MCF7 Luc: SCUBE2-Driven Osteoblastic Niche Remodeling Governs Luminal Breast Cancer Bone Metastasis
Introduction
Breast cancer remains the leading cause of cancer-related deaths in women worldwide, and distant metastasis is the primary driver of this mortality. Among all metastatic sites, bone is the most frequently affected organ, with up to 70% of late-stage breast cancer patients developing skeletal involvement. Intriguingly, this propensity for bone metastasis is disproportionately concentrated in estrogen receptor-positive (ER+) luminal breast cancer (LBC), a subtype that paradoxically carries a lower overall risk of metastasis compared to more aggressive subtypes. The molecular mechanisms underpinning this organ-specific tropism had remained poorly defined — a critical gap that constrained the development of effective preventive and therapeutic strategies. The study published in Cell Research identified SCUBE2 as a pivotal, ER-regulated secretory factor that reshapes the bone microenvironment to facilitate the selective colonization of MCF7 Luc cells, offering a compelling mechanistic explanation for LBC's bone tropism.
Looking for a reliable MCF7 Luc cell line for your breast cancer bone metastasis or bioluminescence imaging studies? Our MCF7 Luc cells are mycoplasma-tested, STR-authenticated, and ready to use. Contact us today to discuss your research needs and request a quote.
Order now: MCF7 Luciferase Cell Line / MCF7-Luc-GFP Cell Line
SCUBE2 Expression Correlates with Bone Metastasis and Is Regulated by ER Signaling in Luminal Breast Cancer
To identify secretory factors specifically upregulated in LBC, the authors interrogated large-scale public clinical datasets. This systematic screen revealed SCUBE2 as the top candidate: a secretory protein whose expression was markedly elevated in ER+ LBC compared to other molecular subtypes and even more pronounced in bone metastatic tumors. Kaplan-Meier survival analysis further demonstrated that high SCUBE2 expression was significantly correlated with increased bone metastasis risk in LBC patients. Mechanistically, SCUBE2 was shown to be a direct transcriptional target of ER signaling — estrogen treatment upregulated SCUBE2 expression, while ER antagonists suppressed it — linking its expression pattern to the LBC-specific hormonal milieu.
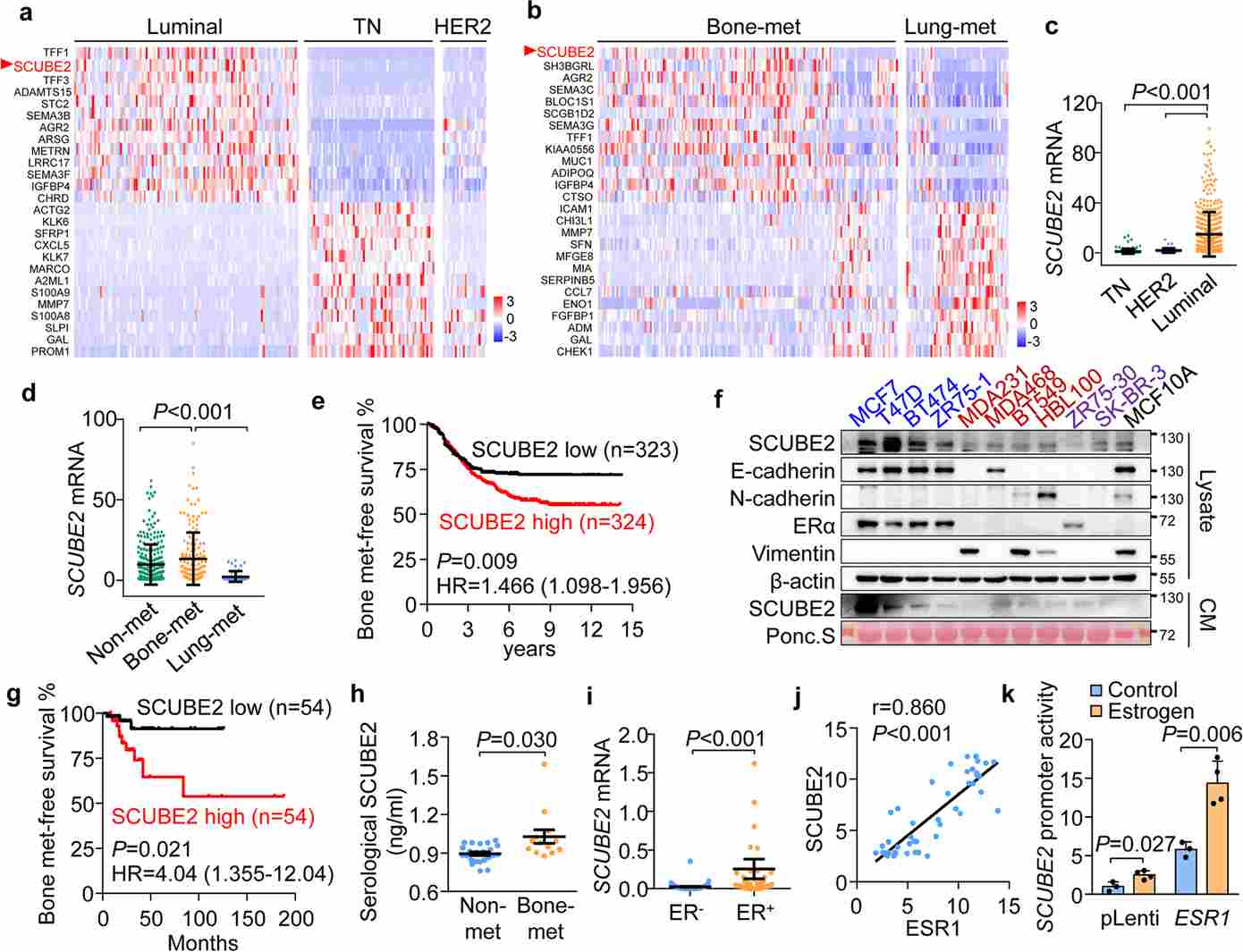
Figure 1. Heatmap of secretory protein-encoding gene expression across different molecular subtypes of breast cancer; Kaplan-Meier analysis linking SCUBE2 expression to bone metastasis risk.
SCUBE2 Promotes Bone Metastasis of Breast Cancer In Vivo
To functionally validate SCUBE2's role in bone colonization, the researchers employed MCF7 Luc cells — stably expressing firefly luciferase — in intracardiac injection models. Bioluminescence imaging (BLI) allowed real-time, non-invasive monitoring of skeletal tumor burden. Overexpression of SCUBE2 in MCF7 Luc cells dramatically accelerated bone metastasis formation, as evidenced by intensified BLI signals in the long bones of injected nude mice. Conversely, SCUBE2 knockdown markedly attenuated bone colonization, with significantly reduced BLI signals and fewer metastatic foci. These gain- and loss-of-function experiments compellingly established that SCUBE2 is both necessary and sufficient to drive LBC bone tropism in vivo.

Figure 2. BLI images and quantification showing bone metastasis in nude mice injected intracardially with MCF7 Luc cells with or without SCUBE2 overexpression/knockdown; representative X-ray images of metastatic lesions.
SCUBE2 Remodels the Osteoblastic Niche to Suppress NK Cell Activity
Single-cell RNA sequencing (scRNA-seq) of early-stage bone metastatic niches from MCF7 Luc-injected mice revealed a striking enrichment of osteoblasts in SCUBE2-high metastatic environments. Mechanistically, SCUBE2 promotes the release of membrane-anchored Sonic Hedgehog (SHH) from tumor cells. This secreted SHH then activates the Hedgehog signaling pathway in adjacent mesenchymal stem cells, driving their differentiation into osteoblasts. The newly differentiated osteoblasts deposit abundant type I collagen (COL1) into the extracellular matrix. This collagen-rich environment suppresses NK cell cytotoxicity by engaging the inhibitory receptor LAIR1 on NK cells, activating the downstream SHP1 phosphatase to dampen NK cell activation. In ex vivo co-culture assays, osteoblast-conditioned matrices derived from SCUBE2-stimulated conditions significantly impaired NK cell-mediated killing of MCF7 Luc organoids.
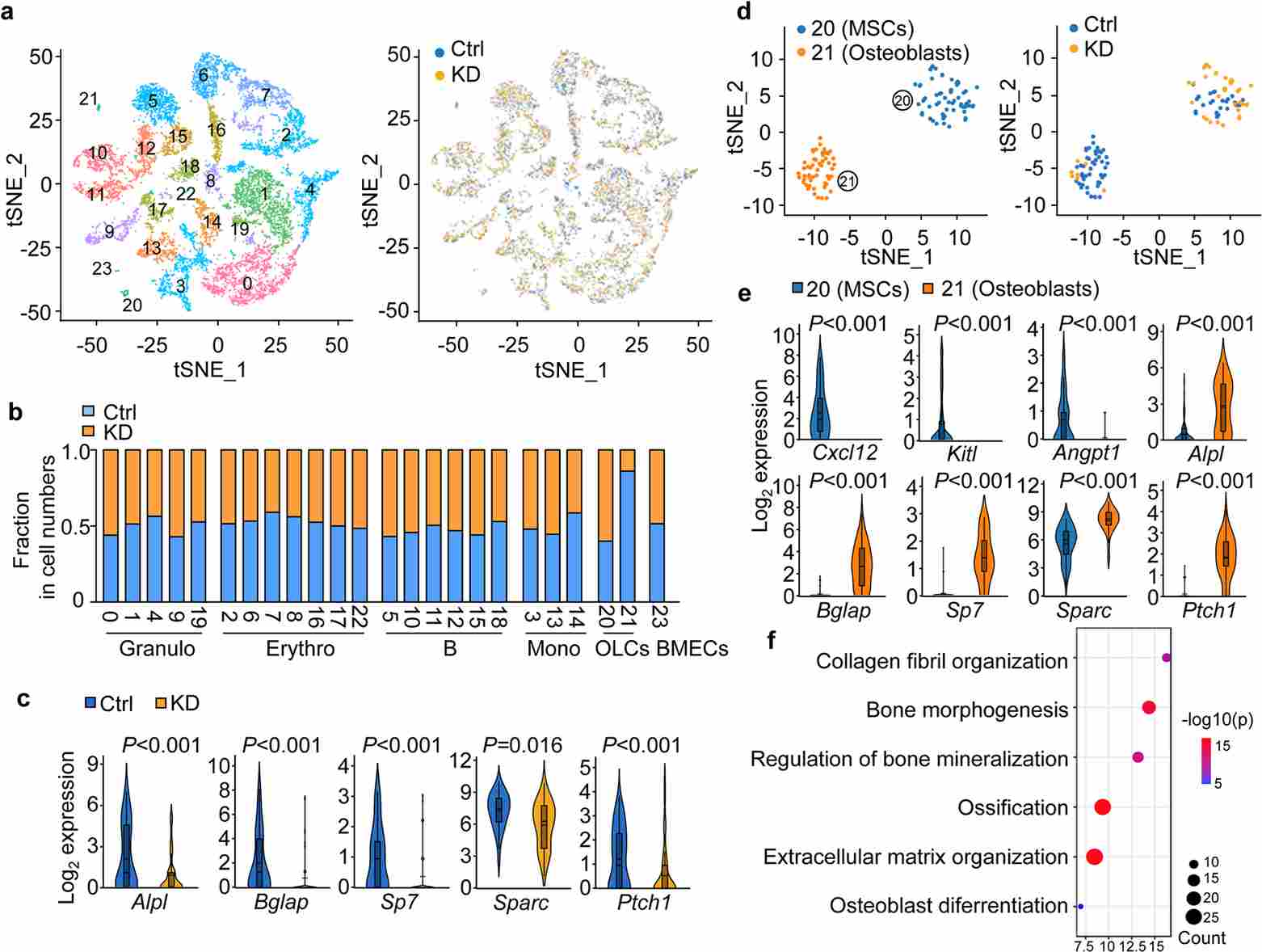
Figure 3. Co-culture assay showing NK cell coverage of MCF7 Luc patient-derived organoids (PDOs) in osteoblast-conditioned versus control conditions; quantification of NK cell cytotoxicity.
Targeting Hedgehog Signaling with Sonidegib Suppresses SCUBE2-Driven Bone Metastasis
Recognizing that the SCUBE2→SHH→Hedgehog pathway is central to osteoblastic niche remodeling, the researchers tested Sonidegib — a clinically approved Hedgehog pathway inhibitor — as a potential anti-metastatic agent. In vitro, Sonidegib treatment effectively blocked osteoblast differentiation induced by SCUBE2-overexpressing MCF7 Luc cells. In vivo, oral administration of Sonidegib to mice bearing SCUBE2-overexpressing MCF7 Luc bone metastases significantly reduced skeletal tumor burden and extended overall survival. Importantly, Sonidegib did not impair primary tumor growth, suggesting a niche-specific mechanism of action.
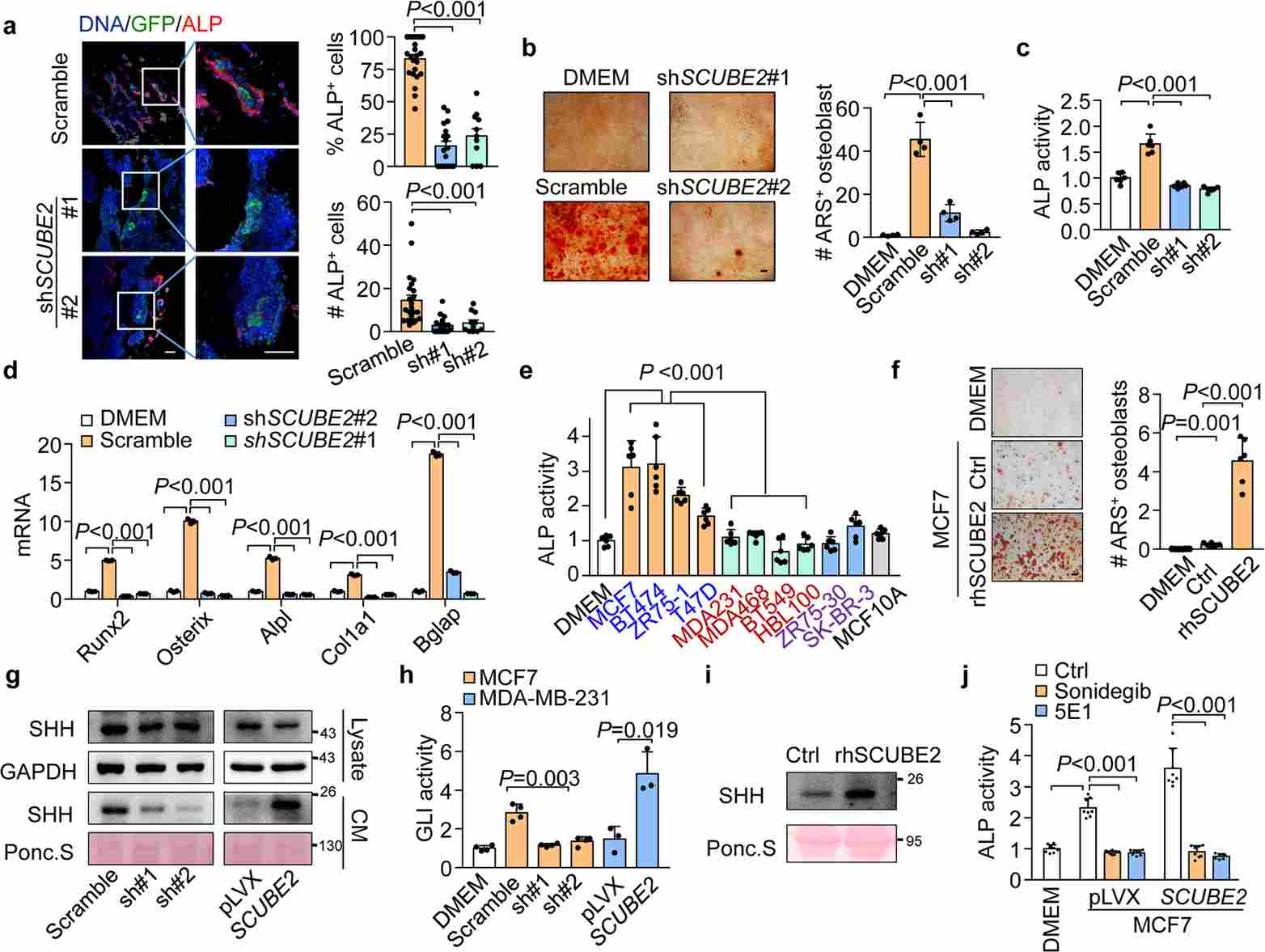
Figure 4. BLI quantification of bone metastasis in Sonidegib-treated versus control mice with SCUBE2-overexpressing MCF7 Luc tumors; Kaplan-Meier survival curves; ALP activity assays.
SCUBE2 Neutralizing Antibody Blocks Bone Metastasis in Orthotopic Models
To complement the pharmacological approach, the team developed a SCUBE2-specific neutralizing antibody. In orthotopic mammary fat pad transplantation models using MCF7 Luc cells, anti-SCUBE2 antibody treatment powerfully suppressed spontaneous bone metastasis without affecting primary tumor growth. This orthogonal validation strengthened the evidence that the SCUBE2 axis is a robust, clinically actionable target with a favorable therapeutic window — an important consideration for future clinical translation.
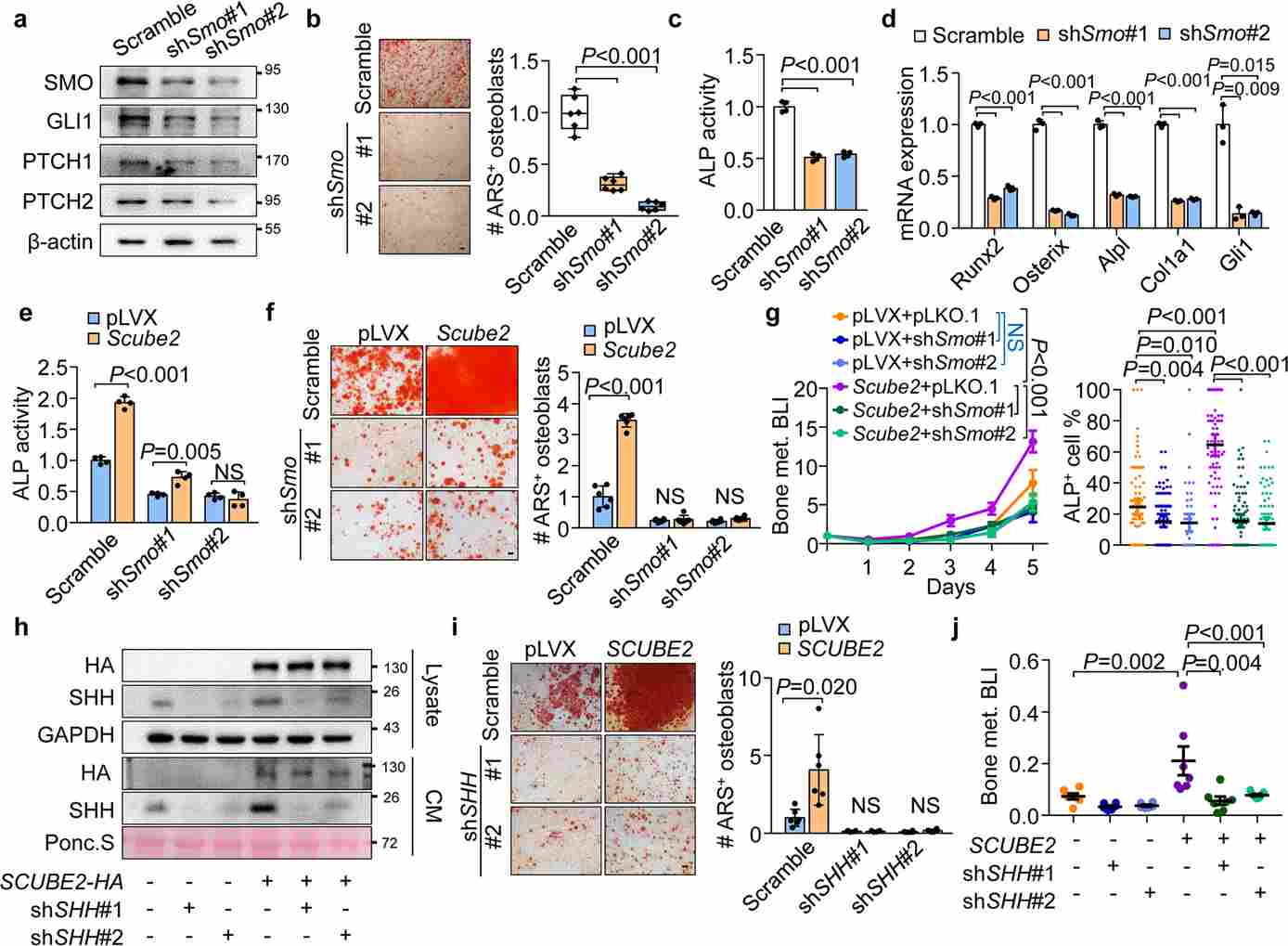
Figure 5. BLI tracking and quantification of spontaneous bone metastasis in orthotopic MCF7 Luc models treated with anti-SCUBE2 antibody versus IgG control; primary tumor growth curves.
Discussion
This study elegantly resolves a long-standing paradox in breast cancer biology: why does ER+ luminal breast cancer, despite its relatively indolent systemic behavior, show such a pronounced predilection for skeletal colonization? The answer lies in a tumor-intrinsic secretory program encoded by SCUBE2 and orchestrated by ER signaling. Rather than directly invading bone, MCF7 Luc cells pre-condition the bone microenvironment by co-opting Hedgehog-driven osteoblastogenesis to create an immunosuppressive COL1-rich niche that silences NK cell surveillance.
The use of MCF7 Luc cells throughout this study underscores the critical role of bioluminescent imaging in capturing the spatiotemporal dynamics of bone metastasis. By allowing non-invasive, longitudinal tracking of single metastatic foci with high sensitivity, BLI enabled precise quantification of the effects of SCUBE2 modulation and therapeutic interventions in live animals. From a clinical standpoint, SCUBE2's serum detectability positions it as a promising liquid biopsy biomarker for stratifying bone metastasis risk in ER+ breast cancer patients. Furthermore, the demonstration that the FDA-approved drug Sonidegib can intercept this pathway provides an immediately translatable therapeutic strategy.
Accelerate your breast cancer research with our MCF7 Luc cell line — a validated, luciferase-expressing model ideally suited for bioluminescence imaging, bone metastasis modeling, and drug efficacy studies. Browse our catalog and find the perfect cell model for your study.
Order now: MCF7 Luciferase Cell Line / MCF7-Luc-GFP Cell Line
Conclusion
This landmark Cell Research study provides a mechanistically complete framework for understanding why ER+ luminal breast cancer preferentially colonizes bone. SCUBE2 emerges as a master regulator of bone tropism: its ER-driven expression in LBC cells initiates a cascade that remodels the osteoblastic niche, suppresses NK cell immunity, and enables tumor engraftment. The MCF7 Luc model was instrumental in delineating this biology in vivo, demonstrating the power of bioluminescent cell lines for bone metastasis research. Targeting SCUBE2 — by neutralizing antibody or by inhibiting its downstream Hedgehog effector pathway with Sonidegib — offers a promising strategy to prevent or treat luminal breast cancer bone metastasis, with potential to meaningfully improve outcomes for a large segment of breast cancer patients.
References
Wu Q, Tian P, He D, et al. SCUBE2 mediates bone metastasis of luminal breast cancer by modulating immune-suppressive osteoblastic niches. Cell Research, 2023; 33(6):476-490. https://doi.org/10.1038/s41422-023-00810-6

