THP-1-Luc: Enabling NF-κB-Driven Luciferase Reporter Assays for Pyrogen Detection and Innate Immunity Screening
Introduction
Pyrogens — fever-inducing contaminants including endotoxins (lipopolysaccharide, LPS) and nonendotoxin molecules (lipoteichoic acid, zymosan) — pose a safety risk in parenteral pharmaceuticals. Regulatory pharmacopoeias mandate pyrogen testing for nearly all injectable drugs. The rabbit pyrogen test (RPT), in use since the 1940s, requires live animals and yields subjective, semi-quantitative results. The monocyte activation test (MAT) offers an in vitro alternative but depends on donor-derived primary monocytes with inherent batch-to-batch variability.
A transgenic THP-1-Luc reporter cell line — stably transfected with an NF-κB-driven firefly luciferase construct — addresses both limitations simultaneously. NF-κB is the central transcription factor mediating inflammatory responses to all known pyrogen classes, making it an inclusive pyrogenic marker. The luciferase readout provides rapid, objective, and quantitative detection of NF-κB activation, directly correlating with the presence and potency of pyrogenic contaminants.
Vitrobiotech supplies validated THP-1-Luc reporter cell lines engineered for NF-κB-driven bioluminescent pyrogen detection and innate immunity screening. Contact us to discuss your research needs and receive technical support.
Order now: THP-1 Cell Line / THP-1 Luciferase Cell Line / THP-1-Luc-GFP Cell line
Establishing the THP-1-Luc Reporter Subclone for Pyrogen Detection
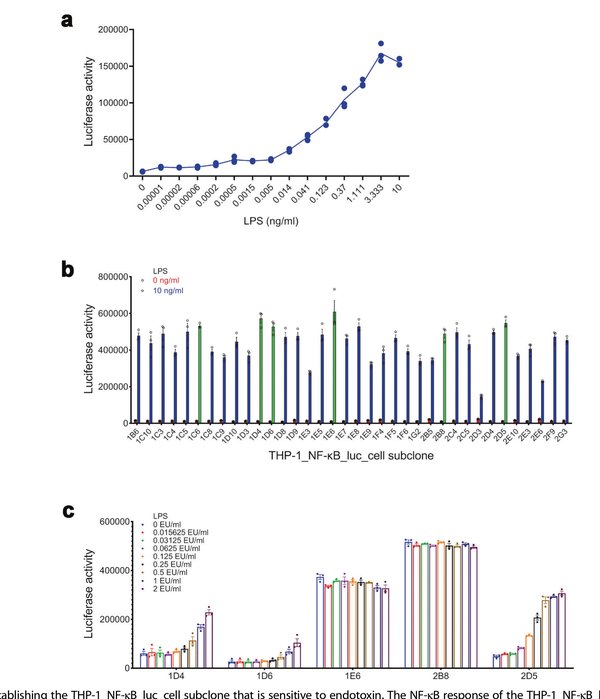
The researchers transfected the human monocytic THP-1 cell line with an NF-κB response element-driven luciferase reporter construct, generating a mixed-cell pool (THP-1_NF-κB_luc). From this pool, 36 single-cell subclones were isolated and screened for NF-κB responsiveness to LPS. The subclone designated 2D5 exhibited the optimal combination of high inducible luciferase activity and low background signal, with a dose-dependent response to LPS concentration. This subclone was selected as the working cell line for all subsequent assay development and validation experiments.
Figure 1. Establishing the THP-1_NF-κB_luc cell subclone sensitive to endotoxin. (a) NF-κB response of the mixed-cell pool to LPS stimulation. (b) Screening of 36 single-cell subclones for LPS-induced NF-κB activity. (c) Dose–response curves of selected subclones, with 2D5 showing optimal signal-to-background ratio.
Validating the THP-1-Luc Assay for Endotoxin and Nonendotoxin Pyrogens
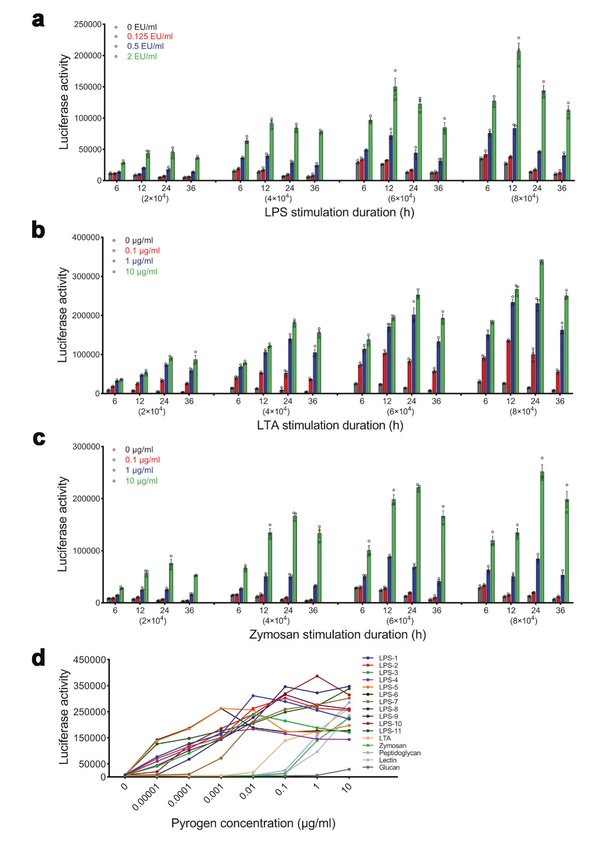
With subclone 2D5 established, the team systematically optimized assay parameters: cell density, stimulation time, and culture medium composition. A density of 4 × 10⁴ cells/well and a stimulation period of 12–24 hours yielded the highest signal-to-background ratio for both endotoxin (LPS, LTA) and nonendotoxin (zymosan) pyrogens. Critically, the assay detected all three pyrogen classes through a single NF-κB-driven luciferase readout, demonstrating its capacity as a broad-spectrum pyrogen detection platform.
The THP-1-Luc reporter cells responded to LPS in a dose-dependent manner with a detection limit of 0.0625 EU/mL, meeting pharmacopoeial sensitivity requirements. The coefficient of variation (CV) of relative light units (RLUs) remained below 20% across all tested concentrations, confirming assay reproducibility. Compared to the RPT, the THP-1-Luc assay reduced processing time from days to hours and eliminated animal use entirely.
Figure 2. Confirmation of experimental parameters and NF-κB response to endotoxin and nonendotoxin pyrogens. (a–c) NF-κB response at different cell densities to LPS (a), LTA (b), and zymosan (c). (d) Dose–response curves confirming broad-spectrum pyrogen detection.
Our THP-1-Luc cell lines deliver reproducible, quantitative NF-κB reporter data for pyrogen detection and innate immunity drug screening. Request a quote now to get started.
Order now: THP-1 Cell Line / THP-1 Luciferase Cell Line / THP-1-Luc-GFP Cell line
Long-Term Stability and Phenotypic Consistency of THP-1-Luc Cells
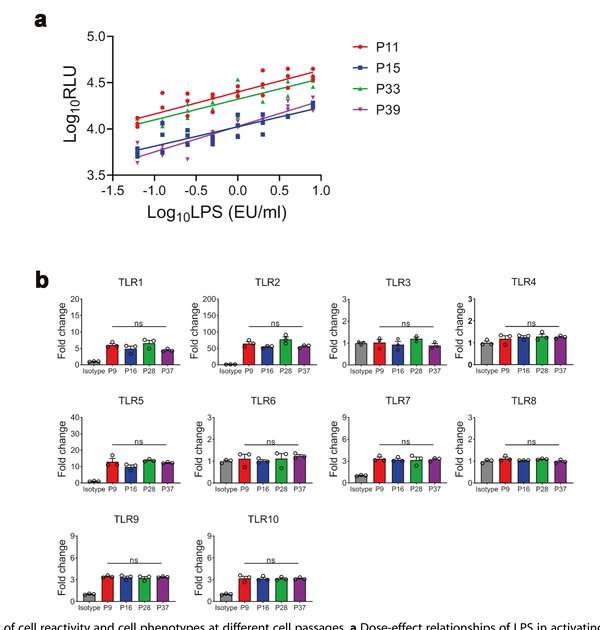
A practical concern with reporter cell lines is phenotypic drift over extended passaging. The researchers evaluated THP-1-Luc subclone 2D5 across passages 6, 16, 28, and 37. The dose–response relationship to LPS remained consistent across all passages tested, with no significant change in NF-κB inducibility. Flow cytometry analysis confirmed that the expression profile of Toll-like receptors (TLR1–10) — the pattern recognition receptors responsible for pyrogen sensing — was stable across passages, indicating that the transfected cells retained their native innate immune signaling machinery throughout extended culture.
This stability is critical for regulatory compliance: a qualified cell bank can serve as a consistent reagent for routine quality control testing, eliminating the donor variability inherent in primary cell-based MATs. The combined results — broad-spectrum pyrogen detection, pharmacopoeial sensitivity, inter-assay reproducibility, and long-term phenotypic stability — position the THP-1-Luc reporter as a validated replacement for the rabbit pyrogen test and a superior alternative to donor-dependent MATs.
Figure 3. Stability of cell reactivity and phenotypes at different passages. (a) Dose–effect relationships of LPS at passages 11, 15, 33, and 39. (b) TLR1–10 expression profiles remain consistent across passages.
Discussion: THP-1-Luc as a Versatile Platform Beyond Pyrogen Testing
While this study focused on pyrogen detection, the THP-1-Luc reporter platform has broader utility in immunology and drug discovery. The NF-κB pathway is activated by diverse innate immune stimuli beyond pyrogens — including cytokines, damage-associated molecular patterns (DAMPs), and pathogen-associated molecular patterns (PAMPs). This makes the THP-1-Luc system a versatile tool for screening anti-inflammatory compounds, characterizing TLR agonists and antagonists, and evaluating immunomodulatory drug candidates in a physiologically relevant monocytic cell context.
The modularity of the reporter construct further extends its applicability. By replacing the NF-κB response element with IRF, AP-1, or other pathway-specific regulatory sequences, the same THP-1 backbone can be adapted to monitor distinct signaling cascades. This modularity, combined with the cell line’s stable phenotype and validated performance, positions THP-1-Luc as a universal screening chassis for innate immunity research and immunopharmacology.
Conclusion
The engineering and validation of the THP-1-Luc (NF-κB-luciferase) reporter cell line demonstrates that a stable, single-cell-derived monocytic reporter can replace animal-based pyrogen tests with a rapid, quantitative, and reproducible in vitro assay. Subclone 2D5 detects all major pyrogen classes at pharmacopoeial sensitivity, maintains phenotypic stability over extended passaging, and provides results within hours. Beyond pyrogen testing, the THP-1-Luc platform offers a versatile, mechanism-based screening tool for innate immunity research, anti-inflammatory drug discovery, and immunomodulatory candidate evaluation.
Explore our catalog of validated THP-1-Luc reporter cell lines and find the perfect screening model for your immunology and drug discovery program. Browse our catalog and find the perfect cell model for your study.
Order now: THP-1 Cell Line / THP-1 Luciferase Cell Line / THP-1-Luc-GFP Cell line
References
He Q, Yu CF, Wu G, et al. A novel alternative for pyrogen detection based on a transgenic cell line. Signal Transduct Target Ther. 2024;9(1):e66. DOI: 10.1038/s41392-024-01744-0.

