PC-3-Luc: Complete Tumor Eradication via Magnetosome-Mediated Nanothermia in Prostate Cancer Models
Introduction
Prostate cancer (PCa) remains one of the most frequently diagnosed malignancies in men worldwide. Despite advances in surgery, radiotherapy, cryotherapy, and high-intensity focused ultrasound (HIFU), current treatments carry significant side effects and require costly hospital infrastructure. The search for minimally invasive, tumor-selective therapeutic modalities continues to drive innovation in cancer nanomedicine.
A groundbreaking 2025 study published in Advanced Therapeutics by El Hedjaj and colleagues introduced a novel treatment strategy using magnetosomes—natural iron oxide nanoparticles produced by magnetotactic bacteria—excited by low-intensity ultrasound (LIU) to achieve localized hyperthermia in prostate tumors. Central to the in vivo validation of this approach were PC-3-Luc reporter cells, which enabled real-time bioluminescence imaging (BLI) monitoring of tumor response throughout the treatment course.
The PC-3-Luc cell line, derived from an androgen-independent human prostate adenocarcinoma, stably expresses firefly luciferase, providing a sensitive, non-invasive readout of viable tumor burden. In this study, 152 athymic nude mice bearing subcutaneous PC-3-Luc xenografts received intratumoral injections of carboxy-methyl-dextran-coated magnetosomes (M-CMD), followed by ultrasound treatment at varying intensities. The results were remarkable: at 1 W cm⁻², 100% of treated mice experienced complete tumor disappearance with no regrowth during 60 days of follow-up.
Advancing cancer nanotherapy research requires reliable in vivo tumor models. Our validated PC-3-Luc cell line provides stable luciferase expression for real-time tumor monitoring.
Order now: PC-3 Luciferase Cell Line
Magnetosome Nanoparticles: Nature’s Superior Magnetic Agents
Unlike synthetic iron oxide nanoparticles, magnetosomes are produced biologically by magnetotactic bacteria (MSR-1 strain), resulting in superior physicochemical properties. The fabrication process involves a two-step bacterial culture: a 9-day iron-free pregrowth phase followed by 4 days of iron-supplemented fed-batch fermentation to stimulate magnetosome production. After extraction via alkaline lysis at 80°C and magnetic separation, the magnetosome minerals are purified through heat treatment and coated with carboxy-methyl-dextran (CMD) to form the final M-CMD product.
Transmission electron microscopy (TEM) characterization revealed chain-organized magnetosomes with a core diameter of 36.4 ± 8.1 nm and a coating thickness of 2.5 ± 0.5 nm. The CMD coating dramatically improved suspension stability, with M-CMD maintaining consistent absorbance for over 7,000 seconds, whereas uncoated magnetosomes rapidly sedimented. Notably, M-CMD achieved 96% iron purity by ICP-MS analysis, with only trace amounts of magnesium (2.8%), silica (0.8%), and aluminum (0.1%). Endotoxin levels were reduced to 90 EU mL⁻¹ mg⁻¹, meeting nonpyrogenic safety standards for biomedical applications.
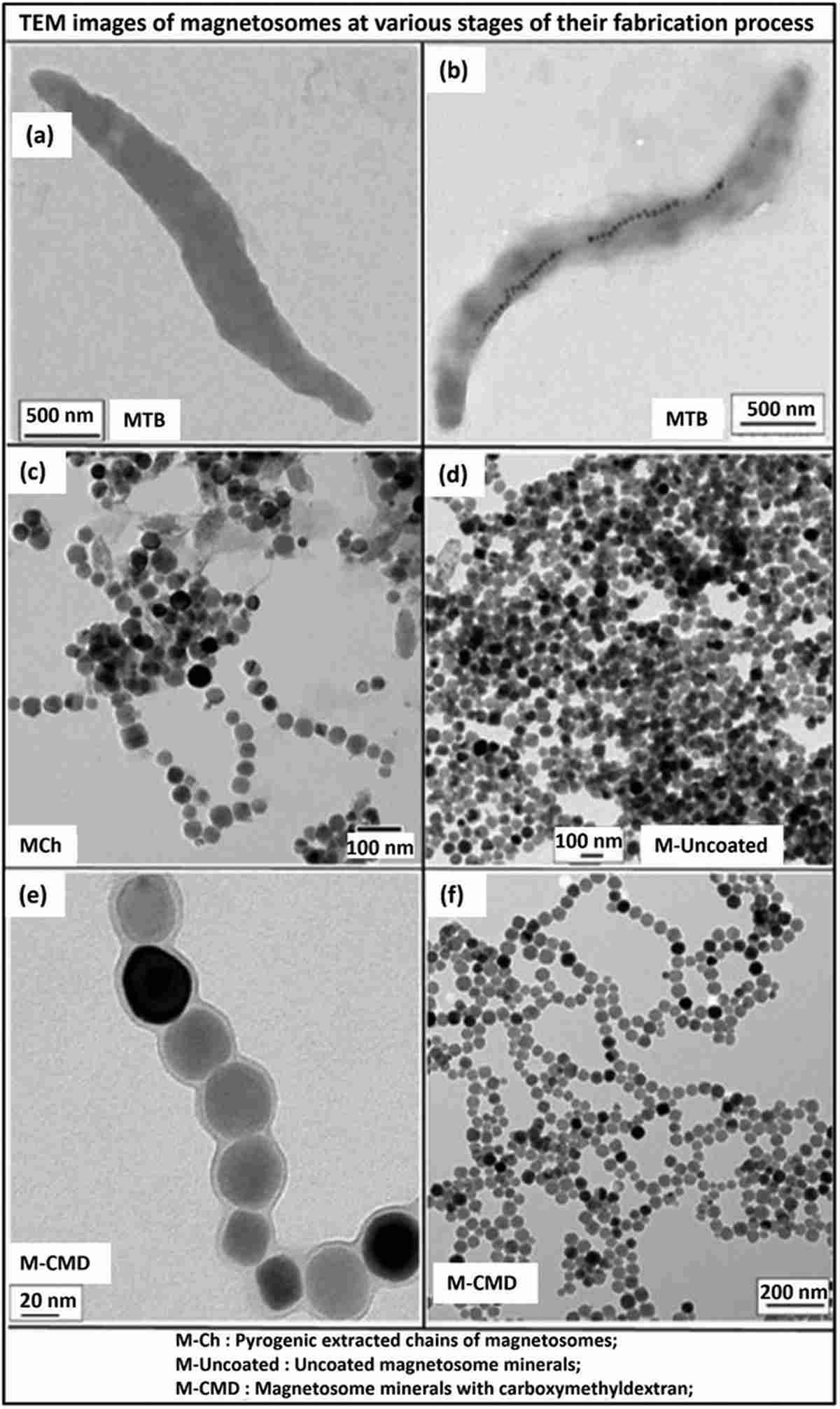
Figure 1. TEM images of magnetosome fabrication and purification. (A–B) Magnetotactic bacteria before and after iron-supplemented growth. (C) Extracted magnetosome chains (M-Ch). (D) Purified individual magnetosome minerals. (E–F) CMD-coated magnetosomes (M-CMD) showing well-dispersed chain organization at different magnifications.
In Vitro Validation: Selective Thermal Cytotoxicity
Before proceeding to in vivo studies, the research team first validated the LIU/M-CMD treatment in cultured PC-3-Luc cells. At an ultrasound intensity of 1 W cm⁻² and M-CMD concentration of 500 µg mL⁻¹, the treatment raised cell culture temperature from 25°C to 45–46°C within minutes and maintained this hyperthermic range throughout the 20-minute exposure. The resazurin viability assay demonstrated approximately 30% tumor cell death—a selective cytotoxic effect entirely dependent on the presence of magnetosomes.
Flow cytometry with propidium iodide and Annexin V staining revealed that cell death occurred predominantly via necrosis rather than apoptosis (28.2% necrotic cells with LIU/M-CMD vs. 0.58% in untreated controls). This finding is consistent with the thermal damage mechanism expected from hyperthermia treatment. ICP-MS analysis confirmed that PC-3-Luc cells internalized 26 ± 3.5 pg of iron per cell from magnetosomes, sufficient to act as intracellular sonosensitizers that amplify the ultrasound-mediated heating effect.
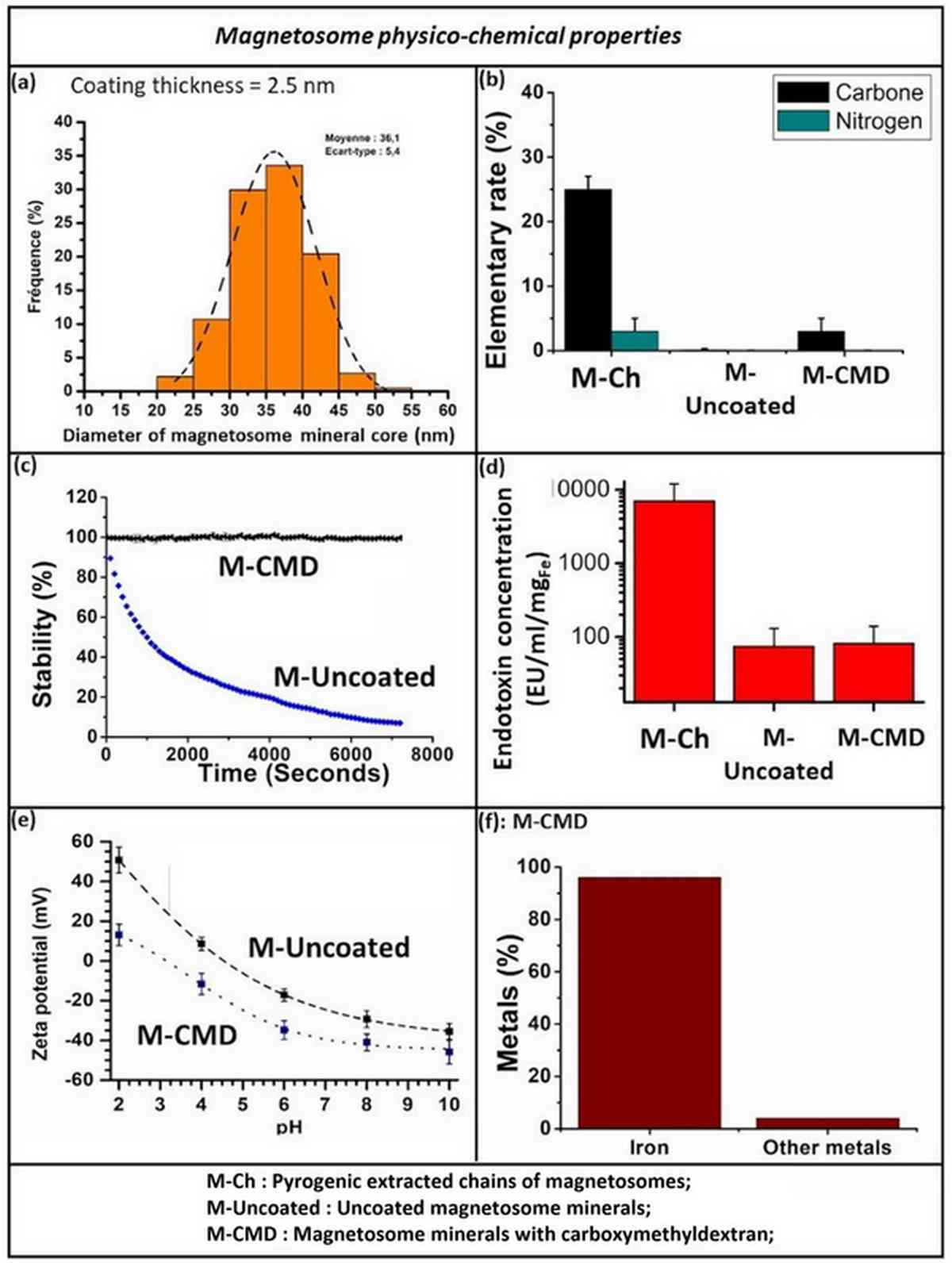
Figure 2. Characterization and in vitro treatment data for M-CMD. (A–B) Temperature elevation profiles showing stabilization at 45–46°C during LIU exposure. (C) Resazurin viability assay demonstrating 30% cell death with M-CMD + LIU versus 0% without nanoparticles. (D) Flow cytometry quantification of necrotic cell populations after treatment.
In Vivo Efficacy: Dose-Dependent Tumor Eradication
The in vivo study employed a rigorous dose-response design using PC-3-Luc xenograft-bearing athymic nude mice. Intratumoral M-CMD was administered at a dose of 3 µg per mm³ of tumor volume—approximately eightfold lower than doses previously required in magnetic hyperthermia studies for glioblastoma. Mice were then treated with low-intensity ultrasound at three different power densities: 0.2, 0.5, and 1 W cm⁻².
The results revealed a clear intensity-dependent therapeutic gradient. At 0.2 W cm⁻² (intratumoral temperature 34–38°C), no antitumor effect was observed, and tumors grew exponentially to over 1,100 mm³. At 0.5 W cm⁻² (41–44°C), tumor growth was delayed but not halted, requiring eventual euthanasia. At 1 W cm⁻² (43–46°C), the combination of M-CMD and LIU achieved complete tumor disappearance in all treated animals. Control groups receiving ultrasound alone at 1 W cm⁻² showed only transient tumor regression followed by rapid regrowth, confirming that the magnetosomes are essential for durable therapeutic efficacy.
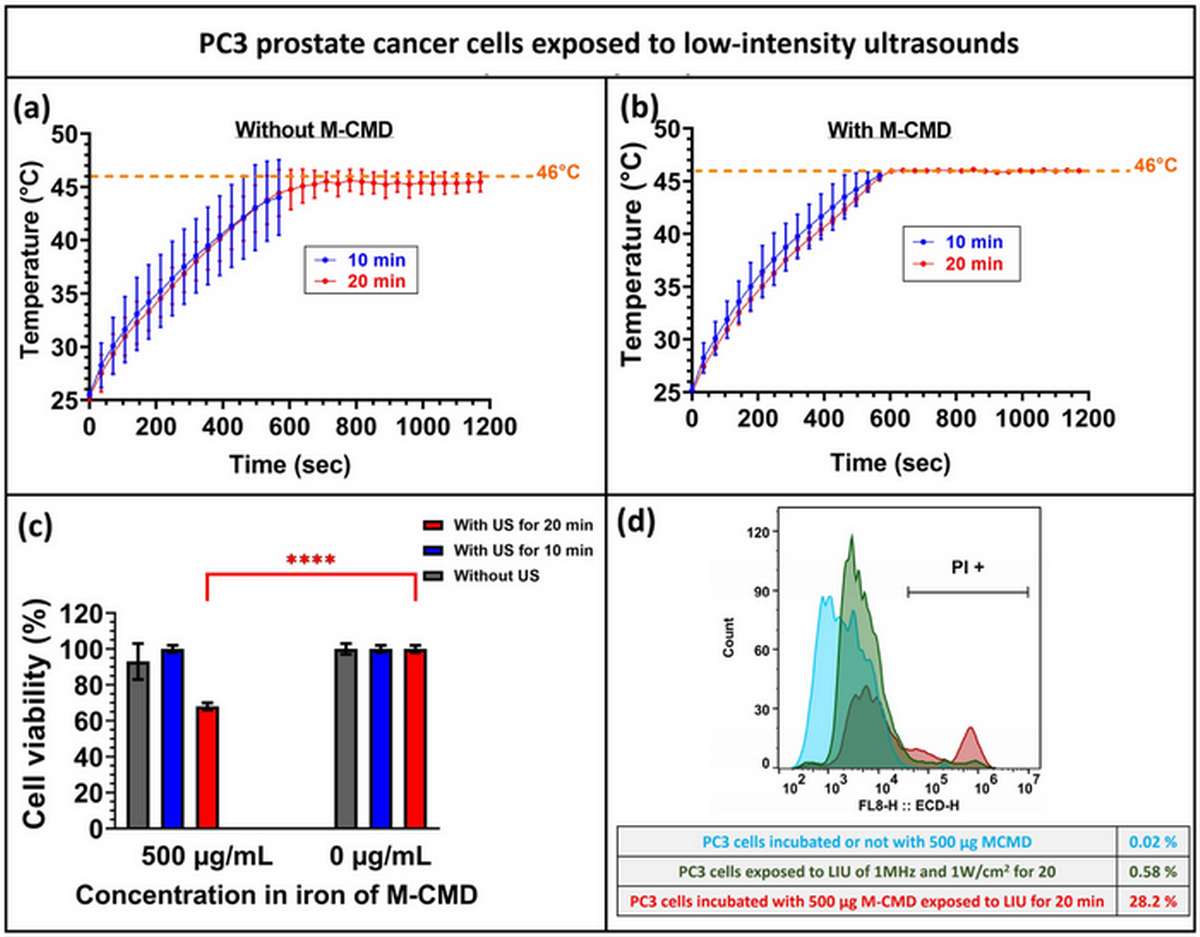
Figure 3. In vivo temperature monitoring and tumor volume evolution at 0.2 W cm². (A,C) Intratumoral and rectal temperature profiles. (B,D) Corresponding tumor volume curves demonstrating no therapeutic effect at this sub-therapeutic intensity.
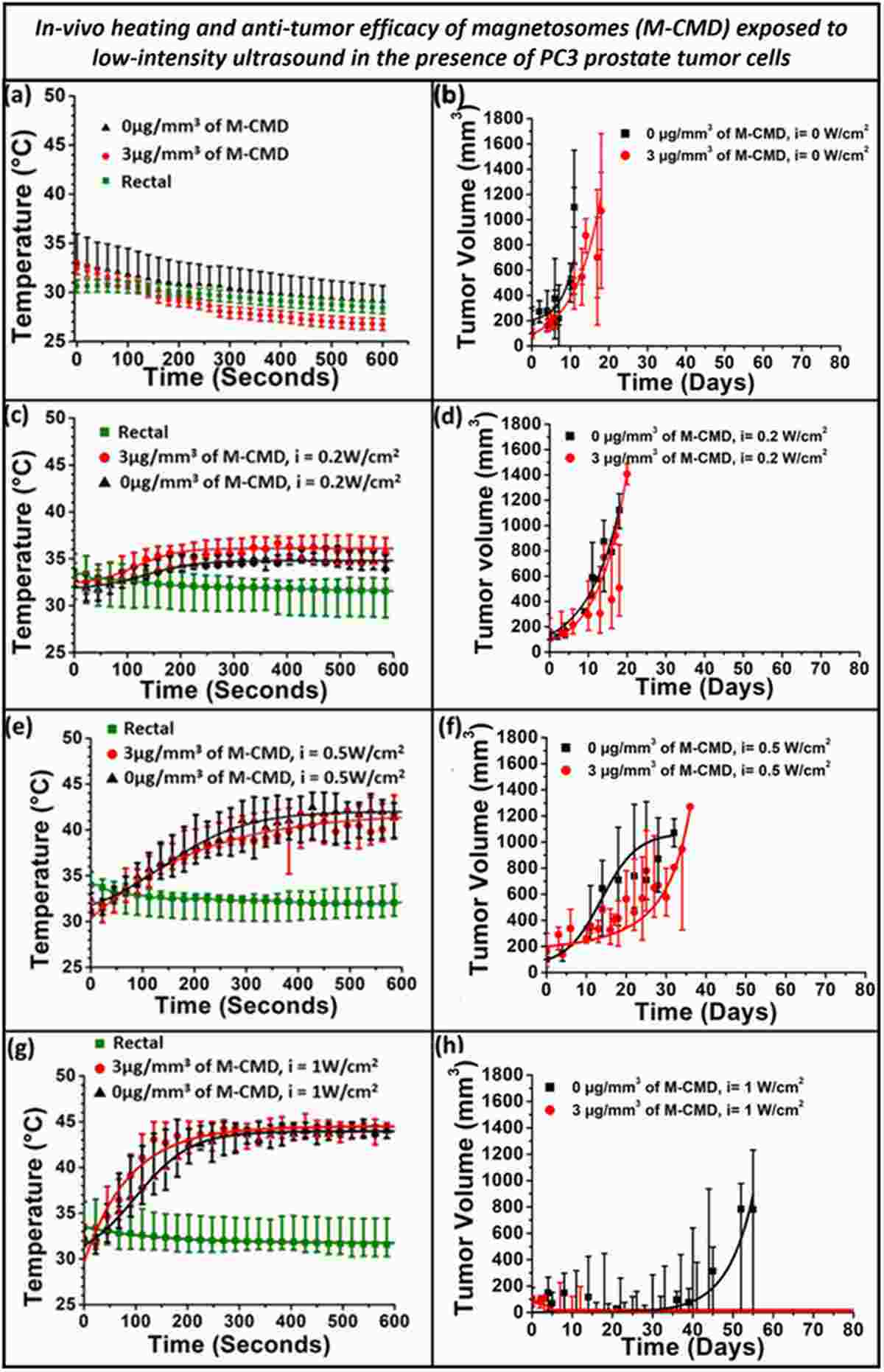
Figure 4. In vivo temperature monitoring and tumor volume evolution at 0.5 W cm². (A,C) Temperature profiles showing partial hyperthermic response. (B,D) Tumor volume curves demonstrating growth delay but incomplete tumor control.
Our PC-3-Luc cells enable precise quantification of tumor response to novel therapeutics through bioluminescence imaging. Whether evaluating nanomedicine, immunotherapy, or combination treatments, PC-3-Luc delivers reliable, real-time in vivo data.
Order now: PC-3 Luciferase Cell Line
Complete Tumor Disappearance and Long-Term Survival
The most striking finding of this study was the complete and sustained eradication of PC-3-Luc tumors in the optimal treatment group (M-CMD + 1 W cm⁻² LIU). Tumor volumes in this group decreased progressively from approximately 500 mm³ to zero, remaining at undetectable levels through day 120 of observation. Bioluminescence imaging confirmed the absence of viable luciferase-expressing tumor cells throughout the follow-up period.
In contrast, mice receiving ultrasound alone at 1 W cm⁻² initially showed tumor regression but experienced exponential regrowth beginning around day 32 post-treatment. Survival analysis demonstrated 100% survival in the M-CMD + LIU group at 60 days post-treatment, while progressive euthanasia was required in all other groups due to uncontrolled tumor burden. These results underscore the synergistic interaction between magnetosome sonosensitization and ultrasound-mediated hyperthermia—neither modality alone achieved comparable outcomes.
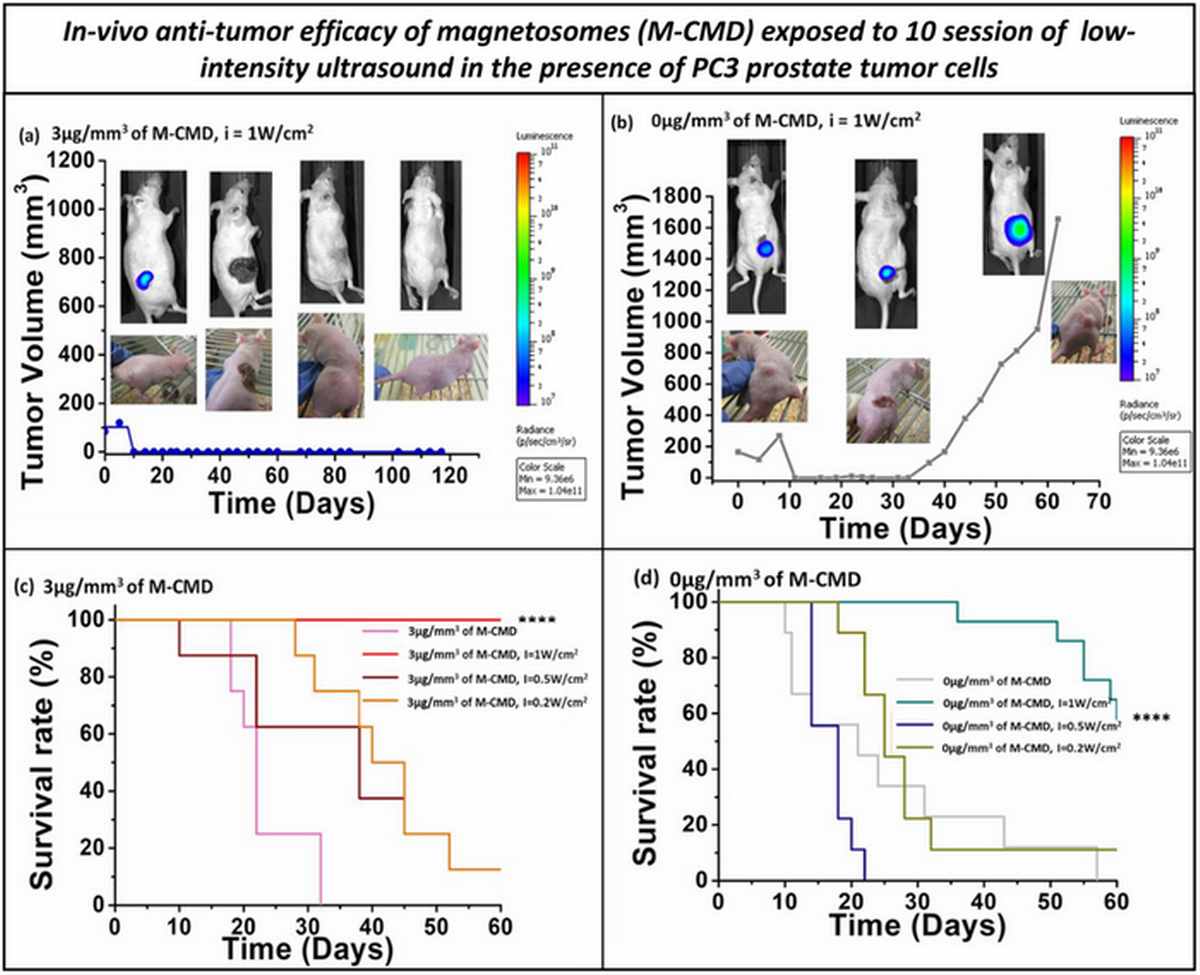
Figure 5. In vivo temperature monitoring and tumor volume evolution at 1 W cm². (A,C,E,G) Intratumoral and rectal temperature profiles for M-CMD + LIU and LIU-only groups. (B,D,F,H) Corresponding tumor volume curves demonstrating complete eradication with M-CMD + LIU versus regrowth with LIU alone.
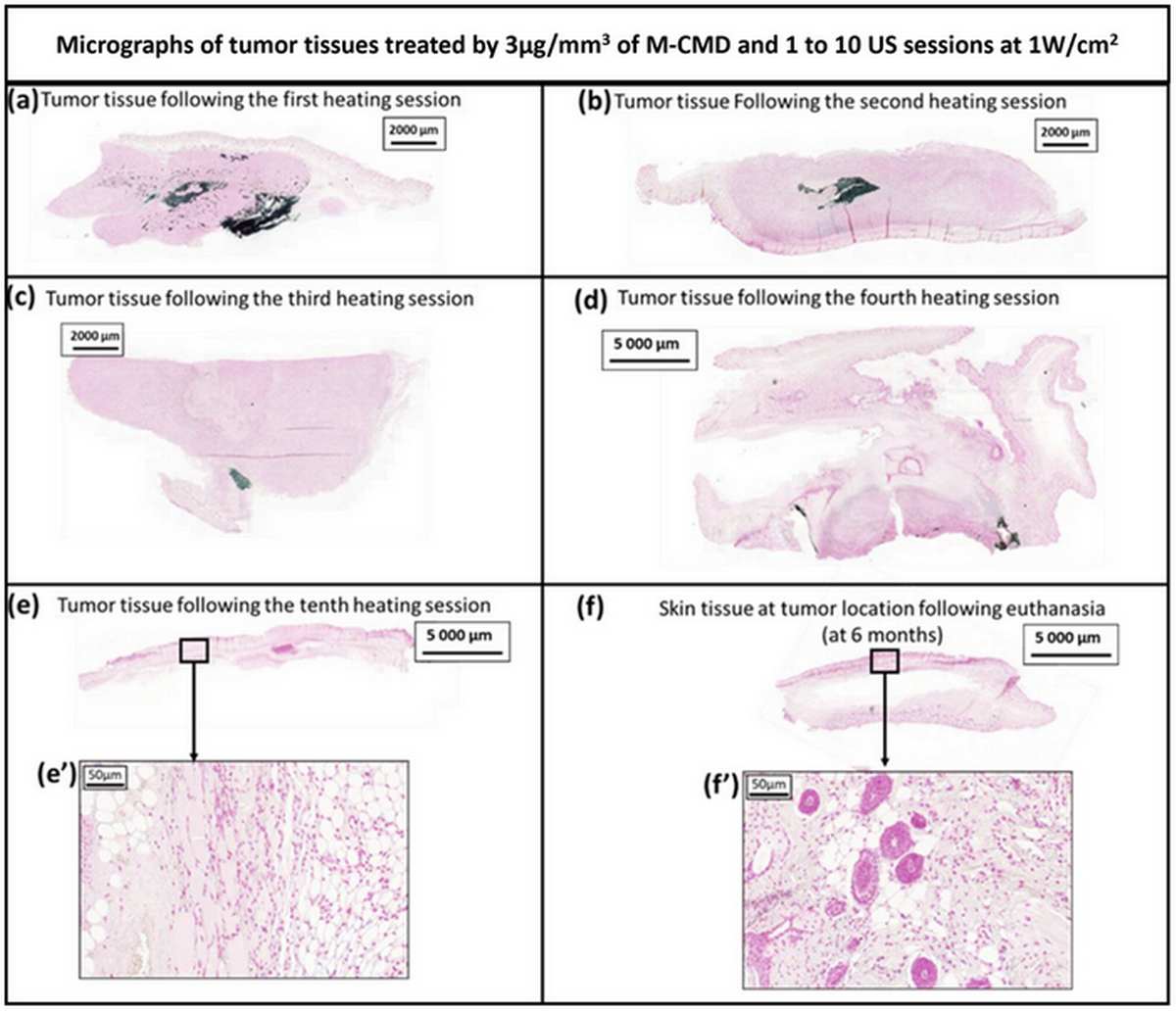
Figure 6. Bioluminescence imaging of PC-3-Luc tumor response. Representative BLI images and quantitative photon flux data showing complete loss of luciferase signal in M-CMD + LIU treated animals, confirming total tumor cell eradication, versus persistent signal in control groups.
Safety Profile and Magnetosome Biodistribution
Long-term safety assessment provided reassuring results. Perls staining of tissue sections collected at multiple time points (S1, S4, S10) and at 6 months post-treatment revealed a heterogeneous, disseminated distribution of iron-positive magnetosomes in the tumor bed at early time points, but complete absence of magnetosomes in the treated tissue at 6 months. Critically, no magnetosome accumulation was detected in the liver, spleen, lung, brain, or kidney at any time point.
Histological examination of all major organs at 6 months revealed no lesions, fibrosis, or pathological changes. Animals maintained normal body weight and behavior throughout the study. Additionally, in vitro cytotoxicity testing showed that M-CMD was non-toxic to L929 fibroblast cells at concentrations up to 1 mg mL⁻¹, and genotoxicity testing on the V79-4 cell line confirmed no DNA damage. These comprehensive safety data support the translational potential of magnetosome-mediated nanothermia as a clinically viable treatment modality.
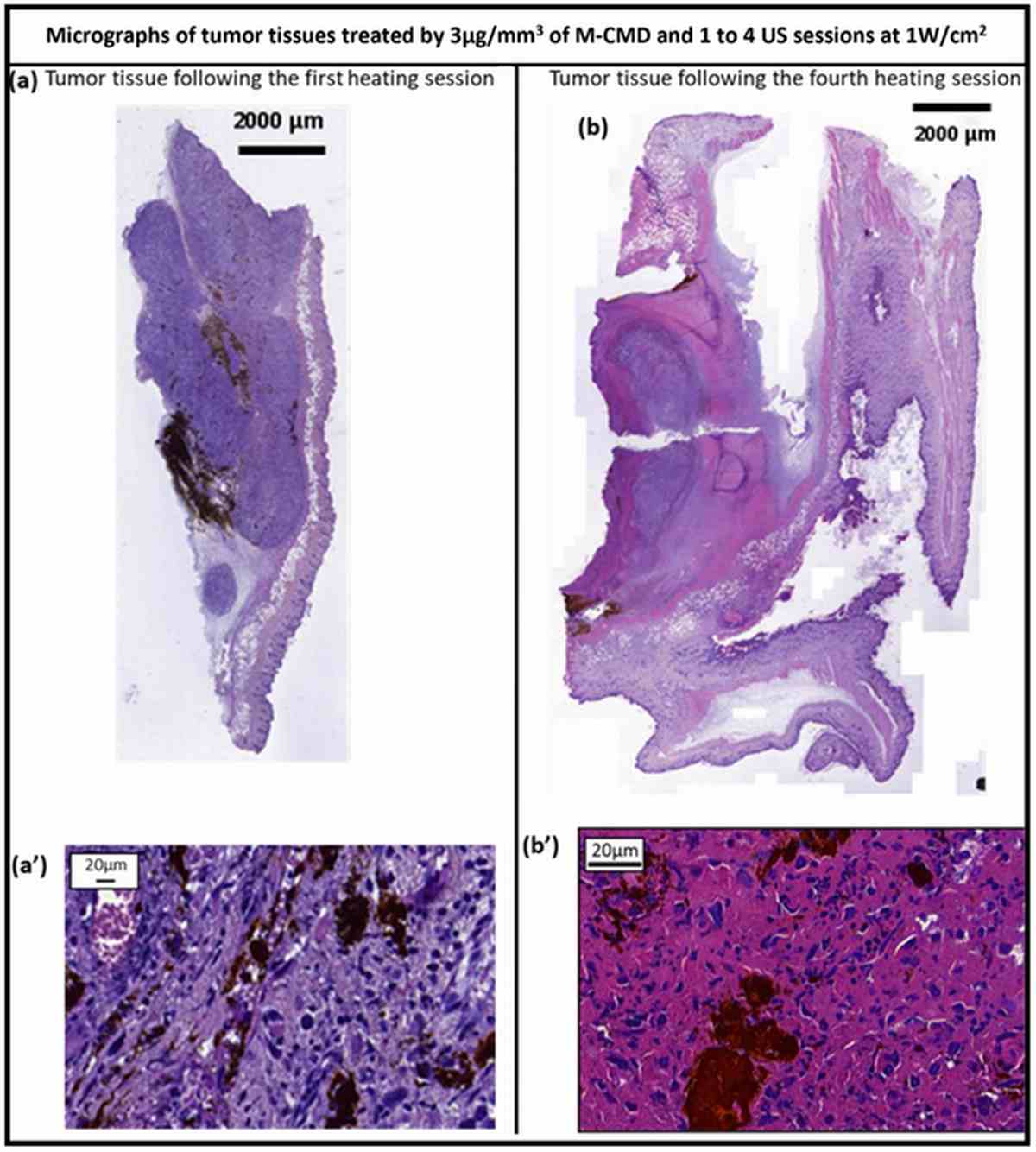
Figure 7. Histological analysis of magnetosome biodistribution via Perls staining. Iron-positive magnetosomes showed heterogeneous distribution at early treatment sessions (S1–S4) but were completely cleared by S10 and undetectable at 6 months post-treatment. No accumulation was observed in liver, spleen, lung, brain, or kidney.
Clinical Translation Potential and Advantages Over Existing Modalities
The magnetosome-mediated LIU approach offers several distinct advantages over current hyperthermia-based treatments. Compared to conventional magnetic hyperthermia using synthetic iron oxide nanoparticles, the required nanoparticle dose is approximately 20-fold lower (3 µg mm⁻³ vs. 50–60 µg mm⁻³), reducing the risk of nanoparticle-related toxicity. The chain arrangement of magnetosomes ensures superior tumor retention compared to discrete synthetic particles.
Unlike HIFU, which targets a very small focal volume (0.5–1 mm³) and typically requires general anesthesia, the LIU/M-CMD approach covers a larger heating area (1–5 cm³) and could potentially be performed as an outpatient procedure. The use of PC-3-Luc reporter cells in the preclinical validation was instrumental in demonstrating these advantages: BLI provided quantitative, longitudinal tumor burden data that confirmed complete eradication and distinguished transient regression from durable cure—a capability that conventional caliper measurements alone could not achieve.
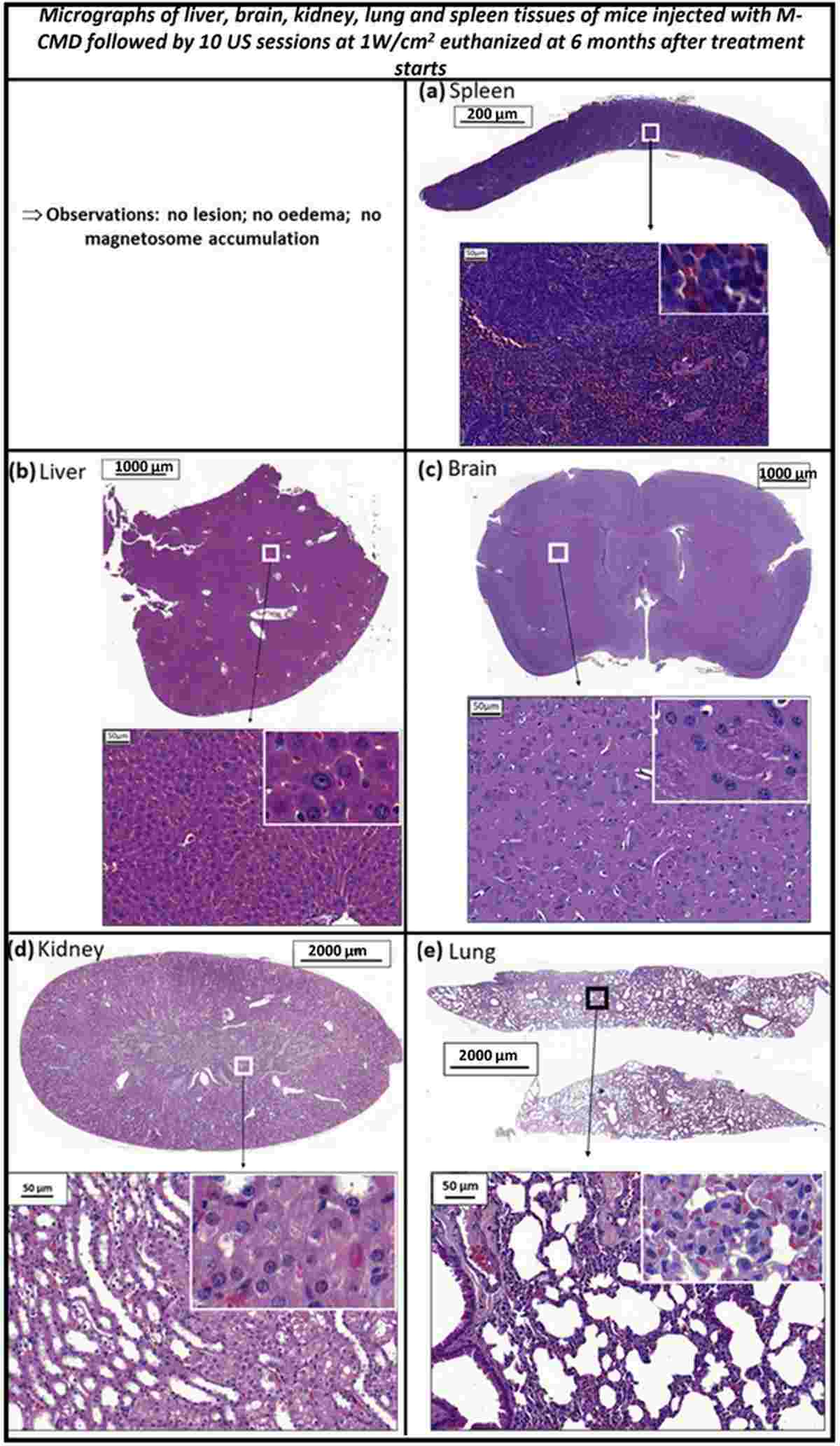
Figure 8. Schematic comparison of magnetosome-mediated LIU nanothermia versus conventional treatments. The approach requires lower nanoparticle doses, covers larger tumor volumes than HIFU, and does not require general anesthesia or hospital operating theater infrastructure, enabling potential outpatient administration.
Discussion
This study represents a significant advance in the field of cancer nanothermia, demonstrating for the first time that magnetosome-mediated hyperthermia under low-intensity ultrasound can achieve complete eradication of PC-3-Luc prostate tumors in vivo. The key innovation lies in the combination of biologically synthesized magnetosomes—which possess superior magnetic properties, chain organization, and biocompatibility compared to synthetic alternatives—with clinically feasible low-intensity ultrasound parameters.
The study’s use of PC-3-Luc cells was pivotal to the research design. Bioluminescence imaging enabled non-invasive, quantitative tracking of tumor burden across the entire treatment course, allowing the investigators to distinguish between true complete response and transient regression. This level of temporal resolution would have been unattainable with endpoint-only measurement methods, underscoring the value of reporter cell lines in preclinical therapy evaluation.
The observation that magnetosome concentration in the tumor bed naturally decreases over time—without accumulating in vital organs—addresses a major safety concern that has limited the clinical translation of nanoparticle-based therapies. The proposed future direction of PLC-controlled sequential ultrasound with active temperature stabilization at 45°C further suggests that this approach could be optimized for clinical use with real-time thermal feedback.
Conclusion
The combination of carboxy-methyl-dextran-coated magnetosomes and low-intensity ultrasound represents a promising new therapeutic platform for localized prostate cancer treatment. In PC-3-Luc xenograft models, this approach achieved 100% complete tumor disappearance with no recurrence, an excellent long-term safety profile, and several practical advantages over existing modalities including lower nanoparticle requirements, larger treatment volumes, and potential outpatient administration.
Reliable in vivo tumor models are the foundation of impactful preclinical research. Our PC-3-Luc cell line offers stable luciferase expression, high signal-to-noise ratio, and proven performance in xenograft studies.
Order now: PC-3 Luciferase Cell Line
References
El Hedjaj C, Barret E, Chebbi I, Le Fèvre R, Maake C, Guscetti F, Guyot F, Aubry JF, Seksek O, Alphandéry E. Full Disappearance of PC3-Luc Prostate Tumors Mediated by Hyperthermia Under Low Intensity Ultrasound Application in the Presence of Magnetosomes. Advanced Therapeutics. 2025;8(2). doi:10.1002/adtp.202400281

