LRBA Degradation Unlocks Antitumor Immunity Through CTLA4 | A Novel Therapeutic Strategy Using MC38-OVA Cell Model
Introduction
While LRBA deficiency is known to cause severe autoimmune disease and CTLA4 loss, its role in antitumor immunity remained unclear. This study systematically demonstrates that LRBA is a "bad actor" in the tumor microenvironment, where high LRBA expression suppresses antitumor immune responses. The research discovered LC427, an oral small-molecule degrader that specifically disrupts the LRBA-CTLA4 interaction, representing a novel therapeutic strategy for cancer immunotherapy.
For your immunotherapy research, consider using our high-quality wild-type MC38 cell line, which maintains optimal growth characteristics and tumorigenicity. Contact us to discuss your research needs and receive expert technical support.
LRBA as a Negative Regulator of Antitumor Immunity
The researchers first sought clinical evidence by analyzing patient samples. In colorectal cancer patients, LRBA and CTLA4 expression showed positive correlation in both tumor and adjacent tissues. This clinical observation prompted further investigation into LRBA's role in tumor immunity.
Database analysis across multiple cancer types revealed striking patterns. In breast cancer and lung cancer microenvironments, high LRBA expression negatively correlated with cytotoxic CD8+ T cell infiltration while positively correlating with regulatory T cells (Tregs). This suggests LRBA creates an immunosuppressive tumor microenvironment.
The most definitive evidence came from genetic knockout mouse models. LRBA-deficient mice were inoculated with MC38 colon cancer cells, and results clearly showed that LRBA knockout significantly inhibited tumor growth. This bidirectional evidence proves that LRBA acts as a "brake" on antitumor immunity.
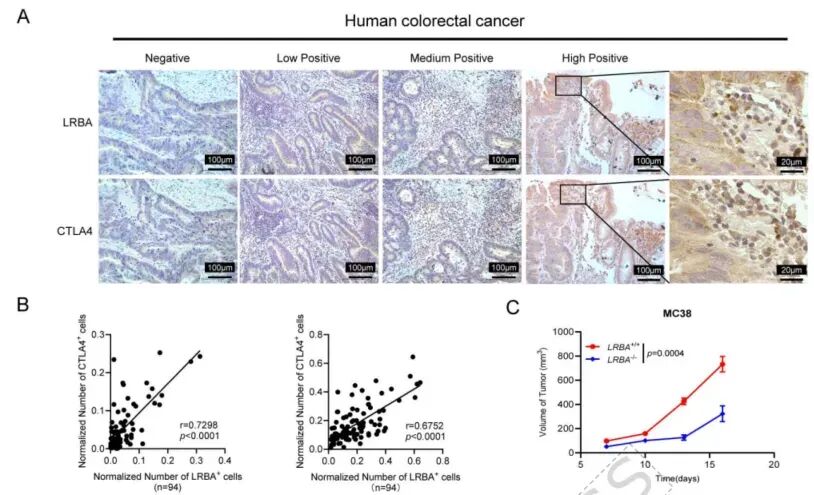
Figure 1. LRBA and CTLA4 Expression Positively Correlates in Tumor Tissues
Discovery and Characterization of LC427
The research team developed an innovative nano-luciferase complementation reporter system to screen for compounds that could disrupt LRBA-CTLA4 interaction. This elegant system works by fusing luciferase fragments to key binding domains of LRBA and CTLA4; only when they interact does the complete luciferase emit light.
From a library of 4,501 compounds, they identified a hit compound WAY-262611. Further optimization yielded LC427, which demonstrated significantly improved activity and reduced cellular toxicity. LC427 efficiently inhibited luminescence in the reporter assay, and effectiveness was abolished when binding site mutations were introduced, confirming specificity.
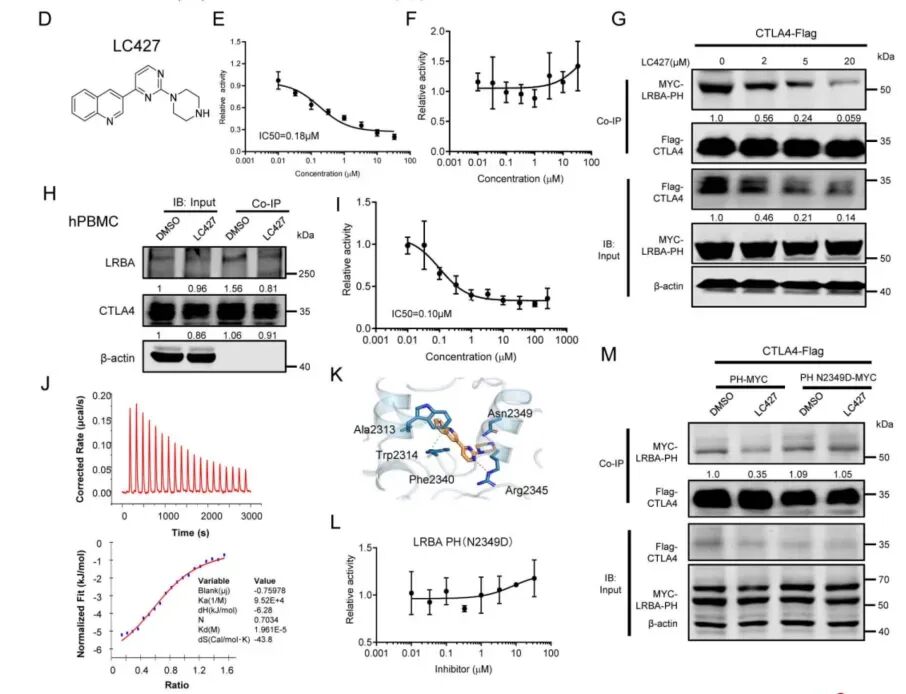
Figure 2. Discovery and Characterization of LC427
Mechanism of Action
Multiple experimental approaches confirmed LC427's mechanism. Co-immunoprecipitation experiments in both murine and human immune cells showed that LC427 disrupts the native LRBA-CTLA4 interaction. In vitro binding assays with purified proteins also demonstrated direct inhibition.
Isothermal titration calorimetry measured a equilibrium dissociation constant (Kd) of 19.6 µM, confirming direct physical binding between LC427 and LRBA protein. Computational molecular docking predicted binding in the PH-BEACH domain of LRBA, and mutation of key binding sites (such as Asn2349) abolished LC427 activity.
A sophisticated experiment separating surface and intracellular CTLA4 revealed that LRBA primarily binds intracellular CTLA4, and LC427 disrupts this interaction. This indicates the mechanism operates mainly within the cell.
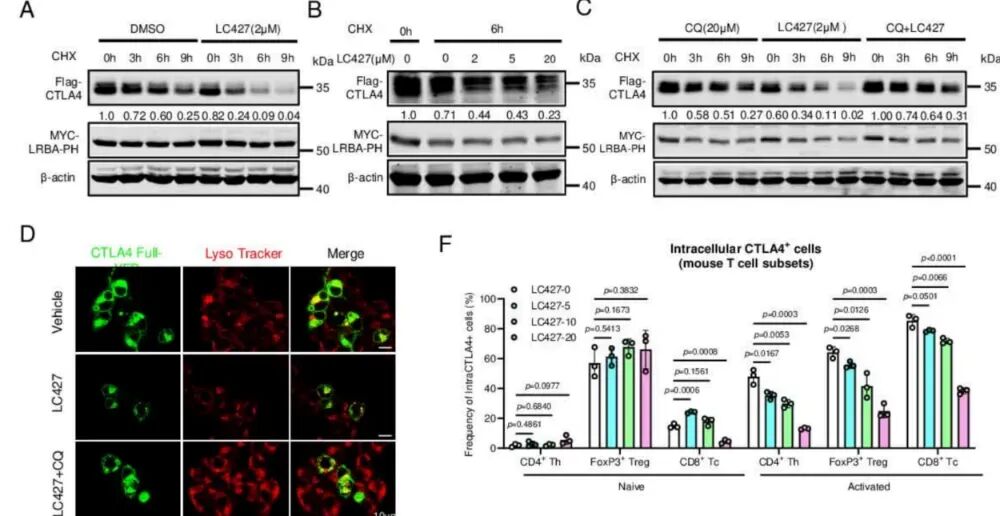
Figure 3. LC427 Discovery, Optimization, and Mechanism of Action
LC427 Induces CTLA4 Degradation
In cells overexpressing LRBA and CTLA4, LC427 treatment reduced CTLA4 protein levels in both time-dependent and dose-dependent manners. The degradation pathway was identified as lysosomal, as the lysosomal inhibitor chloroquine completely rescued CTLA4 reduction.
Fluorescence microscopy directly demonstrated increased CTLA4 localization in lysosomes following LC427 treatment. The mechanism is clear: by disrupting LRBA's protection of CTLA4, LC427 redirects CTLA4 to lysosomal degradation.
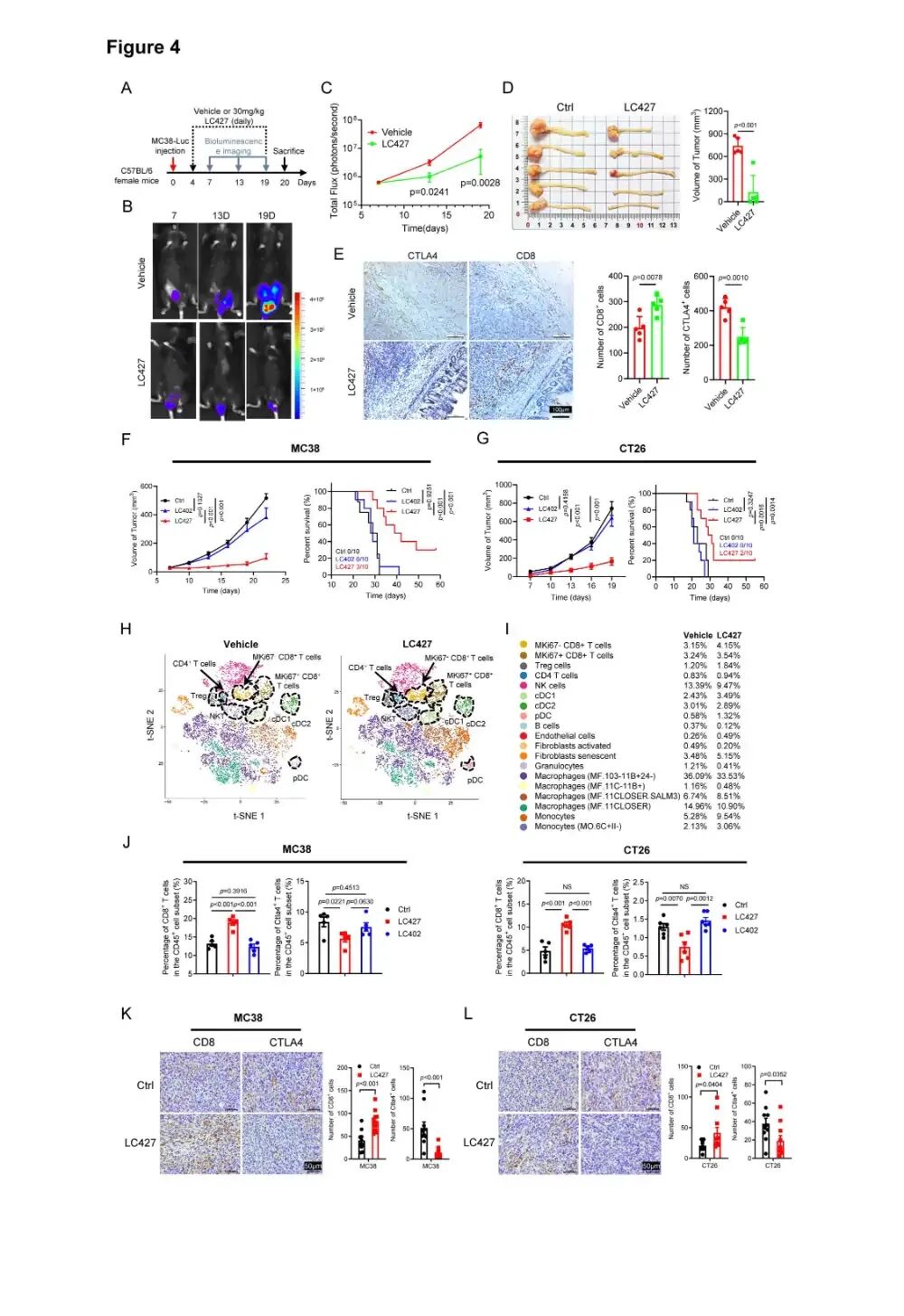
Figure 4. LC427 Mechanism and Impact on T Cell Function
Our MC38 wild-type cell line provides reliable and consistent results for cancer immunotherapy research.Request a quote now to get started with your preclinical studies.
In Vivo Antitumor Efficacy
Oral administration of LC427 significantly inhibited tumor growth in multiple murine tumor models, including MC38 orthotopic colon cancer, subcutaneous tumors, and CT26 colon cancer. Some mice achieved complete tumor regression. The structurally similar compound LC402 served as a negative control and was ineffective, confirming the specificity of LC427.
Single-cell sequencing and flow cytometry analysis revealed that LC427 treatment increased cytotoxic CD8+ T cells and natural killer T cells (NKT) in tumors while reducing CTLA4+ T cells, transforming the tumor microenvironment from "cold" to "hot".
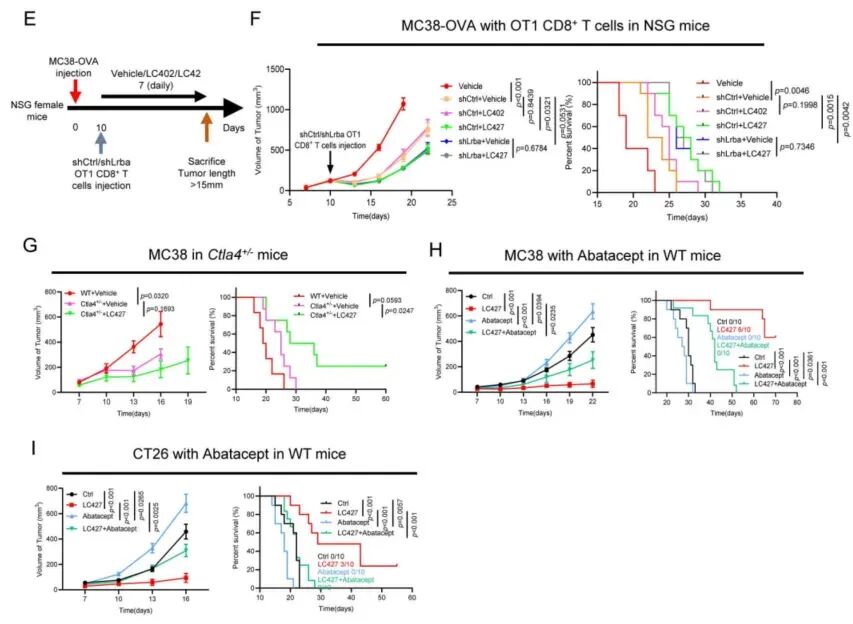
Figure 5. LC427 Inhibits Tumor Growth Through T Cell and LRBA-CTLA4 Axis
Mechanism Dependence on T Cells and LRBA-CTLA4 Axis
Rigorous genetic and cellular experiments provided multilayered evidence. In immunodeficient NSG mice, LC427 was completely ineffective, proving dependence on an intact immune system. Antibody-mediated depletion of CD8+ or CD4+ T cells abolished LC427 efficacy.
In adoptive transfer experiments, LC427 lost efficacy when transferred T cells had LRBA knocked down. In Ctla4 heterozygous mice (with partial loss of function), LC427 efficacy was significantly reduced. These experiments conclusively demonstrate that LC427 exerts antitumor effects through the LRBA-CTLA4 axis, acting on T cells, particularly CD8+ T cells.
Conclusion
This study presents a novel oral small-molecule strategy to overcome CTLA4 antibody toxicity by targeting LRBA, the "regulator" of CTLA4, rather than CTLA4 itself. While the path from mouse to human remains long, including verification in different cancer types, combination therapies, and clinical trials, this work highlights that targeting immune checkpoint "regulators" rather than the checkpoints themselves may represent a more promising therapeutic approach.
References
1. Ge X, et al. Targeting LRBA triggers CTLA4 degradation and antitumor immunity for cancer immunotherapy. Nature Communications. 2023 Dec 14;14(1):8001.

