U-87 MG Luc Reveals IFNγR-Dependent Immune Synapse Formation as the Key Mechanism of CAR T Cell Efficacy in Glioblastoma
Introduction
Glioblastoma (GBM) is the most lethal primary brain tumour in adults, with a median survival of 14–16 months under standard chemoradiotherapy. CAR T cell therapy has transformed treatment of haematologic malignancies, yet its efficacy against solid tumours—including GBM—has remained elusive. Understanding the tumour-intrinsic barriers to CAR T cell killing in GBM is therefore an urgent scientific priority.
To systematically identify resistance mechanisms, the Maus laboratory at Massachusetts General Hospital performed genome-wide CRISPR knockout screens in U-87 MG glioblastoma cells, revealing loss of interferon-γ receptor (IFNγR) signalling as the dominant escape mechanism. The resulting Nature paper, published 13 April 2022, demonstrates how IFNγR-driven ICAM-1 upregulation stabilises immune synapses—a finding with direct relevance to next-generation CAR T cell design and patient stratification.
Vitrobiotech's U-87 MG Luc cell line delivers stable firefly luciferase expression across passages, validated for orthotopic intracranial BLI imaging in immunodeficient mice. It is the essential in vivo platform for evaluating CAR T cell therapies, combination immunotherapy strategies, and drug delivery systems in GBM. Contact us to discuss your research needs.
Order now: U 87 MG Cell Line / U-87 MG Luciferase Cell Line
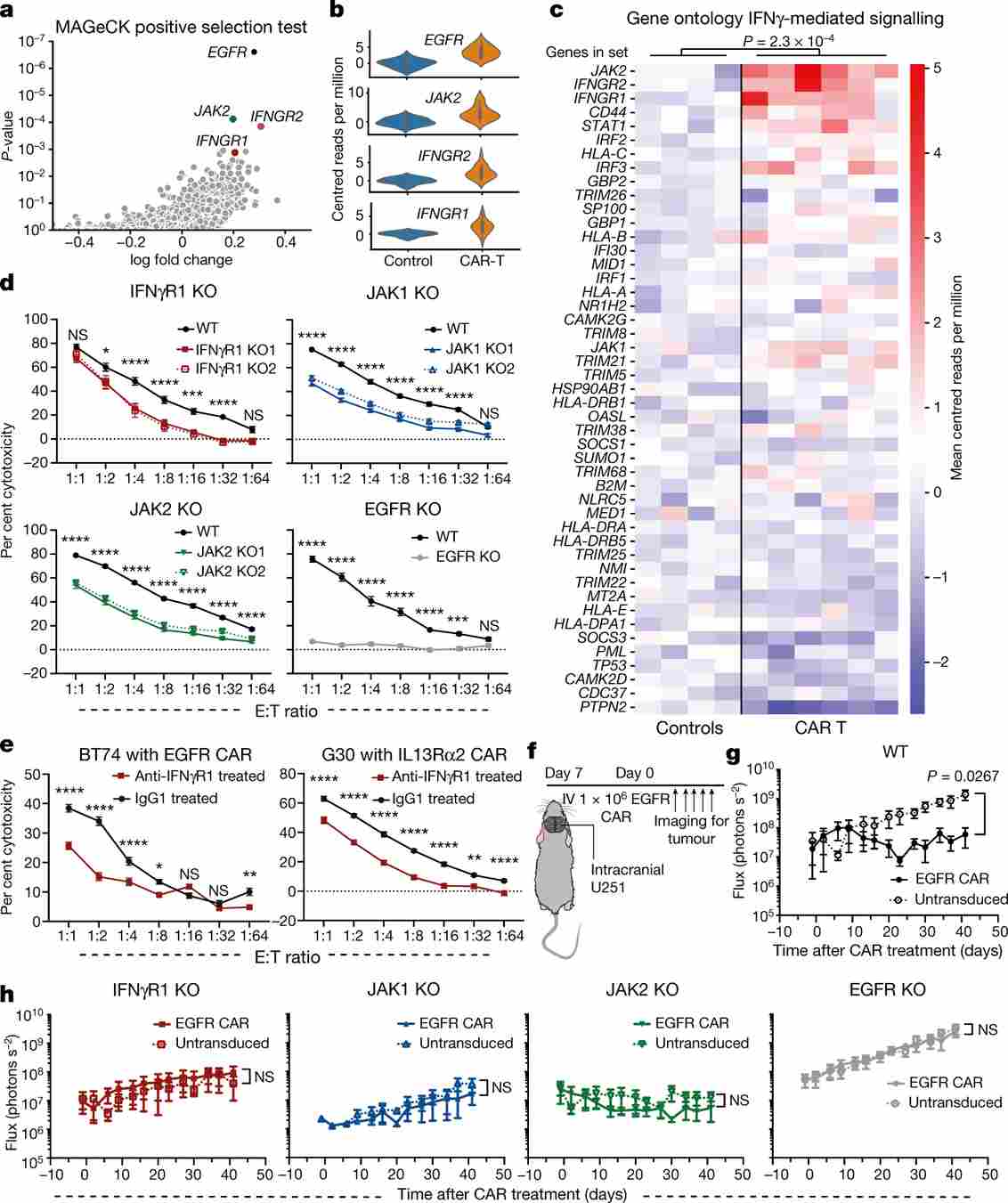
Figure 1. Genome-wide CRISPR screen in U-87 MG glioblastoma identifies loss of IFNγR signalling (IFNGR1, JAK1, JAK2) as a major resistance mechanism to CAR T cell cytotoxicity in solid but not liquid tumours.
IFNγR Signalling Is Selectively Required for CAR T Cell Killing in Solid Tumours
Using a genome-scale lentiviral CRISPR library, the authors screened U-87 MG cells co-cultured with EGFRvIII-targeting CAR T cells. Deep sequencing of surviving cells enriched guides targeting IFNGR1, JAK1, and JAK2—the core IFNγR–JAK signalling axis. No enrichment of these genes was detected when the same screen was performed in Nalm6 leukaemia cells, establishing a solid-tumour-specific dependency.
Individual IFNGR1 knockout U-87 MG cells were dramatically resistant to CAR T cells targeting EGFRvIII, EGFR, or IL13Rα2—three distinct antigens—confirming a broad, antigen-independent resistance mechanism. This dependency extended to pancreatic, lung, and other solid tumour lines, but was absent in liquid tumour models.
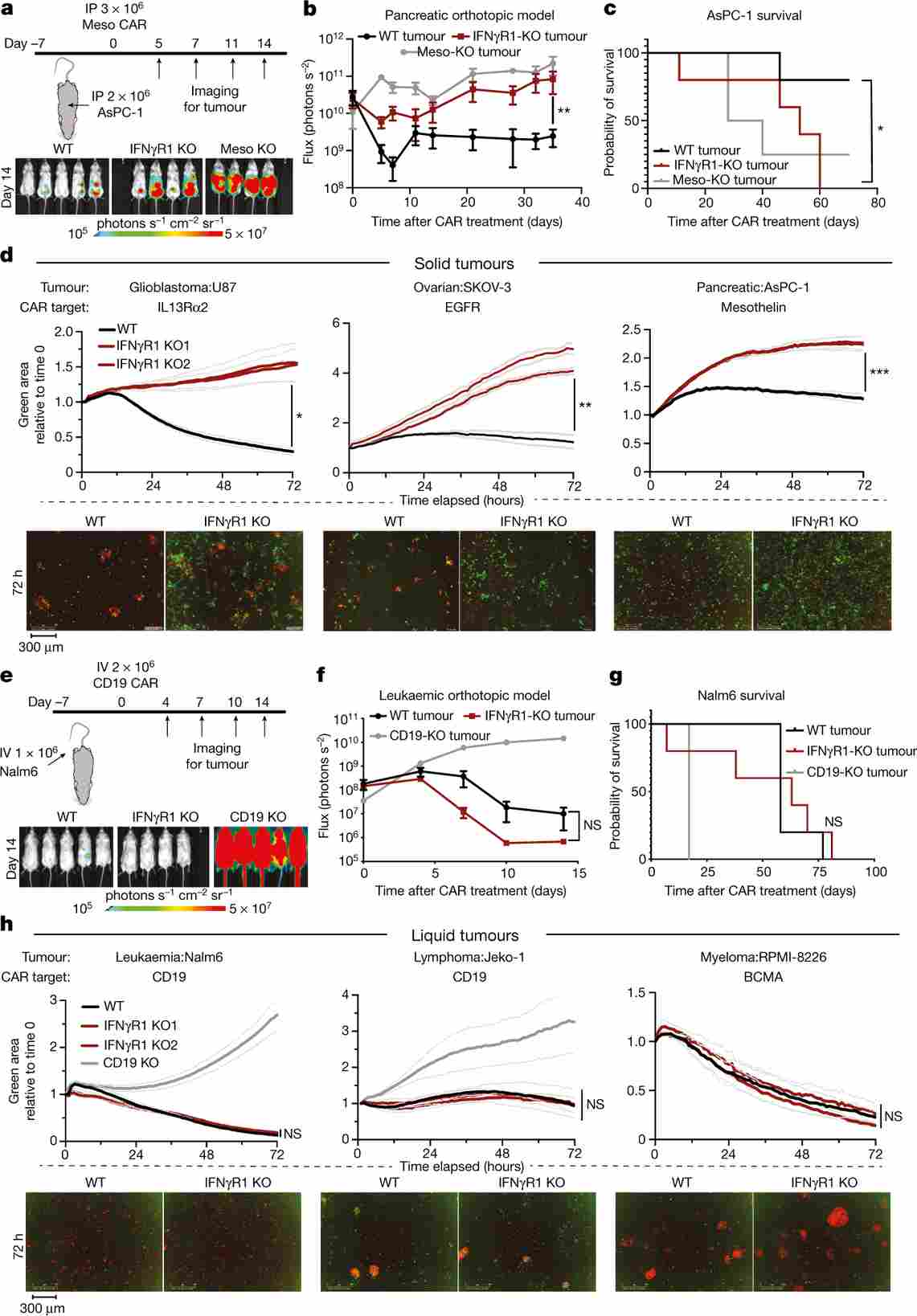
Figure 2. IFNGR1 knockout in U-87 MG confers resistance to multiple CAR T cell products. Wild-type U-87 MG cells are efficiently killed; IFNGR1-KO cells survive across a range of effector-to-target ratios and CAR T constructs.
IFNγ Drives ICAM-1 Upregulation and Immune Synapse Stabilisation
Mechanistic studies showed that CAR T cell–secreted IFNγ signals through IFNGR1 on tumour cells to transcriptionally upregulate ICAM-1. ICAM-1 then engages LFA-1 on CAR T cells to form stable immunological synapses, enabling sustained cytotoxic granule delivery. In IFNGR1-KO U-87 MG cells, IFNγ-driven ICAM-1 induction was abolished, conjugate formation was reduced, and CAR T cell dwell time at the tumour interface was shortened.
Exogenous ICAM-1 overexpression in IFNGR1-KO cells fully rescued CAR T cell sensitivity, directly linking ICAM-1 to synapse stability and cytotoxicity. Conversely, anti-ICAM-1 or anti-LFA-1 blocking antibodies suppressed killing of wild-type U-87 MG cells, confirming the centrality of this adhesion axis.
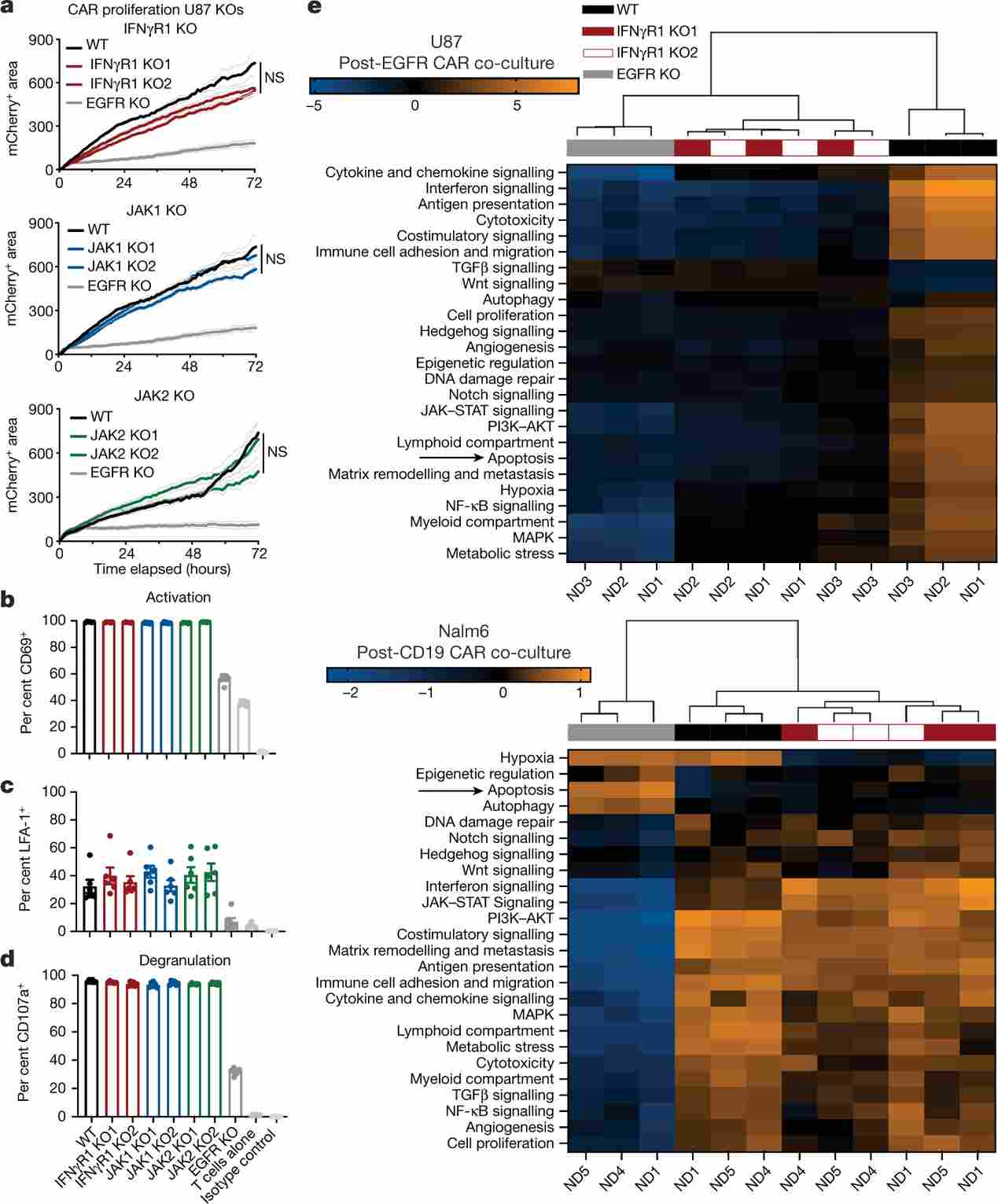
Figure 3. Mechanistic model: IFNγ → IFNGR1 → JAK/STAT → ICAM-1 upregulation → LFA-1 engagement → immune synapse stabilisation in U-87 MG cells. Live imaging shows reduced CAR T cell–tumour conjugation in IFNGR1-KO U-87 MG cells versus controls.
In Vivo Validation: U-87 MG Luc Intracranial BLI Model
For in vivo proof of concept, U-87 MG Luc cells (IFNGR1-competent or IFNGR1-KO) were stereotactically implanted into the striatum of immunodeficient mice. Tumour burden was tracked by weekly bioluminescence imaging (BLI). Mice bearing IFNGR1-expressing U-87 MG Luc tumours showed significant BLI signal reduction and prolonged survival following EGFRvIII CAR T cell therapy. By contrast, IFNGR1-KO U-87 MG Luc tumours progressed despite CAR T cell infusion, with photon flux indistinguishable from vehicle-treated controls—directly translating the in vitro mechanism to an orthotopic brain tumour setting.
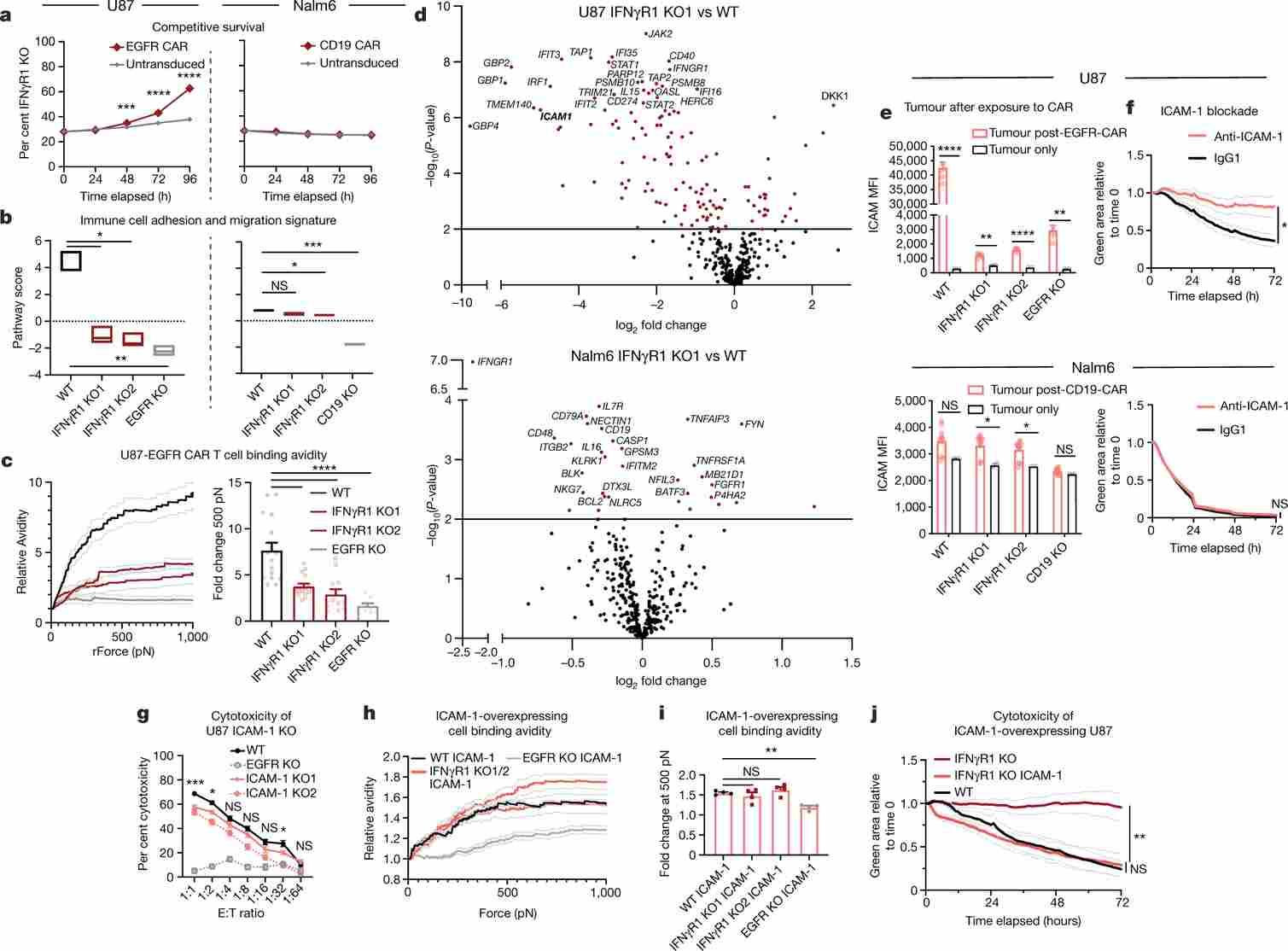
Figure 4. In vivo BLI tracking of U-87 MG Luc intracranial tumours. Mice receiving EGFRvIII CAR T cells against IFNGR1-competent tumours show significant tumour regression and extended survival; IFNGR1-KO tumour-bearing mice are non-responsive.
Discussion and Translational Implications
These findings reframe the role of IFNγ signalling in cancer immunotherapy. Rather than modulating antigen presentation—traditionally its principal immunological function—IFNγR here controls the physical adhesion required for productive CAR T cell cytotoxicity. This distinction is clinically important: tumours that downregulate IFNGR1 or acquire JAK1/2 mutations (known resistance mechanisms in anti-PD-1 therapy) are likely pre-adapted to evade CAR T cells.
From a therapeutic standpoint, the IFNγ–ICAM-1 axis provides actionable targets. Strategies to induce ICAM-1 on tumour cells—via cytokine pre-conditioning, epigenetic modulators, or engineered CAR constructs secreting IFNγ locally—could restore CAR T cell efficacy in IFNγR-deficient GBM. Conversely, selective blockade of IFNγR in healthy tissues may mitigate the cytokine release syndrome seen in liquid tumour CAR T therapies, where IFNγ drives toxicity without contributing to tumour cell adhesion.
Ready to explore CAR T cell resistance mechanisms or next-generation immunotherapy combinations in GBM? Vitrobiotech's U-87 MG Luc cell line—with validated, passage-stable luciferase expression—is optimised for orthotopic intracranial BLI assays in NCG/NSG mice. Browse our catalog today and find the perfect GBM for your study.
Order now: U 87 MG Cell Line / U-87 MG Luciferase Cell Line
Conclusion
Employing U-87 MG Luc in genome-wide CRISPR screens and orthotopic intracranial BLI models, Larson et al. uncovered a fundamentally new requirement for IFNγR-dependent ICAM-1 upregulation in CAR T cell immunological synapse formation and GBM killing. The work establishes a mechanistic roadmap for IFNγR-targeted sensitisation strategies and provides a strong rationale for incorporating BLI-compatible U-87 MG Luc models in the preclinical evaluation of next-generation solid tumour CAR T therapies.
References
Larson RC, Kann MC, Bailey SR, et al. CAR T cell killing requires the IFNγR pathway in solid but not liquid tumours. Nature. 2022;604(7906):563-570. doi:10.1038/s41586-022-04585-5

