HepG2-Luc: NIR-II Fluorescence-Guided Liver Cancer Surgery via HDAC6-Targeted Molecular Imaging
Introduction
Hepatocellular carcinoma (HCC) ranks as the third leading cause of cancer-related mortality worldwide. Surgical resection remains the only potentially curative option for eligible patients, yet post-operative recurrence exceeds 70% within five years due to residual microscopic disease invisible under white-light illumination. The critical unmet need is an intraoperative imaging modality capable of identifying sub-millimeter tumor foci in real time.
Histone deacetylase 6 (HDAC6) has emerged as a promising imaging biomarker, showing significant overexpression in HCC tissues while remaining minimally expressed in healthy liver parenchyma. The second near-infrared window (NIR-II, 1000-1700 nm) offers superior tissue penetration and signal-to-background ratio compared to first-window fluorophores. Wang et al. developed SeCF3-IRD800, a small-molecule HDAC6-targeted probe operating in the NIR-II window, and validated its performance across preclinical models including HepG2-Luc orthotopic xenografts.
Contact us to discuss your research needs. Our HepG2-Luc luciferase stable cell line provides consistent bioluminescence signal for your HCC in vivo imaging studies — request a quote now to get started.
Order now: HepG2 Cell Line / HepG2 Luciferase Cell Line / HepG2-Luc-GFP Cell Line
HDAC6 Overexpression in Hepatocellular Carcinoma
The investigators systematically evaluated HDAC6 expression across a panel of liver cancer cell lines and clinical specimens. Western blot analysis revealed that among four tested lines—HepG2, HCCLM3, primary HCC cells, and normal LO2 hepatocytes—HepG2-Luc exhibited the highest HDAC6 expression level, with a densitometric ratio of HDAC6/GAPDH reaching 1.30. This was approximately two-fold greater than HCCLM3 cells (ratio 0.65) and over sixty-fold higher than LO2 normal hepatocytes (ratio 0.02). Immunohistochemical staining of 29 paired clinical samples confirmed that 79.3% of HCC tissues showed positive HDAC6 expression versus matched adjacent non-tumor tissue (p<0.0001), establishing HDAC6 as a robust and selective target for HCC-specific molecular imaging.
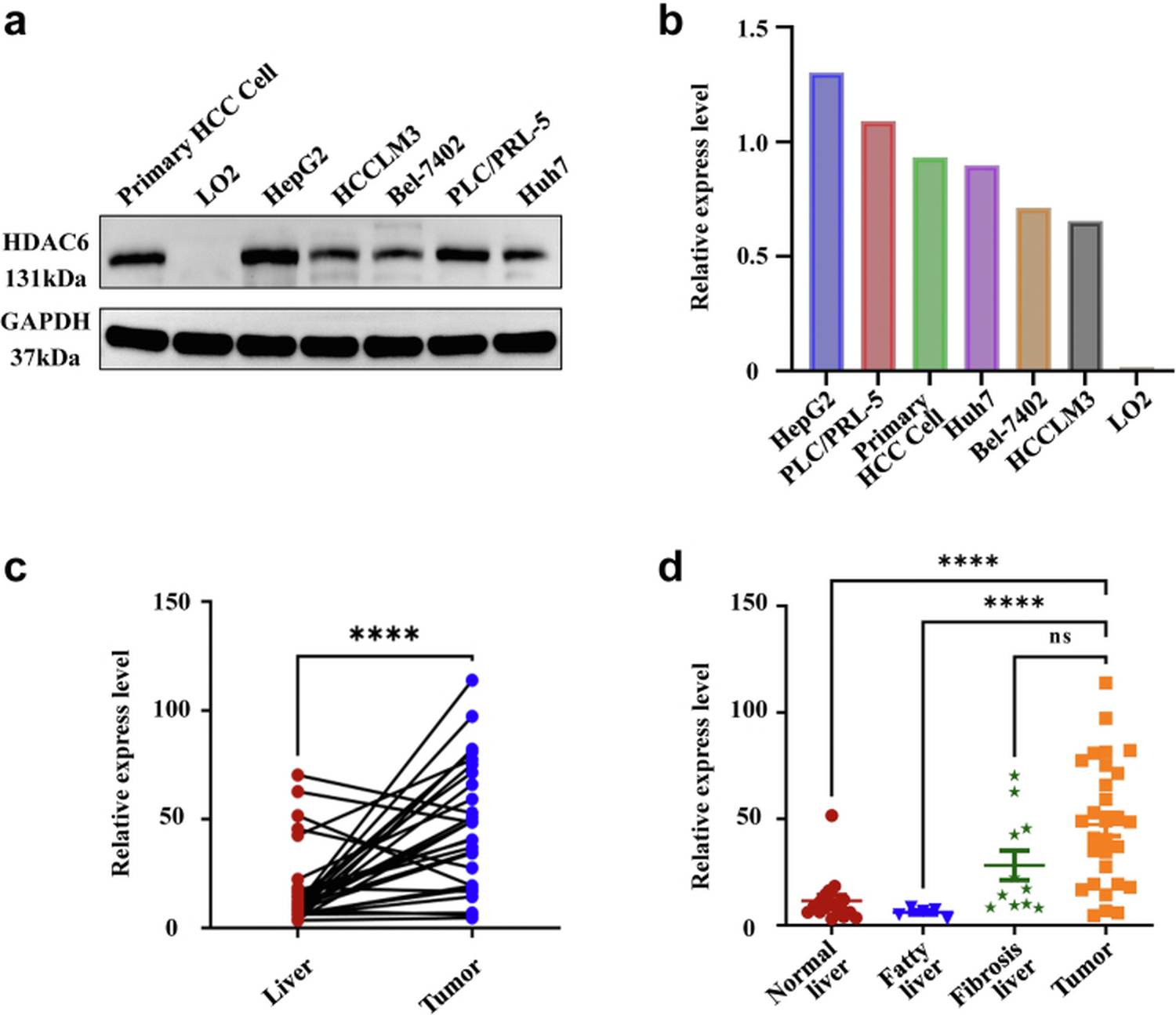
Figure 1. HDAC6 expression profiling across liver cell lines and clinical HCC specimens. (A) Western blot demonstrating highest HDAC6 level in HepG2 cells. (B) IHC quantification of 29 patient pairs showing significant overexpression in tumor vs. adjacent tissue.
The selection of HepG2-Luc as the primary model cell line for this study was therefore well-founded—its high endogenous HDAC6 expression provides optimal contrast for evaluating HDAC6-targeted probes both in vitro and in vivo.
SeCF₃-IRD800: Design and Optical Properties
SeCF3-IRD800 was synthesized by conjugating a trifluoromethyl-modified HDAC6 inhibitor (SeCF3) to the NIR dye IRDye800cw-NHS ester through amide coupling, achieving an overall yield of 84% with >98% purity. The probe exhibits an absorption maximum at 773 nm and emission extending into the NIR-II window (>1000 nm), which provides deeper tissue penetration and reduced autofluorescence compared to conventional NIR-I dyes such as indocyanine green (ICG). Concentration-dependent fluorescence demonstrated excellent linearity (R²=0.9873), enabling reliable quantitative imaging across a broad dynamic range.
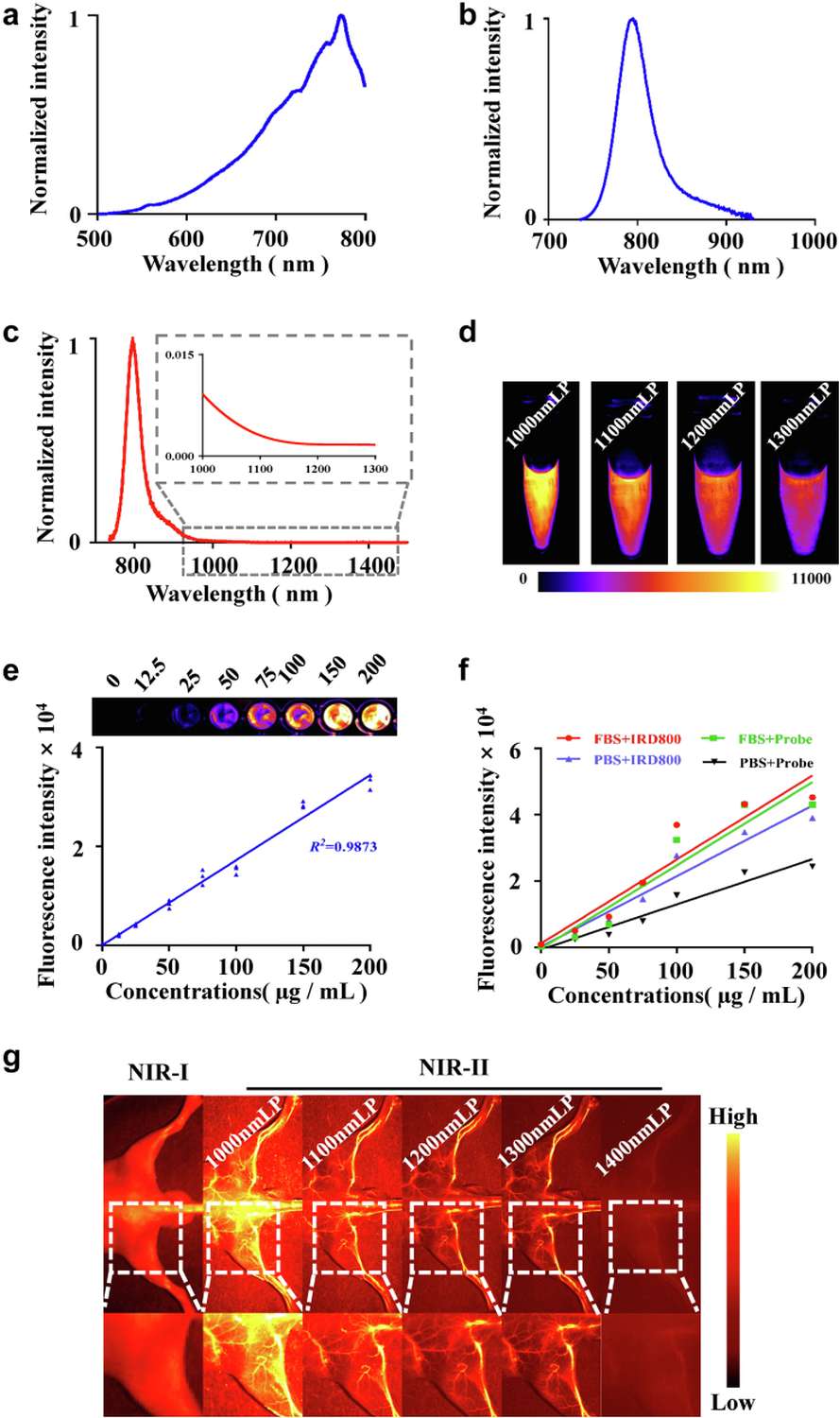
Figure 2. Characterization of SeCF₃-IRD800. (A) Chemical synthesis scheme and structure. (B-C) Absorption and emission spectra. (D) Linear correlation between concentration and fluorescence intensity. (E) NIR-II vascular imaging advantage over NIR-I window.
In Vivo Imaging and Biodistribution
Pharmacokinetic profiling was conducted using HepG2-Luc subcutaneous xenograft models (n=12). Following intravenous injection of SeCF3-IRD800, serial NIR-II imaging revealed that tumor fluorescence peaked at 8 hours post-injection with a mean intensity of 7,658 ± 933 arbitrary units and a tumor-to-background ratio (TBR) of 5.20 ± 1.04. In direct comparison with ICG administered at equivalent dose, SeCF3-IRD800 showed significantly higher tumor signal at all time points from 4 h onward (p<0.05). Critically, hepatic background signal declined rapidly with SeCF3-IRD800, whereas ICG produced persistent and intense liver accumulation (2,417 ± 770 AU at 48 h), which would severely compromise intraoperative discrimination of liver tumors from surrounding parenchyma. Ex vivo organ biodistribution confirmed predominant renal clearance, with tumor-to-liver ratios averaging 6.86 (maximum 10.97), substantially exceeding those of non-target organs (p<0.0001).
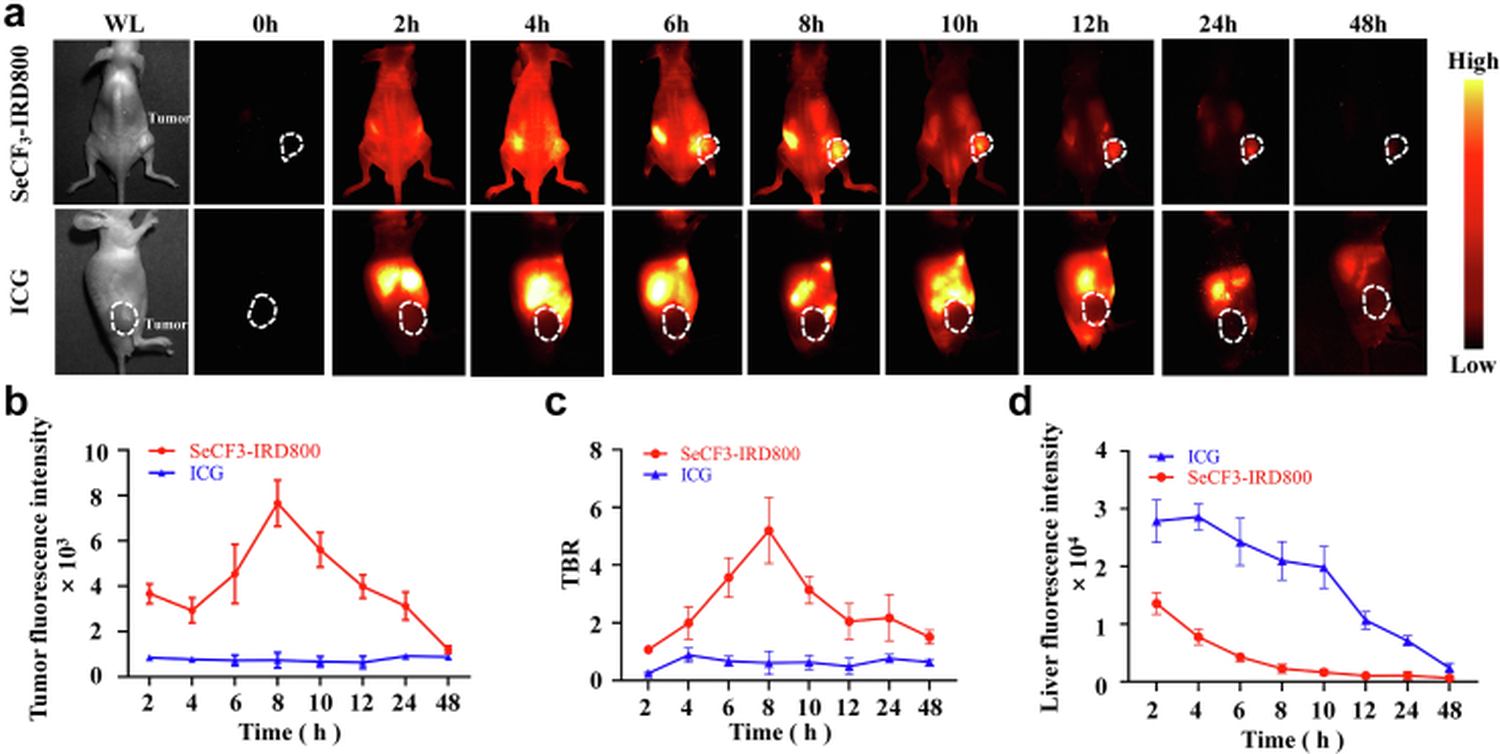
Figure 3. In vivo pharmacokinetics of SeCF₃-IRD800 in HepG2-Luc subcutaneous tumor models. (A) Serial NIR-II imaging time course showing peak tumor signal at 8 h. (B) Tumor fluorescence intensity comparison between SeCF₃-IRD800 and ICG. (C) Ex vivo organ distribution confirming renal clearance pathway.
HDAC6-Specific Tumor Targeting Validation
To confirm that tumor uptake was specifically mediated by HDAC6 binding rather than passive accumulation via the enhanced permeability and retention (EPR) effect, three experimental groups were compared: (1) HepG2-Luc xenografts with high HDAC6 expression; (2) HCCLM3-Luc xenografts expressing low HDAC6 levels; and (3) HepG2-Luc xenografts pre-blocked with excess SAHA (vorinostat), a pan-HDAC inhibitor, at 55-fold molar excess 45 minutes prior to probe injection. At the 8-hour imaging window, the HepG2-Luc group exhibited the highest mean tumor fluorescence (15,029 ± 1,841 AU, TBR = 4.55 ± 0.49). The low-HDAC6 HCCLM3 group showed significantly reduced signal (11,216 ± 976 AU, p<0.001), and SAHA pretreatment nearly abolished tumor accumulation (5,543 ± 1,452 AU, TBR = 0.98 ± 0.37, p<0.001 vs. unblocked HepG2). This tripartite validation demonstrates that SeCF3-IRD800 localizes to tumors through active, HDAC6-dependent targeting mechanisms.
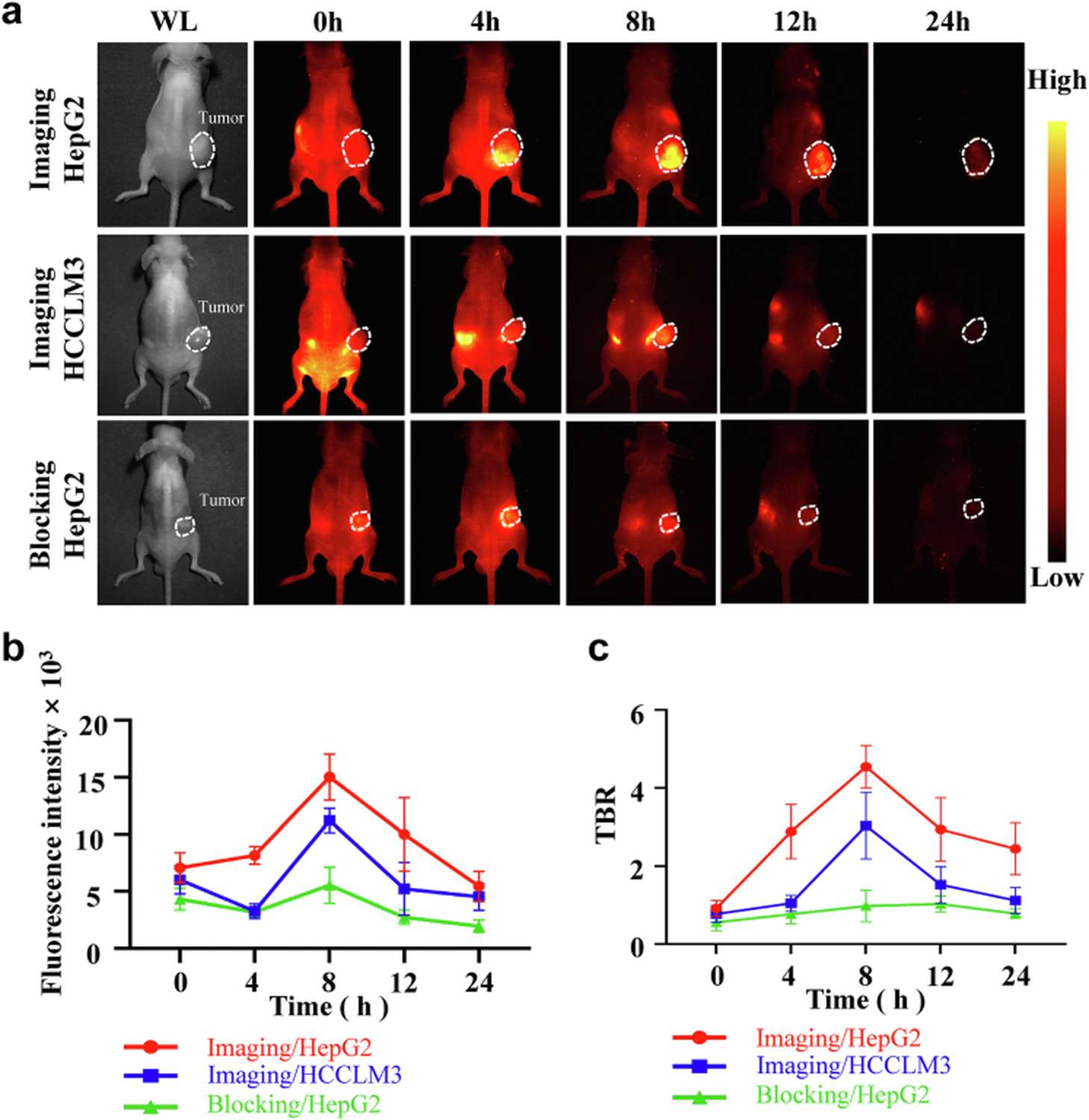
Figure 4. HDAC6-specific targeting validation. (A) Schematic of three-arm specificity experiment. (B-D) Representative images and quantitative fluorescence intensity at 8 h post-injection comparing high-HDAC6 (HepG2-Luc), low-HDAC6 (HCCLM3-Luc), and SAHA-blocked groups.
Place your order and receive expert technical support. Our HepG2-Luc cell line is validated for both subcutaneous and orthotopic HCC model establishment — browse our catalog and find the perfect luciferase reporter model for your study.
Order now: HepG2 Cell Line / HepG2 Luciferase Cell Line / HepG2-Luc-GFP Cell Line
NIR-II Fluorescence-Guided Surgical Resection
The ultimate clinical application of SeCF3-IRD800 was evaluated through fluorescence-guided surgery (FGS) protocols in HepG2-Luc tumor-bearing mice. A standardized four-step workflow was established: (1) initial white-light inspection to identify grossly visible lesions; (2) primary resection of visible tumor mass; (3) NIR-II fluorescence surveillance of the surgical bed to detect residual foci; (4) secondary excision of any fluorescence-positive tissue. Quantitative analysis confirmed that NIR-II fluorescence intensity in tumor regions significantly exceeded that in adjacent liver tissue (p<0.0001), providing clear intraoperative demarcation of tumor margins that were indistinguishable under white light alone.
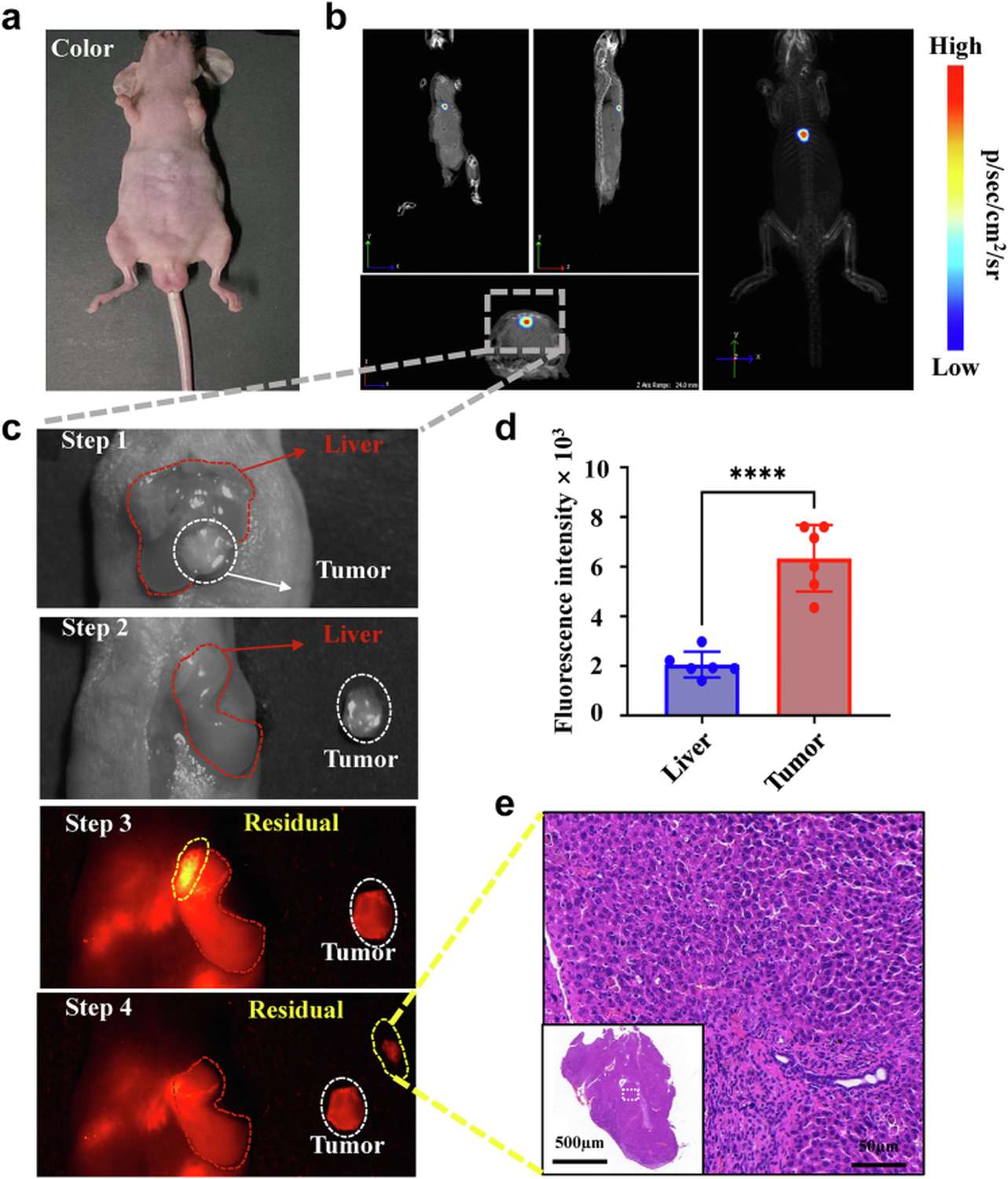
Figure 5. NIR-II fluorescence-guided surgery workflow in HepG2-Luc orthotopic models. (A) Four-step FGS protocol diagram. (B-E) Representative white-light and corresponding NIR-II images at each surgical stage, with quantitative fluorescence intensity comparison between tumor and liver tissue.
Detection of Sub-Millimeter Residual Lesions by Dual-Modal Imaging
Perhaps the most compelling demonstration of this technology involved the detection of residual micro-lesions following what appeared to be complete tumor resection under white light. In HepG2-Luc orthotopic models (n=6), bioluminescence imaging (BLI) confirmed the presence of viable residual tumor cells post-resection, but BLI is not translatable to human surgery. When the same animals were imaged with the NIR-II system, two distinct foci were identified that were completely invisible to the naked eye. Histopathological examination (H&E staining) subsequently confirmed both lesions as malignant tissue, measuring only 2.0 mm and 2.5 mm in diameter respectively. This capability to detect sub-2 mm residual disease represents a potential paradigm shift in HCC surgical oncology, where current recurrence rates are driven precisely by such occult deposits left behind during standard resection.
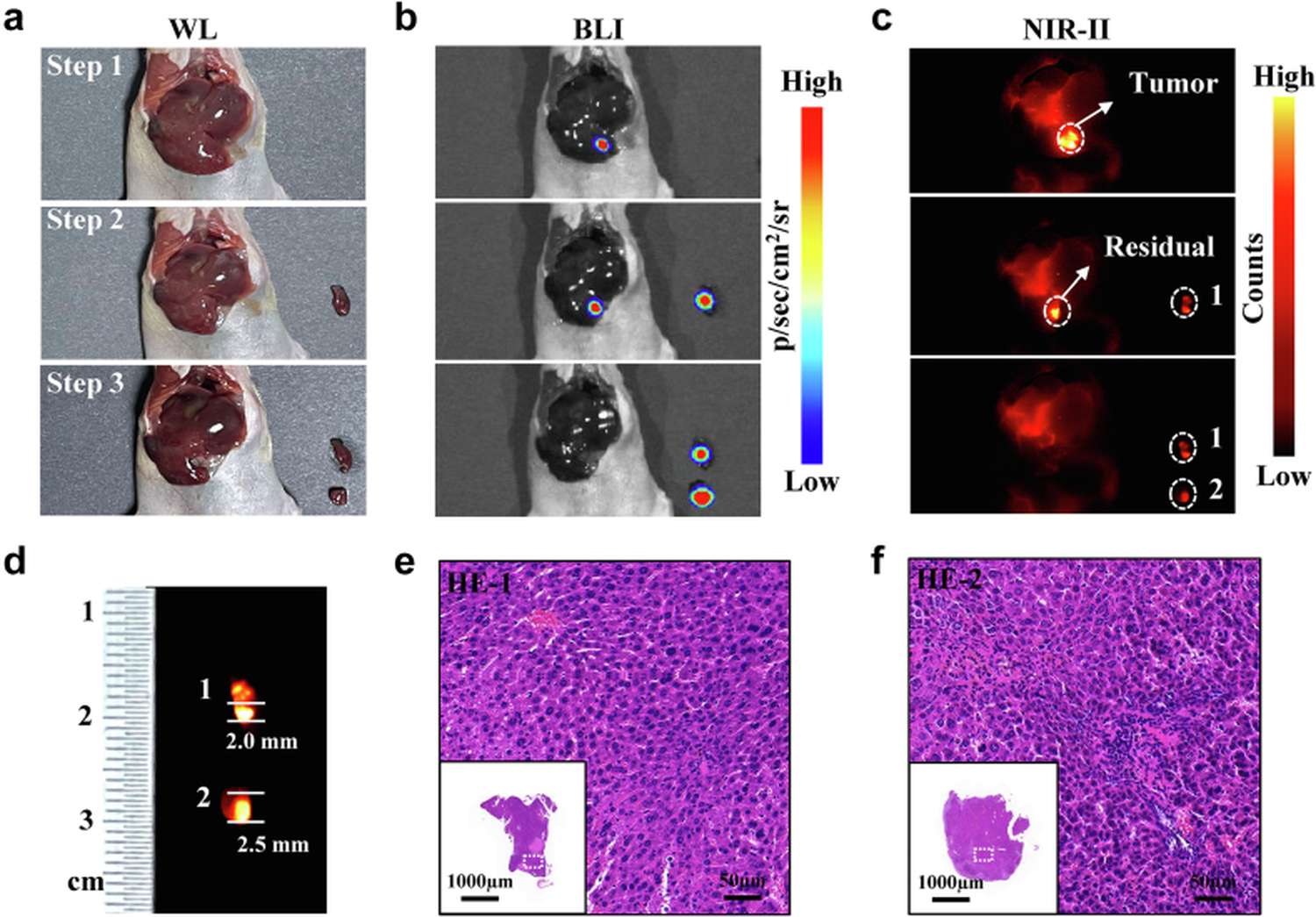
Figure 6. Detection of white-light-invisible sub-millimeter residual lesions in HepG2-Luc orthotopic HCC model using dual-modal BLI + NIR-II fluorescence. (A) Experimental timeline. (B) White-light appearance of resection bed showing no visible residual tumor. (C) BLI confirming presence of viable residual disease. (D) NIR-II fluorescence revealing two distinct micro-foci (arrows). (E-F) H&E histopathology confirming malignancy of 2.0 mm and 2.5 mm lesions respectively.
Performance Across Pathologically Diverse Liver Backgrounds
A notable practical concern for any liver-directed imaging agent is whether underlying pathology—such as cirrhosis or fibrosis, present in >80% of HCC patients—affects imaging performance. The investigators addressed this directly by testing SeCF3-IRD800 in three contexts: normal liver, fibrotic liver (induced by CCl4 administration), and HCC-bearing liver. Importantly, no significant difference in background fluorescence was observed between fibrotic and normal liver tissue. Furthermore, a strong linear correlation (R² = 0.8124) was observed between HDAC6 protein expression levels and corresponding NIR-II fluorescence intensities across all tissue types, indicating that probe signal reliably reflects underlying tumor biology regardless of the host liver's pathological state.
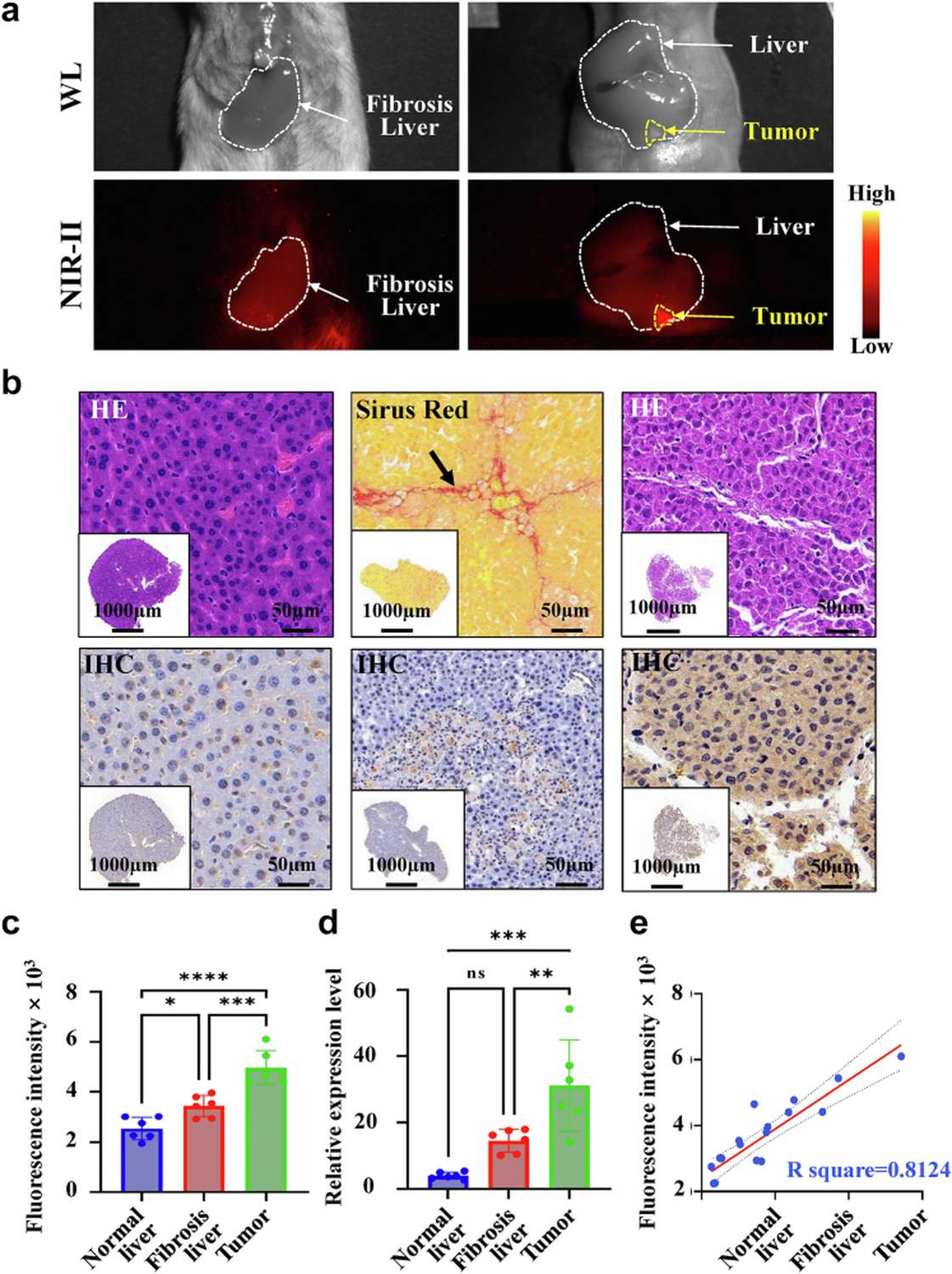
Figure 7. Imaging performance in diverse liver pathologies. (A) Experimental design with normal, fibrotic, and HCC groups. (B-D) Representative images across conditions. (E) Correlation plot demonstrating R²=0.8124 relationship between HDAC6 expression and NIR-II fluorescence intensity.
Ex Vivo Validation Using Clinical HCC Specimens
To bridge the translational gap toward clinical application, freshly resected HCC specimens from patients were incubated ex vivo with SeCF3-IRD800 and imaged using the same NIR-II platform. Tumor regions displayed markedly stronger fluorescence than adjacent non-tumor liver tissue, and this differential signal was competitively abolished by SAHA co-incubation, confirming HDAC6-dependent binding in human tissue. Normal liver samples from the same patients served as negative controls and exhibited minimal background fluorescence. These data provide essential preliminary evidence supporting the feasibility of translating this approach into intraoperative use during human hepatectomy procedures.
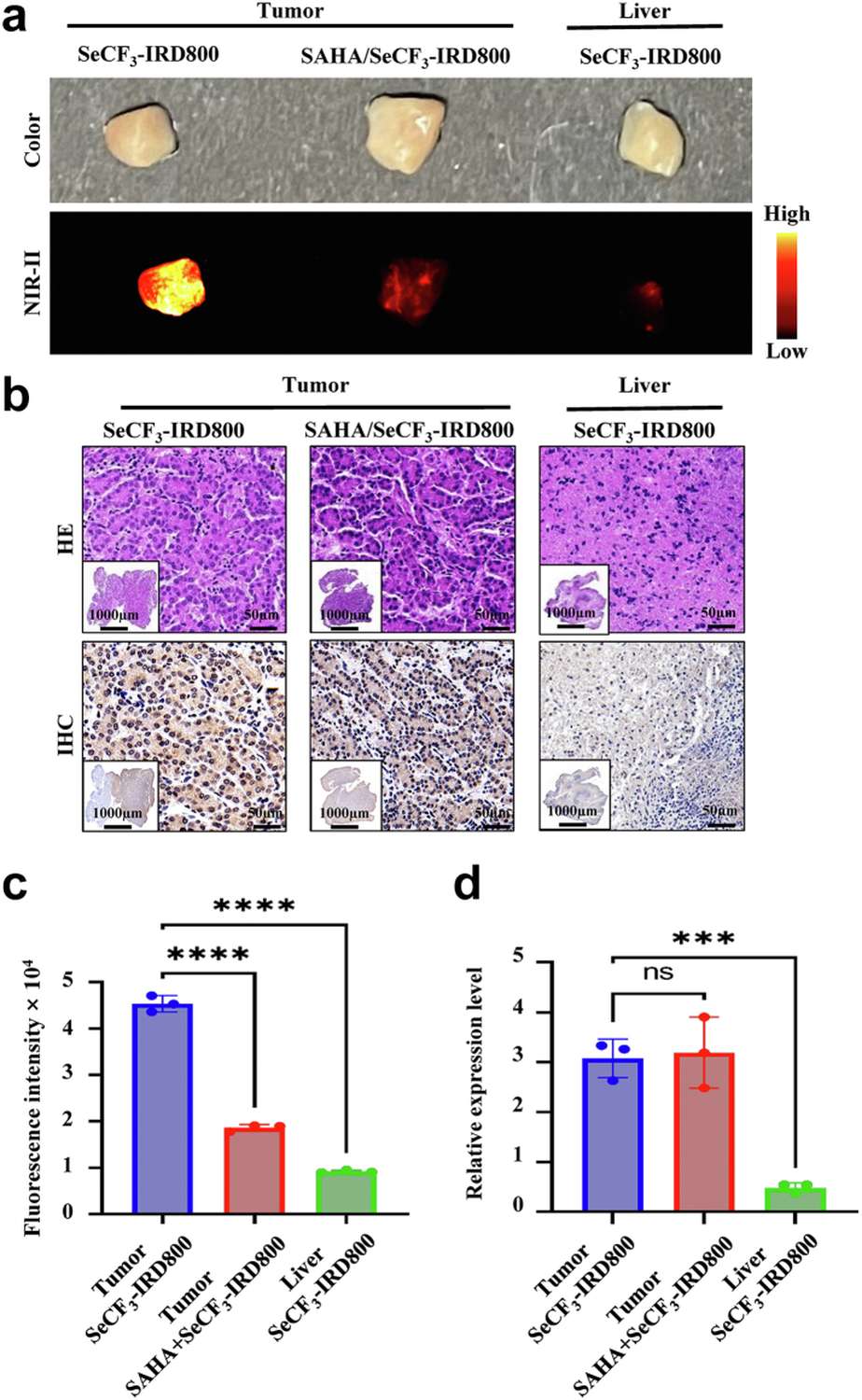
Figure 8. Ex vivo validation using resected human HCC specimens. (A-B) NIR-II fluorescence images of patient-derived HCC vs. normal liver tissue. (C) SAHA blocking control confirming HDAC6 specificity in human tissue. (D) Quantitative fluorescence intensity comparison across specimen groups.
Conclusions and Perspectives
This study presents a rationally designed HDAC6-targeted NIR-II fluorescent probe with demonstrated utility for real-time image-guided liver cancer surgery. Key advantages include rapid renal clearance (eliminating problematic liver background signal common with ICG), an 8-hour optimal imaging window compatible with surgical scheduling, HDAC6-dependent specific tumor accumulation, and most critically, the ability to identify residual foci smaller than 2.5 mm that are invisible under standard white-light illumination. The HepG2-Luc bioluminescent orthotopic model proved instrumental in validating these capabilities through dual-modal cross-verification, where BLI provided ground-truth confirmation of tumor presence while NIR-II fluorescence delivered clinically actionable guidance. Future directions include first-in-human clinical trials and potential extension to robotic-assisted laparoscopic hepatectomy systems equipped with NIR-II detection channels.
References
1. Wang B, Tang C, Lin E, Jia X, Xie G, Li P, Li J, Hu Z, Tian J, Li J. NIR-II fluorescence-guided liver cancer surgery by a small molecular HDAC6 targeting probe. EBioMedicine. 2023 Dec;104:104880. doi: 10.1016/j.ebiom.2023.104880. PMID: 38035463.

