TCF1 Dependency in Tumor Immunotherapy: Insights from MC38-OVA Model
Introduction
Cancer immunotherapy has emerged as one of the most transformative approaches in modern oncology, with immune checkpoint blockade (ICB) therapies demonstrating remarkable success in treating various malignancies. However, despite these advances, a significant proportion of patients fail to respond to ICB treatment, highlighting the urgent need to understand the underlying mechanisms that determine therapy response. The identification of predictive biomarkers and the elucidation of immune response determinants have become central challenges in cancer research.
TCF1 (T-cell factor 1), encoded by the TCF7 gene, is a transcription factor historically associated with T-cell development and memory formation. Recent studies have revealed that TCF1-expressing CD8+ T cells represent a distinct population with stem-like properties, capable of self-renewal and generating effector T-cell responses. These TCF1+ T cells have been shown to be essential for maintaining immune responses during chronic infections and cancer. However, the precise role of TCF1 in determining ICB therapy outcomes across different tumor types remained unclear.
The MC38-OVA cell line represents a valuable preclinical model for studying cancer immunotherapy. MC38 is a murine colon adenocarcinoma cell line derived from C57BL/6 mice, which expresses the model antigen ovalbumin (OVA) as a transgene. This OVA expression allows researchers to track antigen-specific CD8+ T cell responses using well-characterized OVA-derived peptides (such as SIINFEKL) and MHC class I tetramers. The MC38-OVA model has been widely used to investigate tumor immunology, immune checkpoint therapy, and combination treatment strategies.
For your research needs, we provide high-quality MC38 wild-type cell lines with guaranteed viability and authentic STR authentication. Our MC38 cells are carefully characterized and validated for use in tumor immunology research. Contact us to discuss your research requirements and receive expert technical support!
Tumor Immunogenicity Determines TCF1 Dependency
The key finding of this study is that the requirement for TCF1 in CD8+ T cells for ICB therapy response is fundamentally dictated by tumor immunogenicity. Researchers utilized two complementary tumor models with differing immunogenic properties: the highly immunogenic MC38-OVA colon carcinoma and the poorly immunogenic B16F10-OVA melanoma. Both cell lines express ovalbumin as a shared neoantigen, allowing direct comparison of T-cell responses under controlled conditions.
In experiments using MC38-OVA tumors, which are highly immunogenic and respond well to ICB therapy, the deletion of TCF1 in CD8+ T cells did not impair therapeutic efficacy. These findings demonstrate that in tumors with strong intrinsic immunogenicity, alternative T-cell populations can compensate for the loss of TCF1+ stem-like cells, ensuring effective anti-tumor immune responses. This remarkable plasticity suggests that highly immunogenic tumors activate multiple pathways for immune-mediated tumor control.
Conversely, in B16F10-OVA tumors, which have low intrinsic immunogenicity and respond poorly to ICB therapy, TCF1 deletion completely abrogated treatment response. This critical difference highlights that low-immunogenicity tumors depend almost exclusively on TCF1+ CD8+ T cells for therapeutic benefit. The stem-like properties of these cells appear to be essential for generating sustained anti-tumor immunity in the context of weak tumor antigens and immunosuppressive microenvironments.
Mechanistic Insights into TCF1 Function
Further investigation revealed the mechanistic basis for these observations. In highly immunogenic tumors like MC38-OVA, the robust antigen presentation and inflammatory cytokine milieu promote the expansion of diverse T-cell populations, including both TCF1+ and TCF1- subsets. The high tumor mutation burden and abundant neoantigen expression trigger extensive dendritic cell activation and optimal CD8+ T cell priming, ultimately generating a diverse effector T cell pool capable of tumor control independent of TCF1.
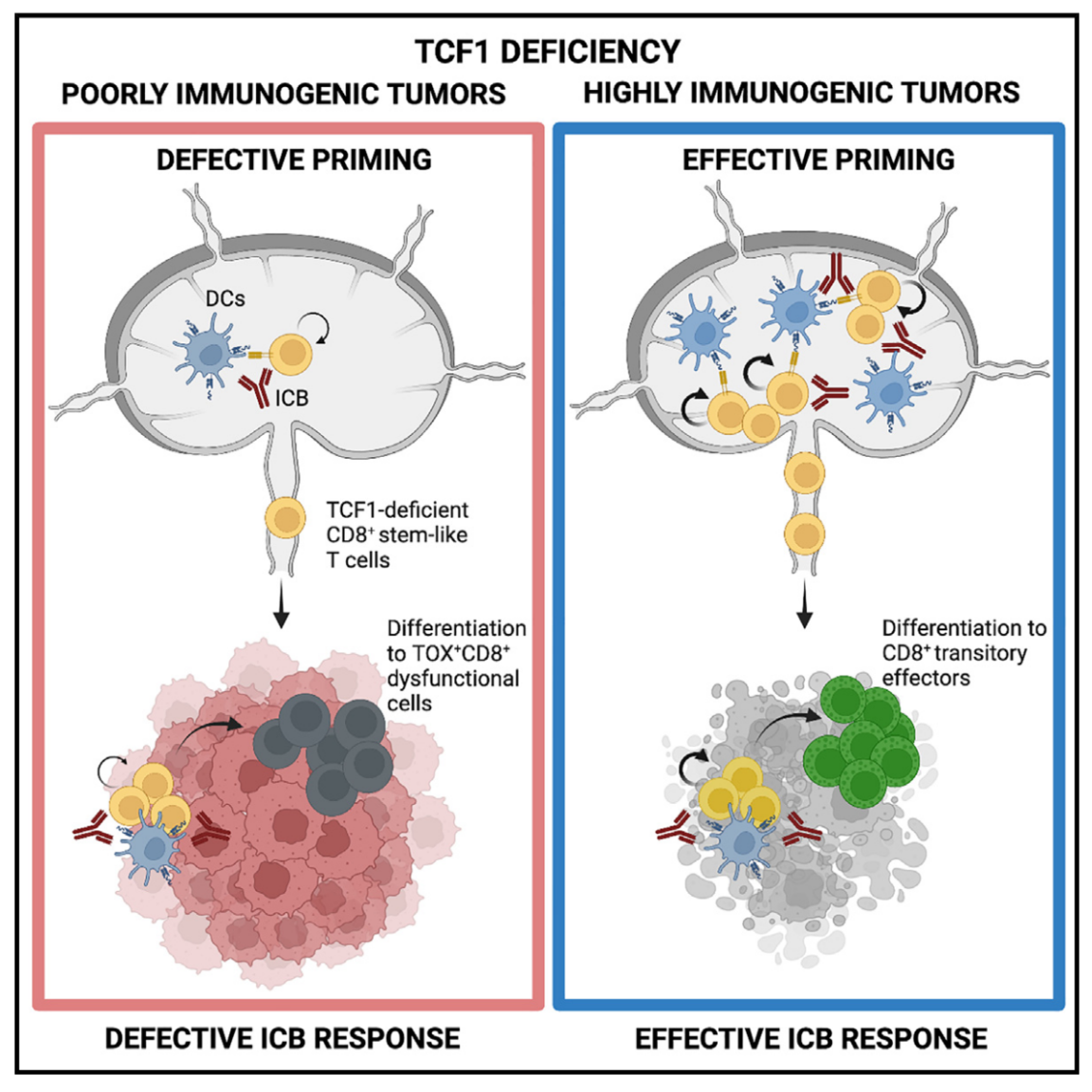
Figure 1
Schematic representation of TCF1-dependent and TCF1-independent pathways in CD8+ T cell response to ICB therapy in tumors with varying immunogenicity. In highly immunogenic tumors (left panel), both TCF1+ and TCF1- T cell populations contribute to tumor control, while in low-immunogenicity tumors (right panel), TCF1+ stem-like cells are essential for therapeutic efficacy.
In contrast, low-immunogenicity tumors such as B16F10-OVA present a significant challenge to the immune system. The sparse neoantigen landscape results in weak T cell priming and preferential reliance on the TCF1+ stem-like population for sustained immune responses. These TCF1+ cells serve as a reservoir for continuous T cell generation, and without them, the immune system fails to mount an effective anti-tumor response even when checkpoint inhibitors are administered.
We offer both MC38 and MC38-OVA cell lines for your immunotherapy research. Whether you need wild-type MC38 for studying native tumor immunology or MC38-OVA for antigen-specific immune response studies, we have you covered. Request a quote now to get started with your research project.
Combination Therapy Strategies
One of the most promising translational applications of this research is the development of combination therapy strategies for patients with low-immunogenicity tumors. The study demonstrated that cancer vaccines could restore ICB therapy response in TCF1-deficient mice bearing low-immunogenicity tumors. This finding suggests that enhancing tumor immunogenicity through vaccination can overcome the TCF1 dependency and enable effective immunotherapy.
Cancer vaccines work by delivering tumor antigens in an immunogenic context, thereby enhancing antigen presentation and activating dendritic cells. When combined with ICB therapy, this approach can potentially convert 'cold' tumors into 'hot' tumors that are responsive to immune checkpoint inhibition. The combination of therapeutic vaccines with anti-PD-1 or anti-CTLA-4 antibodies represents a promising strategy for treating patients who would otherwise not benefit from immunotherapy alone.
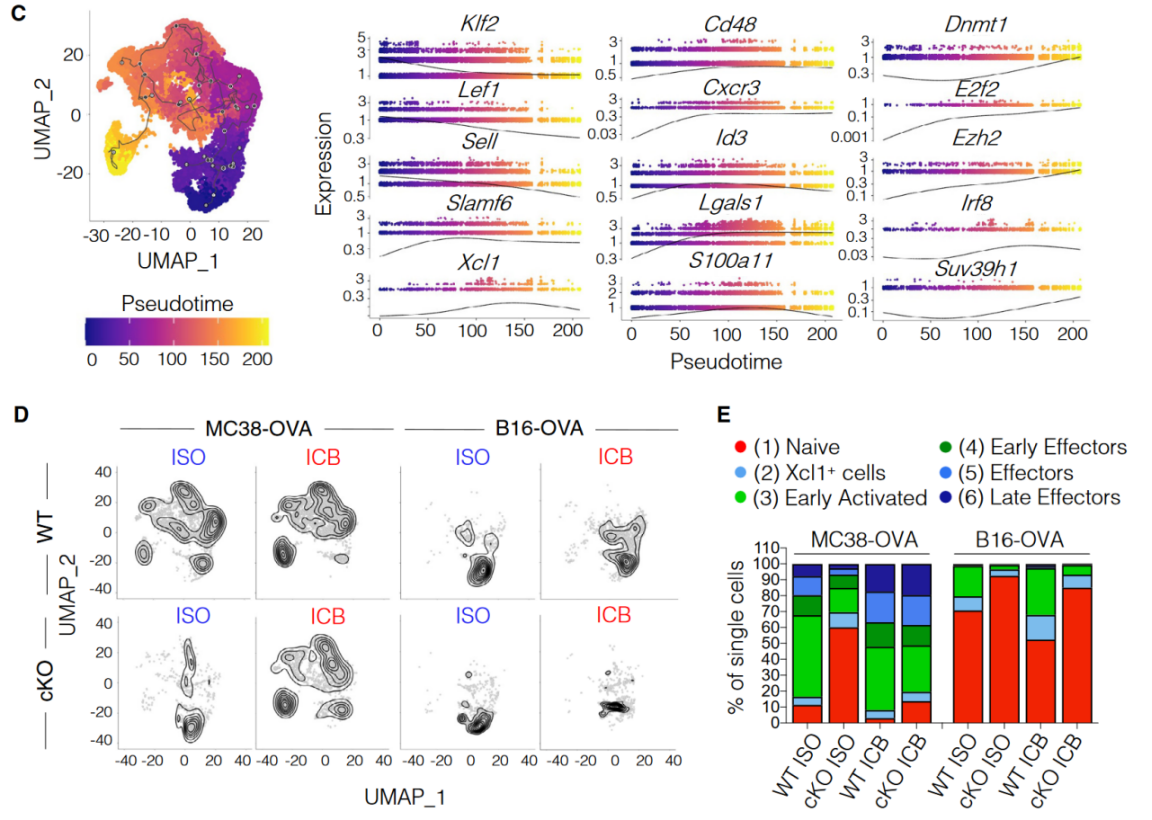
Figure 2
Tumor growth curves in MC38-OVA and B16F10-OVA models showing differential response to ICB therapy with and without TCF1 deletion. Panel A demonstrates that MC38-OVA tumors respond to anti-PD-1 therapy regardless of TCF1 status, while Panel B shows that B16F10-OVA tumors lose therapeutic response upon TCF1 deletion.
Clinical Implications and Future Directions
The findings from this study have significant clinical implications for patient stratification and treatment selection. By assessing tumor immunogenicity through measures such as tumor mutation burden, neoantigen load, and immune gene expression signatures, clinicians may be able to predict which patients are likely to benefit from ICB monotherapy and which require combination approaches. Patients with low-immunogenicity tumors might benefit from sequential or concurrent treatment with cancer vaccines, adoptive T cell therapy, or other immunomodulatory agents.
Furthermore, the identification of TCF1+ CD8+ T cells as key mediators of ICB response in certain tumor types provides a potential biomarker for treatment monitoring. By tracking the frequency and functional status of these cells in peripheral blood or tumor biopsies, clinicians may be able to predict treatment response and adjust therapy accordingly. Flow cytometry analysis of TCF1 expression in CD8+ T cells could become a standard diagnostic tool in immunotherapy oncology.
Figure 3
Flow cytometry analysis of tumor-infiltrating CD8+ T cells showing TCF1 expression patterns in MC38-OVA versus B16F10-OVA tumors. The data reveals distinct T cell populations and their correlation with therapeutic response.
Our commitment to quality ensures that every cell line we provide meets the highest standards for research applications. With comprehensive technical support and flexible ordering options, we make it easy to get the materials you need for your immunotherapy research. Place your order now>>
Conclusion
This research provides fundamental insights into the relationship between tumor immunogenicity and TCF1-dependent immune responses. The discovery that TCF1 dependency varies with tumor immunogenicity has important implications for understanding cancer immunotherapy mechanisms and developing effective treatment strategies. The MC38-OVA model serves as an excellent tool for studying antigen-specific immune responses in highly immunogenic tumors, while the comparative analysis with B16F10-OVA reveals the critical role of tumor intrinsic properties in determining treatment outcomes.
For researchers working in cancer immunology and immunotherapy, the MC38 and MC38-OVA cell lines remain essential tools for preclinical investigation. These models enable the study of complex immune-tumor interactions, the testing of novel therapeutic combinations, and the translation of basic science findings into clinical applications. As our understanding of tumor immunogenicity continues to deepen, the development of personalized immunotherapy approaches that account for individual tumor characteristics will become increasingly important for improving patient outcomes.