Mechanistic Insights into Tumor-Antigen-Specific T Cell Deployment Using EO771-OVA Breast Cancer Models
Introduction
Immune checkpoint blockade therapy has achieved remarkable success in cancer treatment by reactivating effector immune cells, yet some patients remain unresponsive due to the "cold" tumor microenvironment phenotype. Effector immune cell deployment (EICD) represents a critical component of immunosurveillance, and defects in this process lead to tumor immune escape. However, the long-distance regulatory roles of adaptive immunity in distant metastasis and their underlying mechanisms have remained unclear. This study aims to elucidate the function of tumor-antigen-specific CD103+CD8+T cells in breast cancer lung metastasis, clarifying the molecular mechanisms governing their initiation, recruitment, and functional regulation while revealing the impact of tumor microenvironment on their fate.
For studying long-distance T cell deployment and metastasis, our extensively characterized EO771 breast cancer model provides an ideal system.
Order Now: EO771-OVA Cell Line / EO771-Luc Cell Line
CD103+CD8+T Cell Abundance in Tumor-Draining Lymph Nodes Correlates with Reduced Lung Metastasis
Analysis of CD8+T cell subpopulations in tumor-draining lymph nodes (TDLNs) from 533 newly diagnosed breast cancer patients revealed that the abundance of CD103+CD8+T cells—a tissue-resident memory phenotype—significantly correlated with reduced lung metastasis risk. Importantly, central memory T cells (TCM) and effector memory T cells (TEM) within the CD8+ population showed no such association. Higher abundance of CD103+CD8+T cells predicted longer lung-specific metastasis-free survival, and this relationship held true across all breast cancer subtypes. Furthermore, infiltration of these cells in metastatic lung tissue was negatively correlated with metastatic burden.
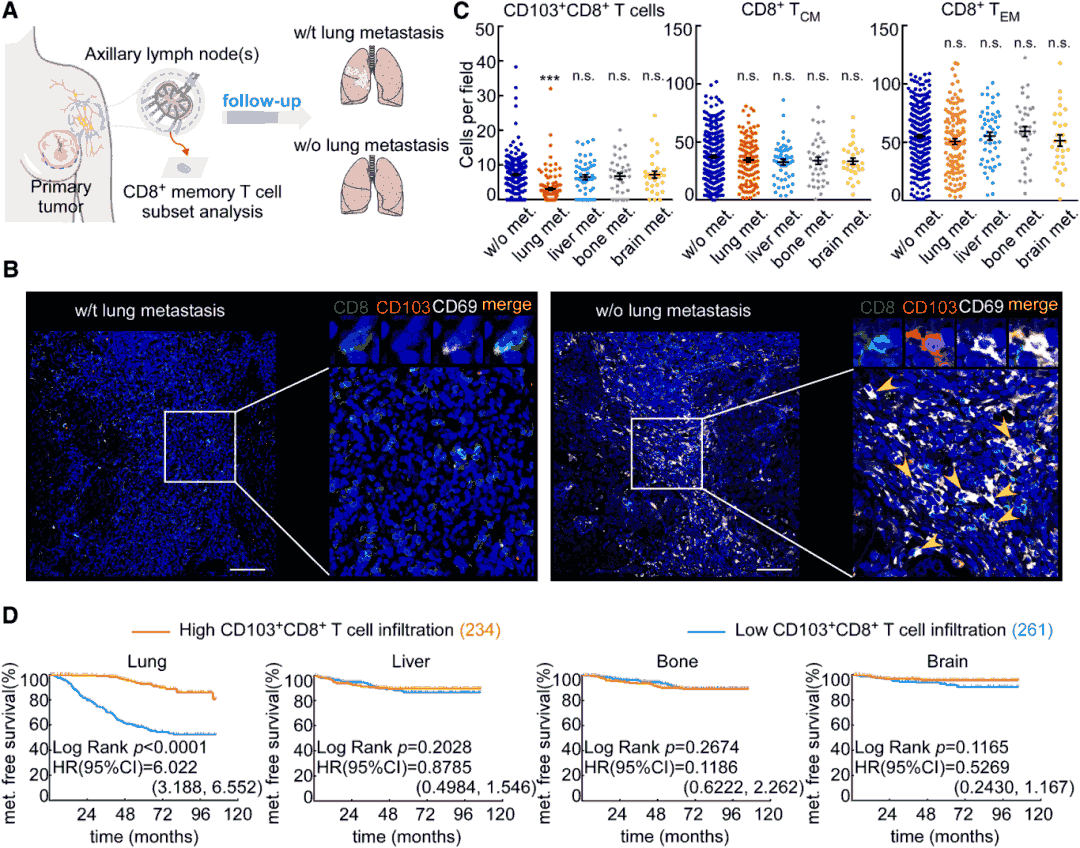
Figure 1. CD103+CD8+T cells in TDLNs correlate with reduced lung metastasis.
CD103+CD8+T Cells Accumulate in Metastasis-Free Lungs Before Declining Prior to Metastasis
In EO771 breast cancer mouse models, CD103+CD8+T cells began accumulating in lung tissue at 2 weeks post-tumor inoculation, reaching peak levels at 21-28 days (representing 51.2% of CD8+T cells), followed by a sharp decline prior to macroscopic metastasis appearance. These cells were predominantly located within lung tissue rather than blood vessels in metastasis-preceding lungs and exhibited enhanced proliferative capacity, cytokine secretion (granzyme B, perforin, IFN-γ), and cytotoxic activity. Consistent findings were obtained in 4T1 breast cancer and B16F10 melanoma models. In clinical samples, CD103+CD8+T cell infiltration was significantly higher in cancer-distant lung tissue (>10 cm from metastatic foci) compared to peritumoral and metastatic tissues in patients with lung metastasis.
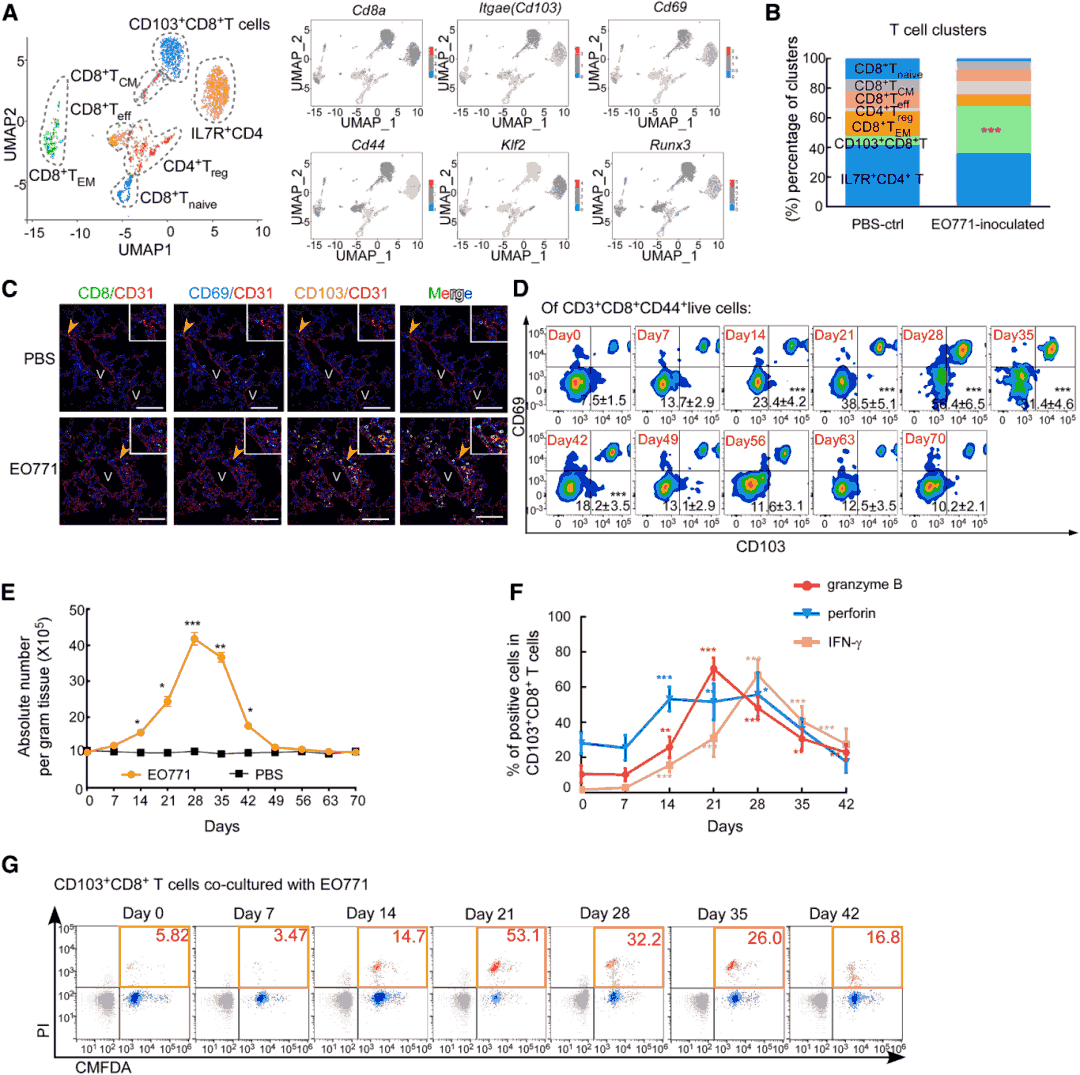
Figure 2. Temporal dynamics of CD103+CD8+T cells in pre-metastatic lungs.
CD103+CD8+T Cells Suppress Lung Metastasis Through Tumor Antigen-Specific Cytotoxicity
Using conditional depletion mouse models (Cd8Cre/+ItgaeloxP/+DTR-GFP), researchers demonstrated that specific elimination of CD103+CD8+T cells significantly exacerbated breast cancer and melanoma lung metastasis while shortening mouse survival. Conversely, adoptive transfer of these cells (but not TCM, TEM, or effector T cells) significantly reduced lung metastatic burden. Antigen specificity experiments revealed that only CD103+CD8+T cells induced by EO771-OVA tumors effectively recognized the OVA257-264 peptide, specifically killed EO771-OVA cells, and secreted IFN-γ, confirming that their anti-metastatic effect depends on tumor antigen specificity.
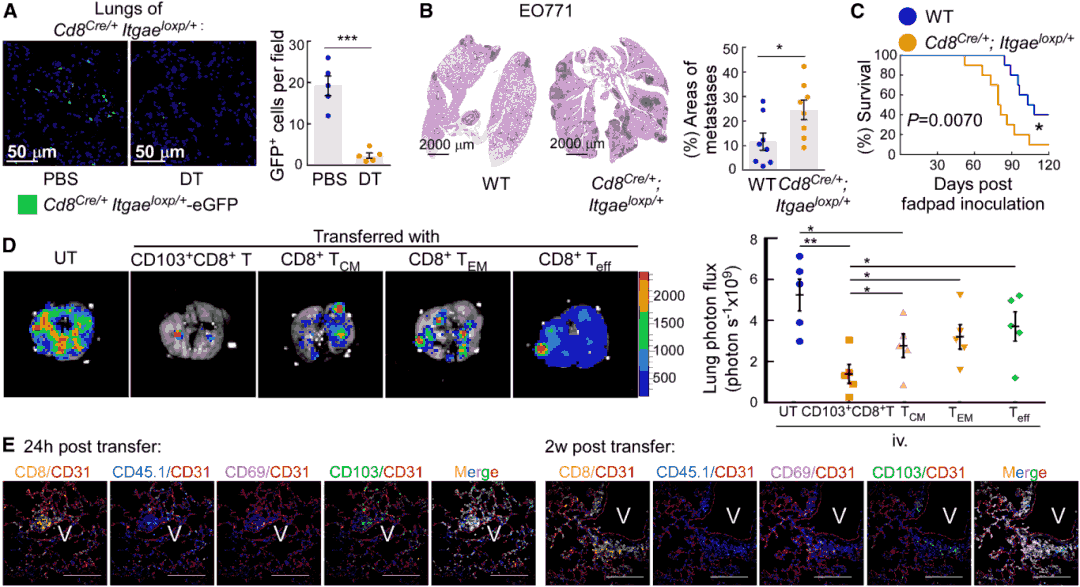
Figure 3. CD103+CD8+T cells inhibit lung metastasis in vivo. (a-b) CD103+CD8+T cell depletion increases lung metastasis and reduces survival in EO771 and B16F10 models. (c-d) Adoptive transfer of CD103+CD8+T cells reduces metastatic burden. (e) Antigen specificity assay showing OVA-specific recognition and killing of EO771-OVA cells.
Tumor-Specific CD103+CD8+T Cells Are Primed in TDLNs and Recruited to Lung Before Metastasis
OT-I T cell tracking experiments demonstrated that tumor antigen-specific CD103+CD8+T cells first accumulate in TDLNs (1 week post-inoculation) and are only detected in pre-metastatic lungs at 3 weeks. Kaede photoconversion experiments confirmed that CD103+CD8+T cells infiltrating the lung originate from TDLNs rather than primary tumors or other lymph nodes. Dendritic cells (DCs) in TDLNs effectively primed CD103+CD8+T cells, and their priming capacity increased with metastatic progression. Inhibition of lymphocyte egress from TDLNs using FTY720 significantly reduced CD103+CD8+T cell infiltration in lungs, confirming that these cells must circulate from TDLNs to exert their function in the lung.
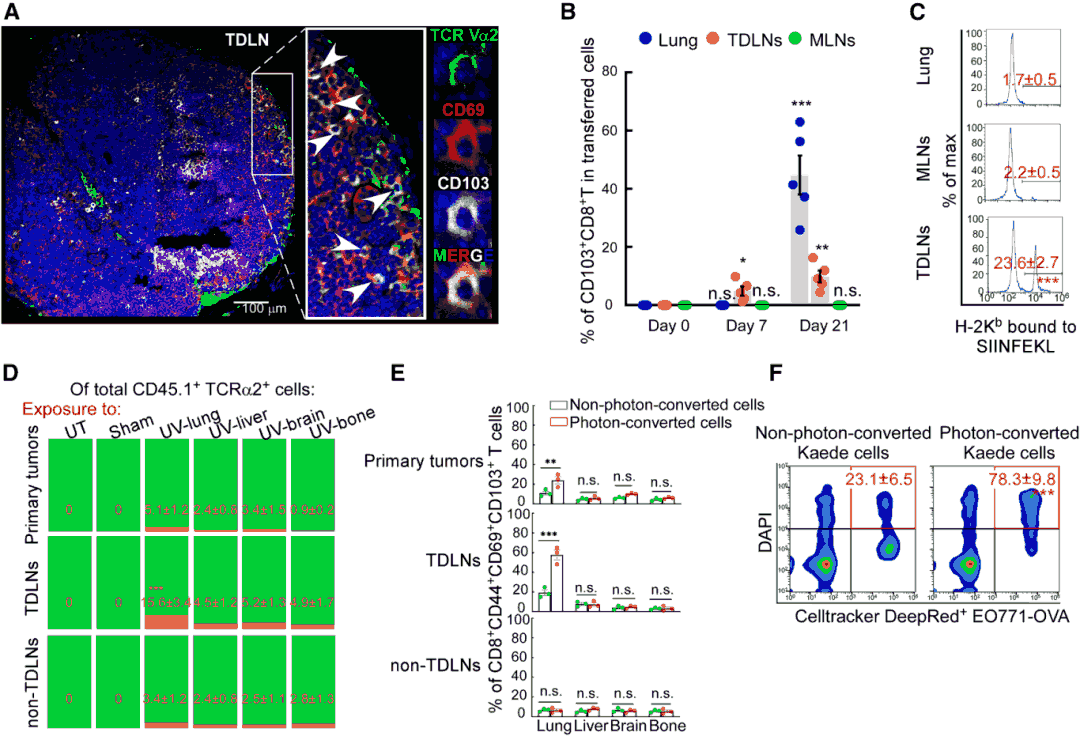
Figure 4. Tumor-specific CD103+CD8+T cells are primed in TDLNs and recruited to lungs.
Alveolar Macrophages Drive Lung CD103+CD8+T Cell Deployment via CCL25/CCR9 and Induce Their Death via IDO1
Single-cell RNA sequencing analysis revealed that CD103+CD8+T cells specifically express CCR9, while their ligand CCL25 is primarily secreted by alveolar macrophages (AMs). CCL25 secreted by AMs is elevated during early metastasis and declines in late stages, consistent with CD103+CD8+T cell infiltration dynamics. Neutralizing CCL25 or CCR9 blocked CD103+CD8+T cell recruitment to lung and accelerated metastasis. During late metastasis, AMs highly express IDO1, reducing CD103+CD8+T cell function by inhibiting cytokine secretion and promoting apoptosis. Treatment with IDO1 inhibitor 1-MT significantly increased CD103+CD8+T cell infiltration in lungs, reduced metastasis, and prolonged survival, but this effect was absent in AM-deficient Csf2-/- mice.
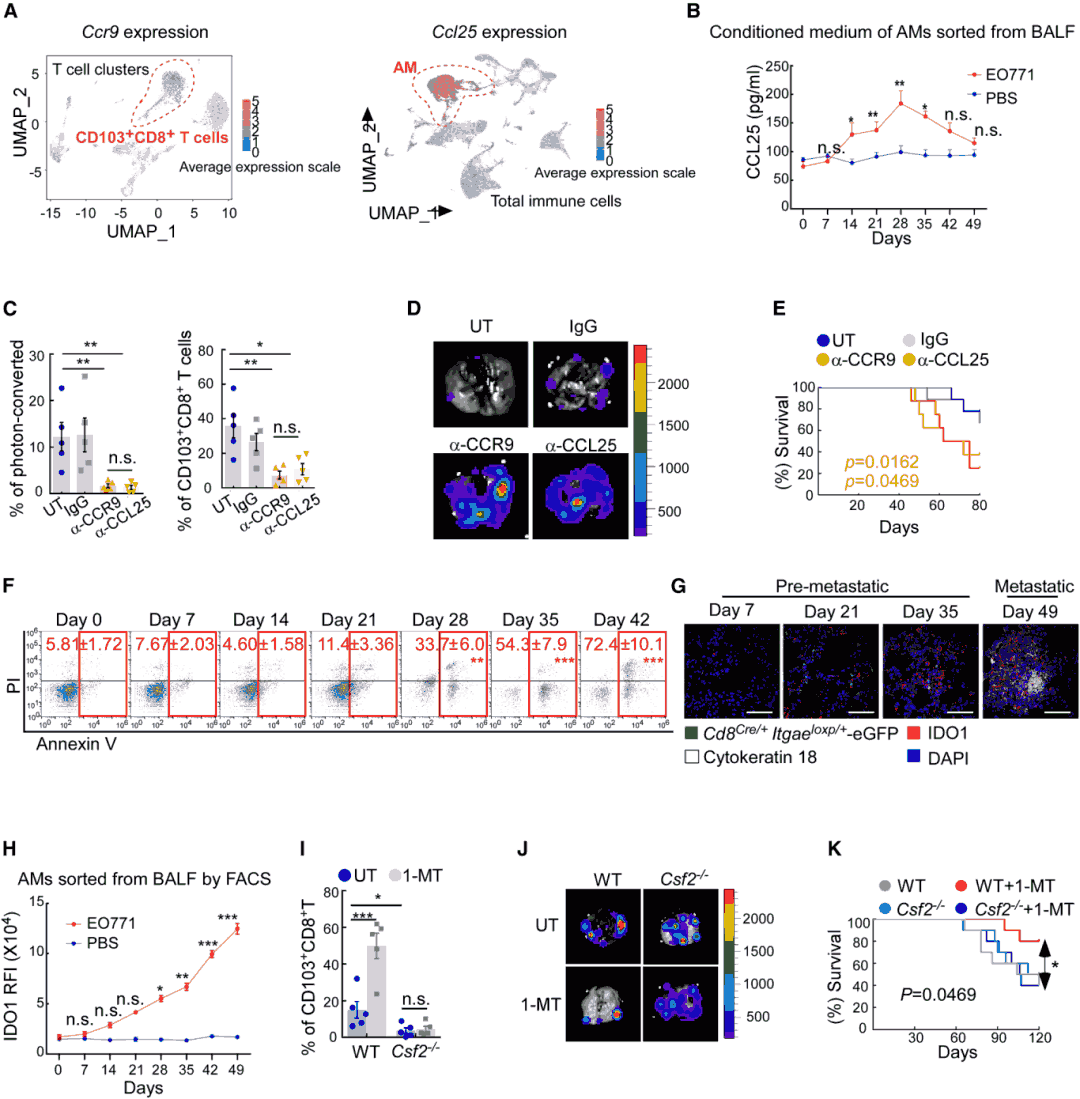
Figure 5. AMs regulate CD103+CD8+T cell deployment and survival.
Tumor-Derived Extracellular Vesicles Differentially Polarize AMs in a Stage-Dependent Manner
Extracellular vesicles (EVs) released by tumors during early and late stages (early-EV, late-EV) showed no significant differences in morphology or diameter but carried distinct cargo: early-EVs were enriched in proteins (such as TNF-α), while late-EVs contained abundant double-stranded DNA. Early-EVs induced AMs to secrete CCL25 through NF-κB pathway activation, whereas late-EVs induced AMs to express IDO1 via AIM2 pathway activation. In vivo experiments confirmed that early-EV injection increased lung CD103+CD8+T cell infiltration and reduced metastasis, while late-EVs had the opposite effect. These effects could be reversed by blocking EV surface molecules ITGα6/ITGβ1 (lung-targeting uptake molecules).
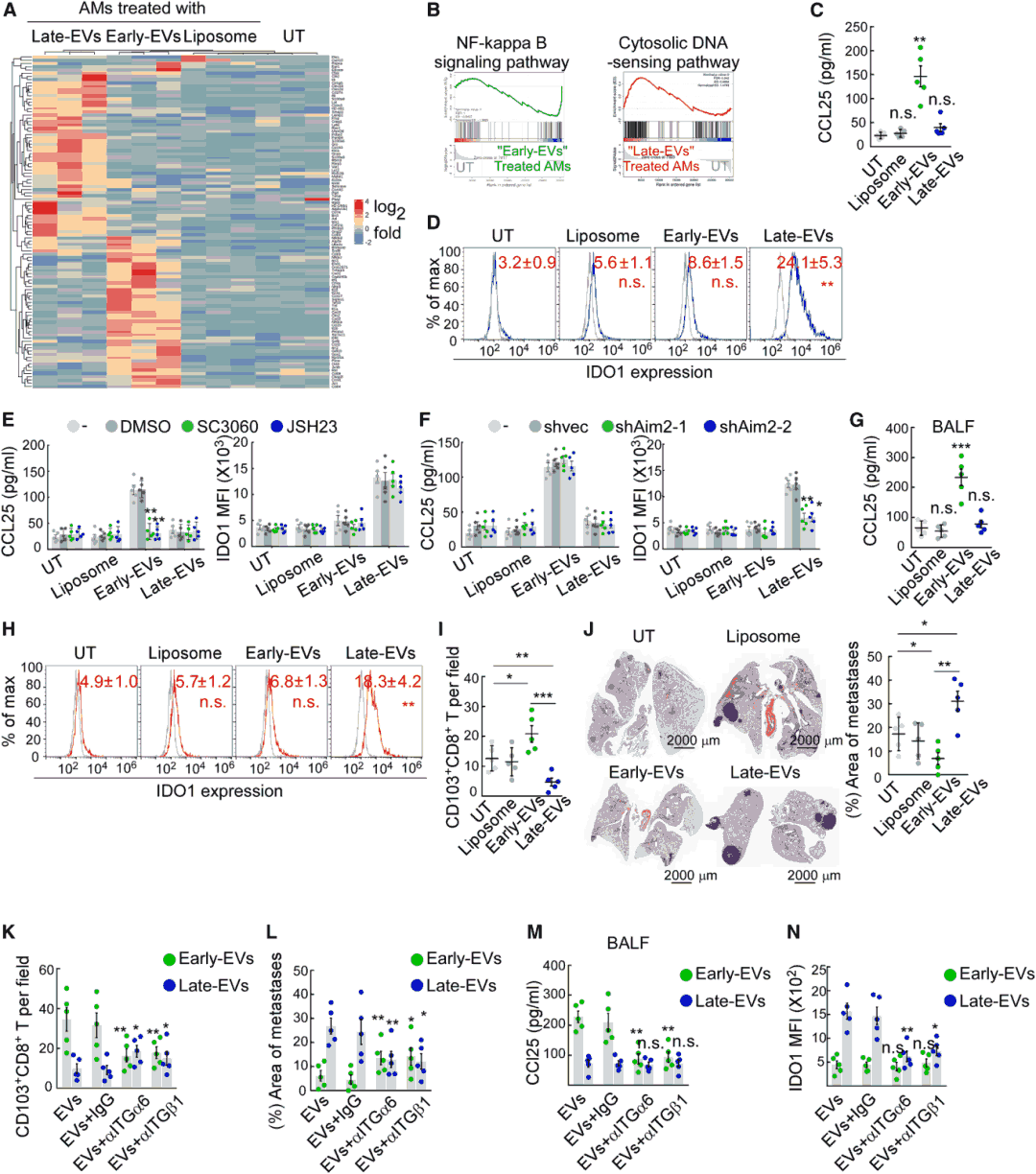
Figure 6. Tumor-derived EVs differentially polarize AMs across metastatic stages.
Discussion: Long-Distance Immunosurveillance and Pre-Metastatic Niche Remodeling
This study provides the first comprehensive characterization of long-distance immunosurveillance by CD103+CD8+T cells in breast cancer lung metastasis. The findings reveal that tumor antigen-specific CD103+CD8+T cells are primed in tumor-draining lymph nodes, then recruited to lung tissue through CCL25/CCR9 signaling to suppress breast cancer lung metastasis via tumor antigen-specific cytotoxicity. Tumor-derived EVs at different stages dynamically regulate the recruitment and survival of these cells through stage-specific cargo that differentially polarizes alveolar macrophages. This work elucidates a novel mechanism of pre-metastatic microenvironment remodeling and suggests that targeting IDO1 or enhancing CCL25/CCR9-mediated cell recruitment may reconstruct effector immune cell deployment (EICD), providing new therapeutic strategies for preventing breast cancer lung metastasis.
For advanced studies of breast cancer metastasis and T cell trafficking, our thoroughly validated EO771 cell lines offer reliable in vivo and in vitro systems.
Order Now: EO771-OVA Cell Line / EO771-Luc Cell Line
References
Xing Y, Zhou Y, Wang R, et al. Long-range deployment of tumor-antigen-specific cytotoxic T lymphocytes inhibits lung metastasis of breast cancer. Dev Cell. Published online August 22, 2025. doi:10.1016/j.devcel.2025.08.003

