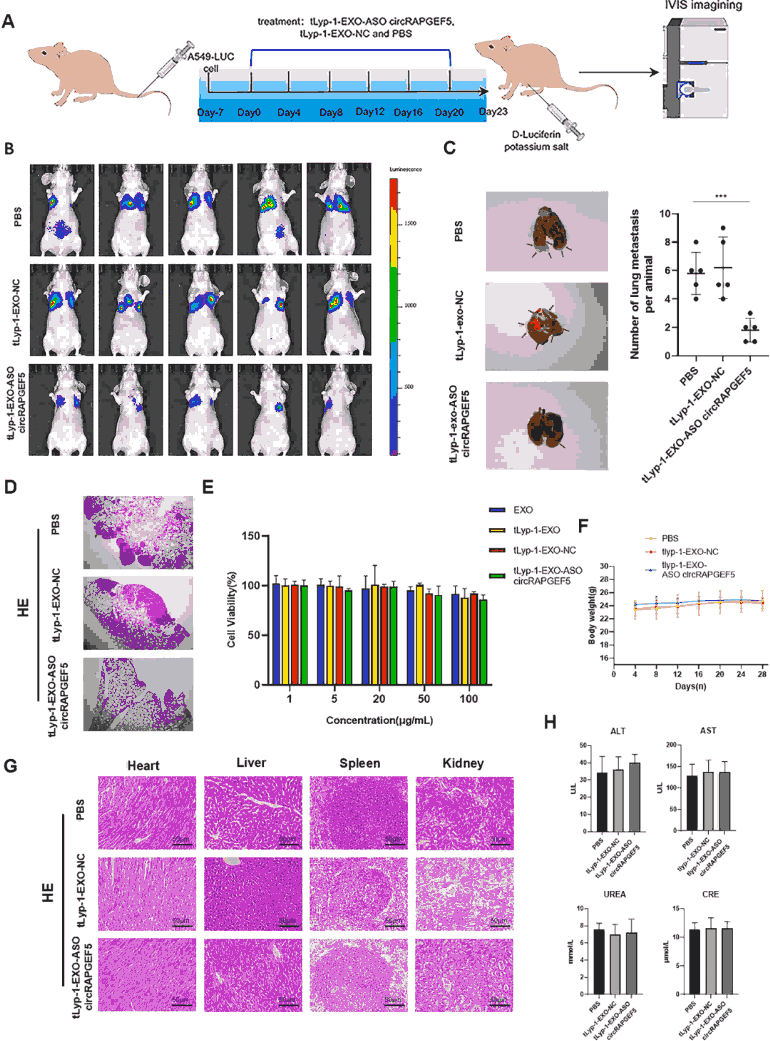
Figure 8. In vivo anti-tumor efficacy and biosafety. (A) Animal experimental design schematic. (B) IVIS imaging showing reduced tumor bioluminescence in tLyp-1-EXO-circRAPGEF5 ASO group. (C-D) Reduced lung metastatic nodule count and attenuated lung tissue injury. (E) CCK-8 showing no significant cytotoxicity in BEAS-2B normal cells. (F) Stable body weight across all groups. (G) H&E staining showing no pathological damage in heart, liver, spleen, and kidney. (H) Normal serum ALT/AST/UREA/CRE levels confirming systemic biosafety.
Biosafety assessment was comprehensive: the tLyp-1-EXO-circRAPGEF5 ASO formulation showed no appreciable cytotoxicity in normal human bronchial epithelial BEAS-2B cells. In vivo, mouse body weight remained stable throughout treatment. H&E staining of major organs (heart, liver, spleen, kidney) revealed no pathological changes, and serum biochemical markers (ALT, AST, UREA, CRE) were within normal ranges, collectively establishing an excellent biosafety profile.
For in vivo metastasis models requiring consistent bioluminescent imaging readouts, our rigorously characterized A549 cells deliver reproducible lung metastasis data.
View more: A549 Cell Line / A549 Luciferase Cell Line
Conclusion
This study successfully developed a tLyp-1-modified exosome delivery system capable of efficiently targeting LUAD cells and delivering circRAPGEF5 ASO to significantly silence circRAPGEF5 expression. Mechanistically, circRAPGEF5 sequesters miR-570-3p via ceRNA activity, releasing SPP1 from post-transcriptional suppression; tLyp-1-EXO-circRAPGEF5 ASO reverses this pathway, inhibiting LUAD cell proliferation, migration, invasion, and EMT. In vivo experiments confirm effective suppression of LUAD metastasis with favorable biosafety. This work not only elucidates the circRAPGEF5/miR-570-3p/SPP1 axis in LUAD metastasis but also provides a highly targeted, safe nucleic acid drug delivery strategy, offering new therapeutic targets and tools for clinical LUAD treatment.
References
Hu T, Zhou C, Chen Y, Fu J, Dai Y, Ling L, Chen Y, Lu Y, He Y, Chen J, Wang Y. TLyP-1-modified exosome-mediated delivery of circRAPGEF5 ASO inhibits lung adenocarcinoma metastasis via regulating the miR-570-3p/SPP1 axis. Materials Today Bio. 2025; 35: 102416. doi:10.1016/j.mtbio.2025.102416

