NALM-6 Luc: Illuminating B-ALL Immunotherapy with Target-Restricted IL-15 Immunocytokines
Introduction
Acute B-cell lymphoblastic leukemia (B-ALL) remains one of the most challenging hematological malignancies in adults, with relapsed or refractory disease carrying dismal prognoses despite the availability of CD19-targeted therapies such as blinatumomab and CAR-T cells. Antibody-dependent cellular cytotoxicity (ADCC) mediated by natural killer (NK) cells is a central mechanism for monoclonal antibody therapies such as rituximab (anti-CD20) and other B-cell-directed agents. However, the inherent NK cell activity driven by conventional Fc-optimized antibodies often falls short in eliminating deeply infiltrated leukemia cells—particularly in the bone marrow or central nervous system niches—due to insufficient immune activation and NK cell exhaustion in the tumor microenvironment.
Interleukin-15 (IL-15) is a potent stimulatory cytokine that promotes the proliferation, activation, and cytolytic capacity of NK cells without inducing the systemic immune suppression associated with IL-2. Yet recombinant IL-15 suffers from an extremely short half-life and dose-limiting toxicities when administered systemically. Engineering of "immunocytokines"—antibody-cytokine fusion proteins—offers an elegant solution to co-deliver cytokine activity precisely to tumor-antigen-expressing cells. The challenge, however, is to restrict IL-15 activity to the tumor site to minimize off-target stimulation.
Preclinical leukemia models depend critically on cell lines that faithfully replicate disease biology while permitting non-invasive longitudinal monitoring. NALM-6 Luc, the luciferase-stably transduced derivative of the canonical pre-B-ALL cell line, is uniquely suited for this purpose: its strong CD19/CD20 surface expression and dissemination pattern in immunodeficient mice closely mimic human B-ALL, and whole-body bioluminescence imaging (BLI) delivers quantitative, real-time tumor burden readouts without sacrificing animals at each time point.
Accelerate your B-ALL immunotherapy research with Vitrobiotech's NALM-6 Luc stable cell line — validated for NSG/NCG tail-vein xenograft models with confirmed luciferase activity. Contact us to discuss your research needs.
Order now: NALM-6 Cell Line / NALM-6 Luciferase Cell Line / NALM-6-Luc-GFP Cell Line
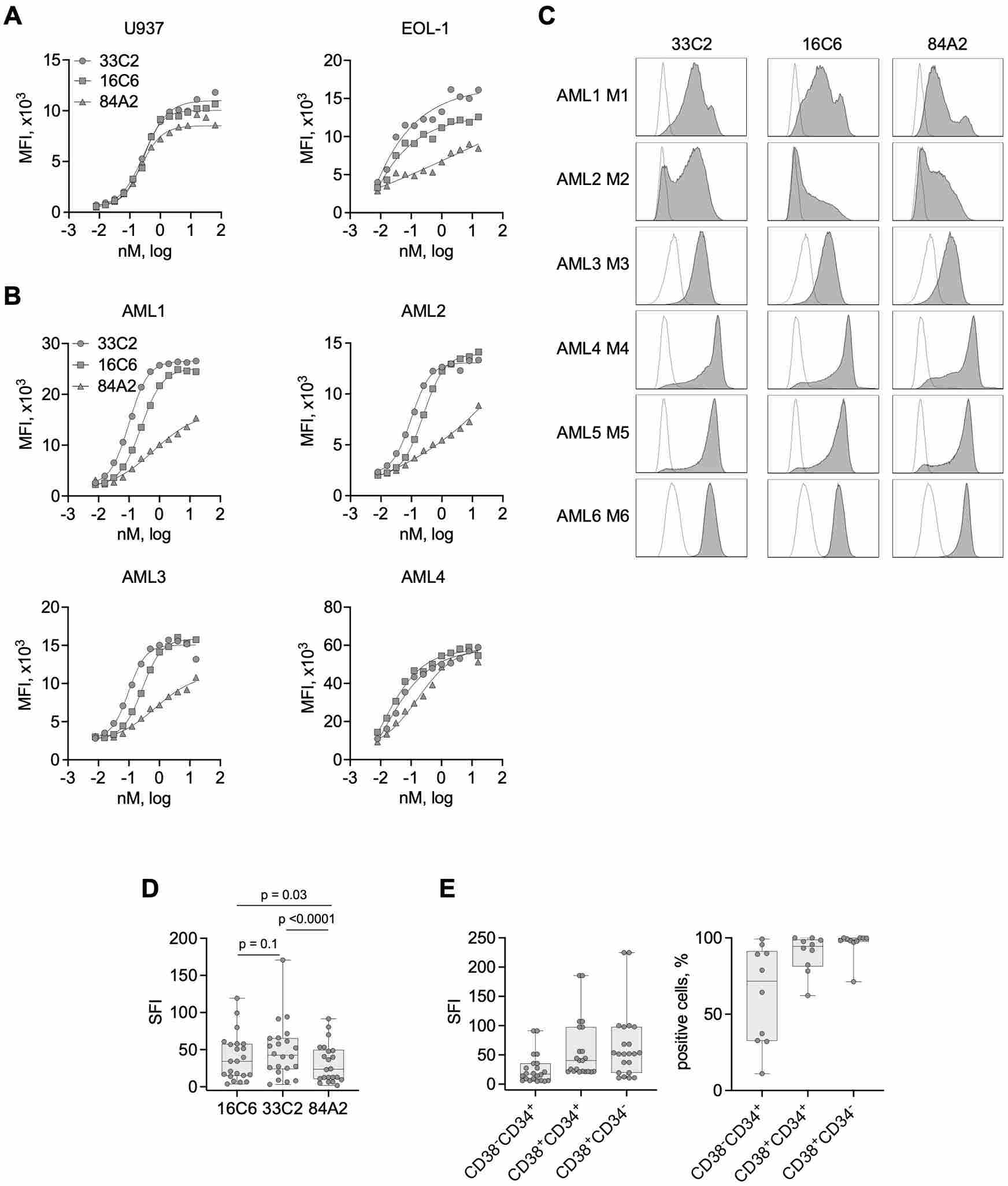
Figure 1. Expression and binding validation of the MIC12/MIC+ immunocytokine platform. CLEC12A/CD19/CD20 expression on leukemia cell lines (left panels) and flow cytometry confirmation of immunocytokine binding to target-antigen-positive cells (right panels). This antibody-engineering validation framework mirrors how NALM-6 Luc is used to confirm MIC+ specificity.
Engineering Conditional IL-15 Activity: The MIC+ Design Principle
The central innovation of MIC+ lies in decoupling IL-15 from its constitutive receptor, IL-15Rα, while preserving signaling through the IL-15Rβγ heterodimer expressed on NK cells. The authors introduced two key mutations into the IL-15 moiety:
(1) E46K: eliminates IL-15Rα binding, preventing the physiological trans-presentation of IL-15 to NK cells in the absence of target cell engagement.
(2) L45E: optimizes IL-15Rβγ agonist activity and improves production yield in CHO manufacturing.
When MIC+ binds to CD19 or CD20 on B-ALL cells via its Fc-optimized antibody domain (SDIE mutations: S239D/I332E, enhancing FcγRIIIA binding and ADCC), the concentrated IL-15 at the cell surface substitutes for physiological trans-presentation, selectively activating NK cells in the immediate tumor vicinity. This architecture eliminates systemic cytokine flooding—a major safety liability of soluble IL-15—while potentiating NK-mediated killing precisely where it is needed.
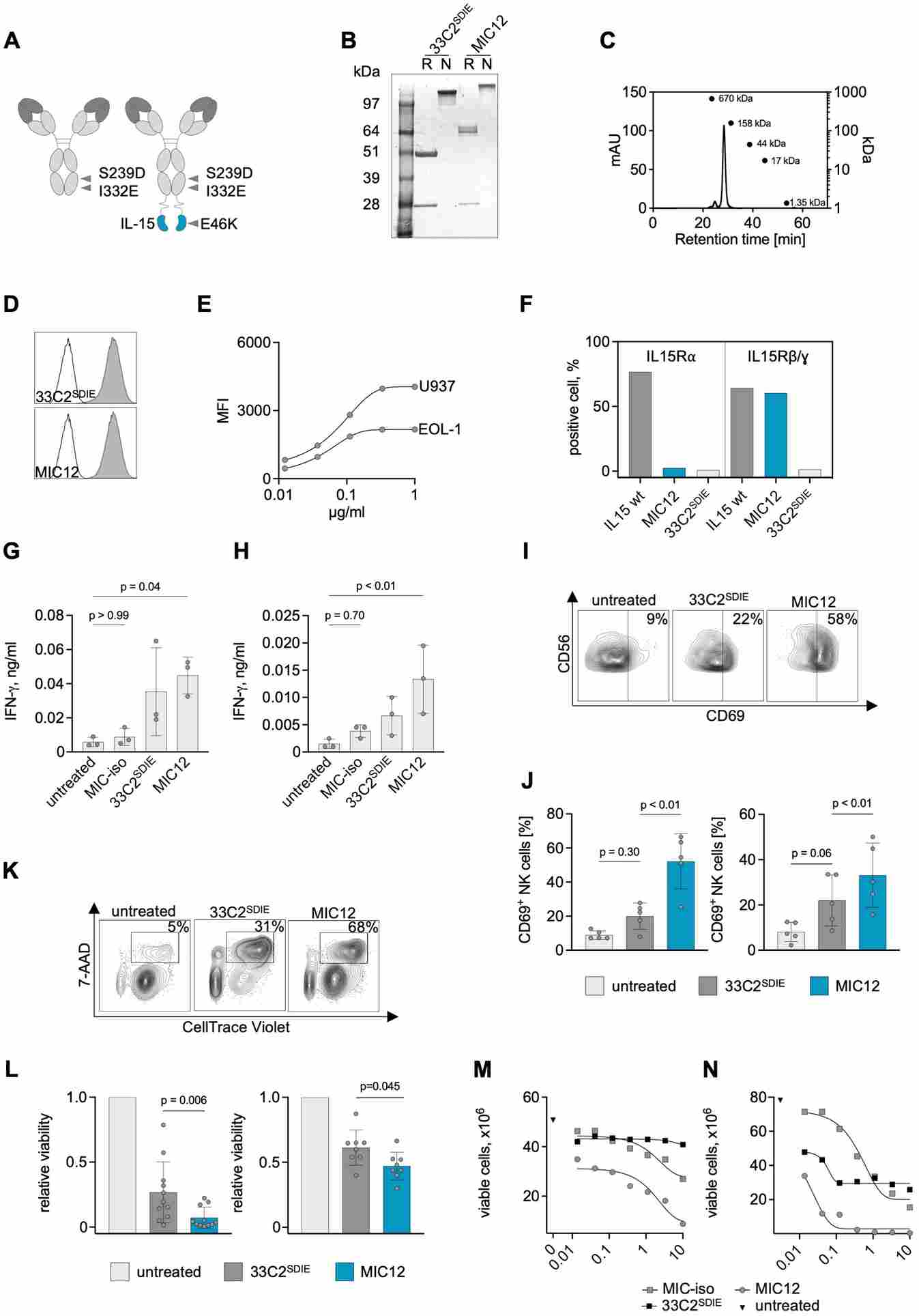
Figure 2. NK cell activation and cytokine secretion induced by target cell-restricted immunocytokine. CD69 upregulation on NK cells (left) and IFN-γ ELISA data (right) comparing MIC+ with Fc-optimized antibody alone. MIC+ selectively amplifies NK activation only in the presence of antigen-positive target cells—a paradigm validated using NALM-6 Luc BLI models in vivo.
Validated In Vivo with NALM-6 Luc Bioluminescence Imaging
The preclinical in vivo efficacy of MIC+ was evaluated using NSG mice engrafted via tail-vein injection with luciferase-expressing human NALM-6 cells (NALM-6 Luc). Bioluminescence imaging was performed serially to track leukemia dissemination and treatment response across the whole body—bone marrow, spleen, liver, and CNS—without terminal sacrifice. Key findings from the NALM-6 Luc xenograft experiments included:
• Superior tumor suppression: MIC+ (anti-CD19-IL-15[L45E/E46K]) showed significantly lower BLI signal than Fc-optimized anti-CD19 alone at all time points, demonstrating enhanced NK-mediated leukemia clearance.
• Target-dependent activity: MIC+ constructs were inactive against CD19/CD20-negative control cell lines, confirming that IL-15 stimulation is strictly contingent on antibody-antigen engagement—a critical safety feature.
• NK cell expansion in vivo: Peripheral blood NK cell counts were significantly higher in MIC+-treated mice compared to antibody-only or PBS controls, reflecting the proliferative IL-15 signal delivered exclusively at the leukemia site.
• Survival benefit: Mice treated with MIC+ showed extended overall survival versus controls in the NALM-6 Luc disseminated leukemia model.
These findings underscore the irreplaceable value of NALM-6 Luc in demonstrating both efficacy and mechanism in a physiologically relevant, disseminated leukemia model. BLI allowed the team to distinguish early leukemia control from complete remission and to monitor potential CNS involvement—features impossible to capture with endpoint-only tumor weight measurements.
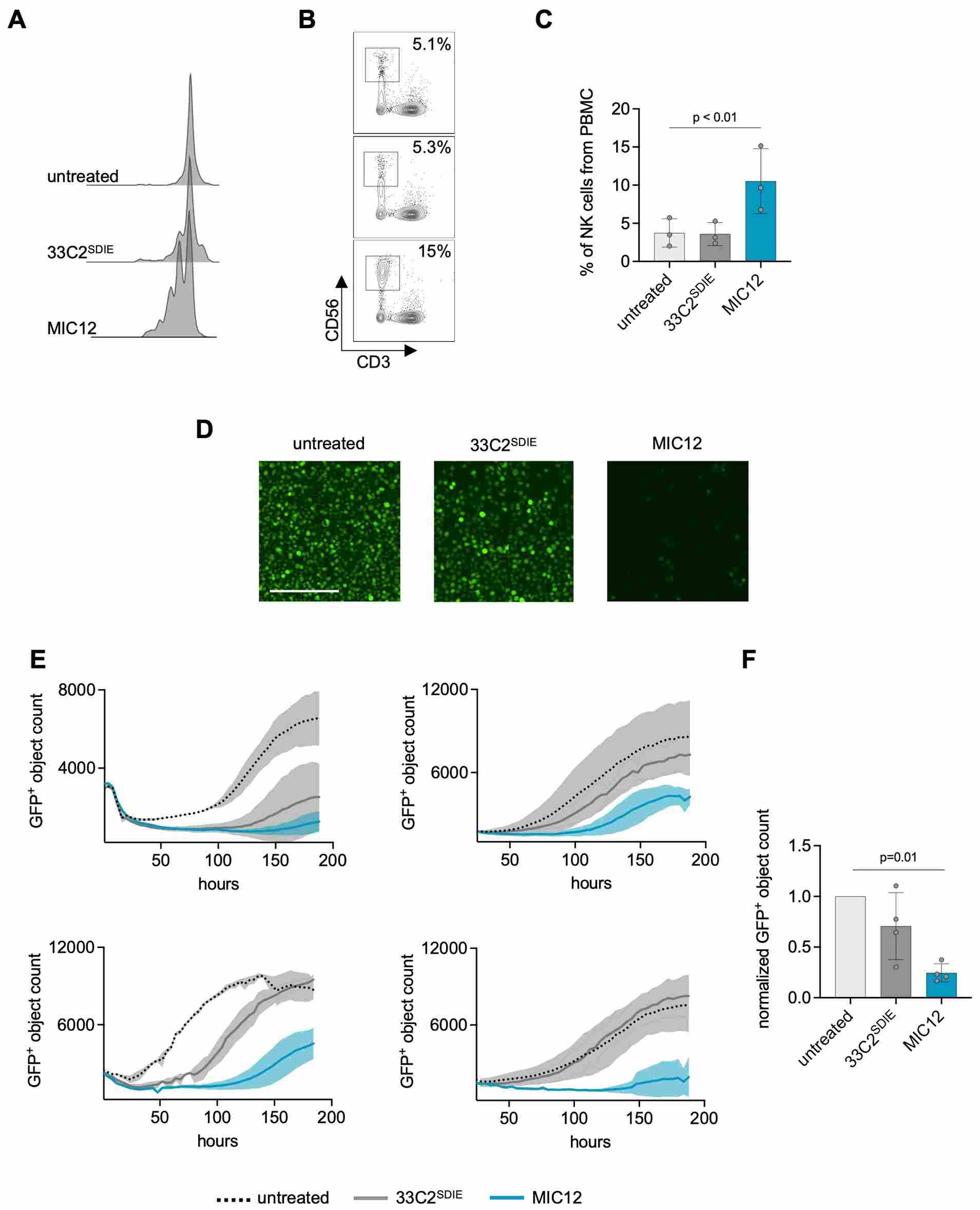
Figure 3. Anti-leukemia cytotoxicity and NK cell proliferation driven by immunocytokine treatment. Flow-cytometry-based cytotoxicity assay (left) and CellTrace Violet-based NK proliferation data (right) from co-culture experiments with MIC+. The NALM-6 Luc cell line provides the CD19/CD20-positive target cell scaffold for both in vitro and BLI in vivo readouts.
Broadening the MIC+ Platform: From NALM-6 Luc to Primary ALL and Solid Tumors
Beyond NALM-6 Luc, the study validated MIC+ activity against:
• Primary patient ALL cells: Fresh bone marrow blasts from B-ALL patients responded to MIC+ treatment with robust NK-mediated killing ex vivo, supporting clinical translatability.
• EL-4 murine lymphoma cells transduced with human CD19/CD20: These syngeneic-like models allowed testing with mouse NK cells and demonstrated that the MIC+ mechanism is conserved across species, facilitating toxicology studies.
• Anti-CD20 MIC+ variants: CD20-targeting MIC+ constructs showed analogous target-restricted activity against CD20+ B-ALL lines, extending therapeutic coverage to a broader antigen landscape.
The successful translation from NALM-6 Luc in vitro screening to NSG in vivo BLI validation to primary patient material exemplifies best-in-class preclinical development pipeline design. Investigators integrating NALM-6 Luc at each step of this workflow benefit from consistent, quantifiable tumor burden data that bridges cellular pharmacology and whole-animal PK/PD modeling.
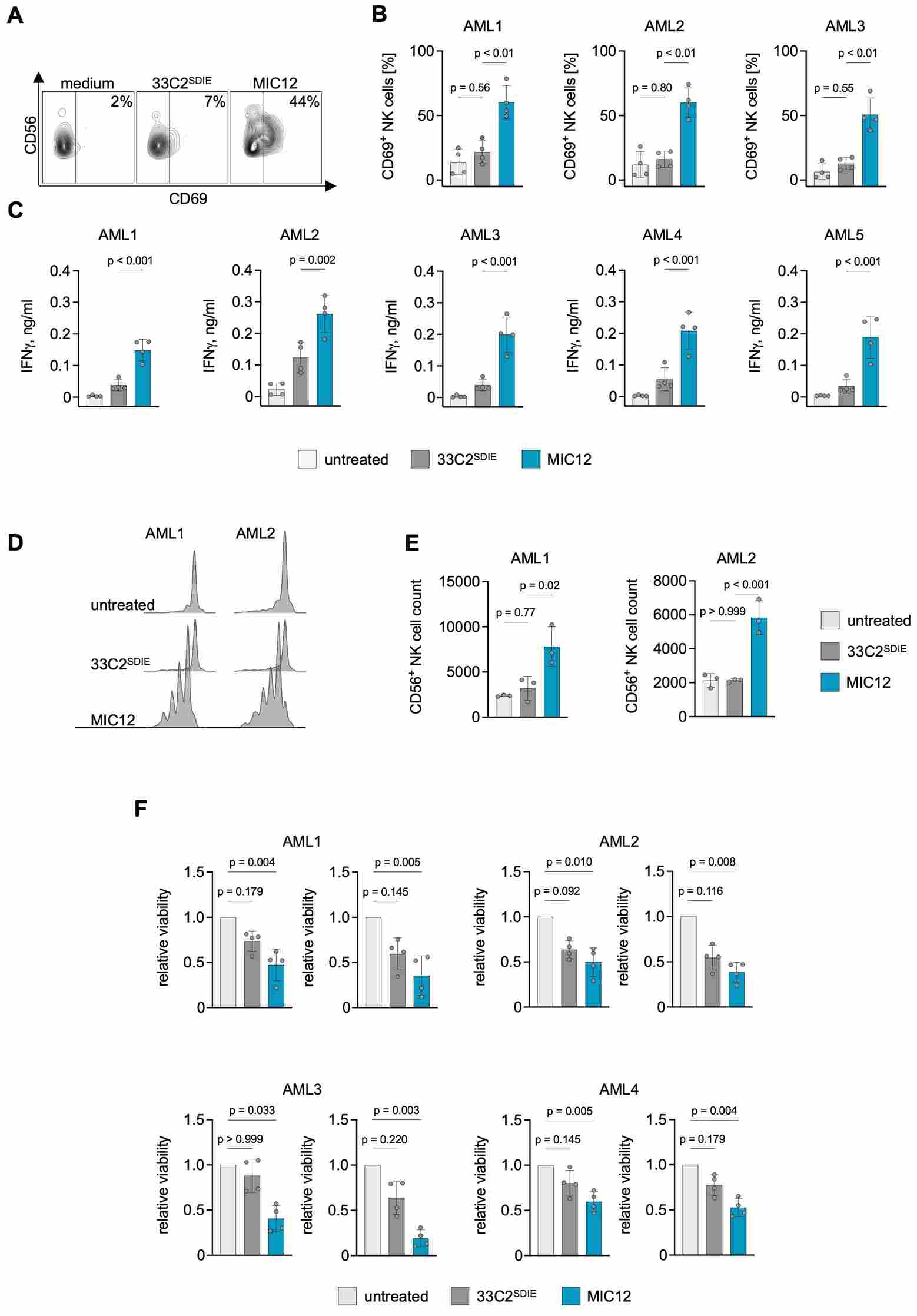
Figure 4. Long-term anti-leukemia efficacy in extended co-culture experiments. IncuCyte-based continuous monitoring of leukemia cell confluence in the presence of immunocytokine plus NK cells versus controls. NALM-6 Luc BLI data in vivo mirrors these kinetics, providing convergent evidence for durable NK-mediated remission.
Mechanistic Insights: Why Conditional IL-15 Outperforms Constitutive Activation
Conventional cytokine immunotherapy strategies face a fundamental paradox: systemic cytokine administration activates immune cells indiscriminately, driving severe toxicities (cytokine release syndrome, vascular leak) that limit dose and duration. MIC+ sidesteps this by functionally coupling IL-15 activity to tumor antigen recognition. The mechanistic advantage is three-fold:
1. Spatial restriction: IL-15 signaling is amplified only at tumor-NK cell conjugates, sparing distal tissues.
2. NK cell proliferation: Unlike Fc-optimized antibodies, which activate but do not expand NK cells, MIC+ drives measurable NK cell proliferation through sustained IL-15Rβγ engagement—functionally reconstituting the "missing" IL-15Rα trans-presentation in the tumor niche.
3. Reduced regulatory T cell activation: Because MIC+ does not engage IL-2Rγ at physiological cytokine concentrations, Treg expansion is minimized, preserving an anti-tumor immune balance—a key advantage over IL-2-based fusion proteins.
The NALM-6 Luc model was critical in revealing these in vivo dynamics: BLI-quantified tumor burden correlated directly with peripheral NK cell expansion kinetics, establishing a pharmacodynamic biomarker framework for clinical translation of MIC+.
Discussion and Translational Outlook
The MIC+ platform represents a convergence of antibody engineering, cytokine biology, and immuno-oncology—addressing the clinical gap between incomplete ADCC (Fc-optimized antibodies) and non-specific toxicity (soluble IL-15). By demonstrating potent anti-leukemia activity in NALM-6 Luc disseminated xenograft models with superior NK expansion and survival benefit, this study provides a compelling preclinical case for MIC+ clinical development in relapsed/refractory B-ALL.
The study also highlights the broader applicability of the MIC architecture: the same conditional IL-15 module has since been adapted for CLEC12A (AML, 2025 Frontiers in Immunology) and potentially other antigen-antibody pairs, illustrating a modular plug-and-play immunocytokine platform. As next-generation cell therapies and antibody formats compete for dominance in B-ALL, MIC+ offers a bispecific-free, manufacturing-accessible approach that augments existing NK biology rather than replacing it.
For researchers designing preclinical B-ALL studies, choice of in vivo model is pivotal. The NALM-6 Luc cell line offers: (i) high CD19/CD20 expression amenable to antibody-based targeting; (ii) efficient engraftment in NCG/NSG/NSG-SGM3 mice via tail-vein injection; (iii) rapid bone marrow infiltration mimicking human B-ALL dissemination; and (iv) robust BLI signal for non-invasive, real-time quantification of leukemia burden and treatment response.
Vitrobiotech's NALM-6 Luc cell line is your gold-standard tool for in vivo B-ALL immunotherapy studies — validated BLI activity, STR-authenticated, and ready for NSG/NCG tail-vein engraftment. Request a quote now or place your order.
Order now: NALM-6 Cell Line / NALM-6 Luciferase Cell Line / NALM-6-Luc-GFP Cell Line
Conclusion
This Science Translational Medicine 2024 study establishes MIC+ immunocytokines as a paradigm-shifting approach that unites the precision of tumor antigen targeting with the regenerative power of IL-15-driven NK cell expansion. The use of NALM-6 Luc in disseminated NSG xenograft models provided the critical in vivo proof-of-concept—demonstrating target-restricted IL-15 activity, robust NK expansion, and superior leukemia suppression compared to current standards. As the immunocytokine field advances toward clinical trials, NALM-6 Luc will remain an indispensable tool for evaluating candidate therapies, characterizing immune-tumor dynamics, and establishing BLI-based pharmacodynamic endpoints that translate to clinical MRD monitoring.
References
1. Zekri L, Hagelstein I, Märklin M, et al. Immunocytokines with target cell-restricted IL-15 activity for treatment of B cell malignancies. Science Translational Medicine. 2024;16(737):eadh1988. PMID: 38446900.

