Jurkat-Luc: Enabling Peptide-Based PD-1/PD-L1 Checkpoint Blockade Discovery
Introduction
The PD-1/PD-L1 immune checkpoint axis is a validated therapeutic target in oncology. Monoclonal antibodies blocking this interaction — nivolumab, pembrolizumab, atezolizumab — have transformed cancer treatment. However, antibody therapies carry limitations: high manufacturing costs, potential immune-related adverse events, limited tissue penetration, and incomplete responses in certain tumor types.
Peptide-based inhibitors offer a compelling alternative: smaller molecular weight enables tumor penetration, lower production costs, and reduced immunogenicity. A central challenge in developing peptide blockers is the need for a robust, quantitative, and mechanism-relevant screening assay. The Jurkat-Luc (PD-1/NFAT) reporter cell system fulfills this requirement by directly measuring the functional consequence of PD-1/PD-L1 blockade — T cell reactivation — through a bioluminescent readout.
Vitrobiotech supplies validated Jurkat-Luc reporter cell lines engineered for PD-1/PD-L1 checkpoint inhibitor screening. Contact us today to discuss your research needs and receive expert technical support.
Order now: Jurkat Cell Line / Jurkat Luciferase Cell Line
The Jurkat-Luc Reporter Platform for PD-1/PD-L1 Functional Screening
The screening strategy employed in this study relied on the JT-PD1 cell line — a Jurkat-NFAT-Luc reporter engineered to stably express human PD-1. When co-cultured with CHO-PDL1 cells presenting both a TCR activator and PD-L1 on their surface, two signals converge on the Jurkat cell: TCR engagement provides an activating signal, while PD-1/PD-L1 binding delivers an inhibitory signal that suppresses NFAT-driven luciferase transcription. The net bioluminescent output therefore reflects the balance between activation and checkpoint-mediated suppression.
When a PD-1/PD-L1 blocking agent is added, the inhibitory signal is removed, NFAT is reactivated, and luciferase expression increases proportionally. This mechanism-of-action (MOA)-based readout directly quantifies the functional potency of checkpoint inhibitors — whether they are antibodies, peptides, or small molecules — in a single, homogeneous assay format.

Figure 1. Functional screening workflow using JT-PD1 (Jurkat-Luc) reporter cells co-cultured with CHO-PDL1 cells. PD-1/PD-L1-blocking peptides relieve NFAT suppression, producing measurable increases in luciferase signal. Peptide arrays spanning the PD-1 and PD-L1 ectodomains are screened to identify functional blockers.
From Peptide Array to Functional Hit: Identification of L7N
The researchers synthesized overlapping peptide libraries (16–18 amino acids, 3–5 residue overlap) covering the extracellular domains of both PD-1 and PD-L1. Each peptide was tested in the Jurkat-Luc co-culture system for its ability to increase NFAT-driven luminescence above baseline — i.e., to functionally block the PD-1/PD-L1 interaction and restore T cell activation.
A peptide designated L7, derived from the PD-L1 ectodomain, emerged as the initial hit. Through systematic substitution of non-conserved hydrophobic residues with hydrophilic counterparts, the team generated an optimized analogue — L7N — with improved solubility and potency. Dose-response curves in the JT-PD1/CHO-PDL1 system confirmed that L7N blocked PD-1/PD-L1 interaction in a concentration-dependent manner, with activity exceeding that of a reference anti-PD-1 antibody at comparable molar concentrations.
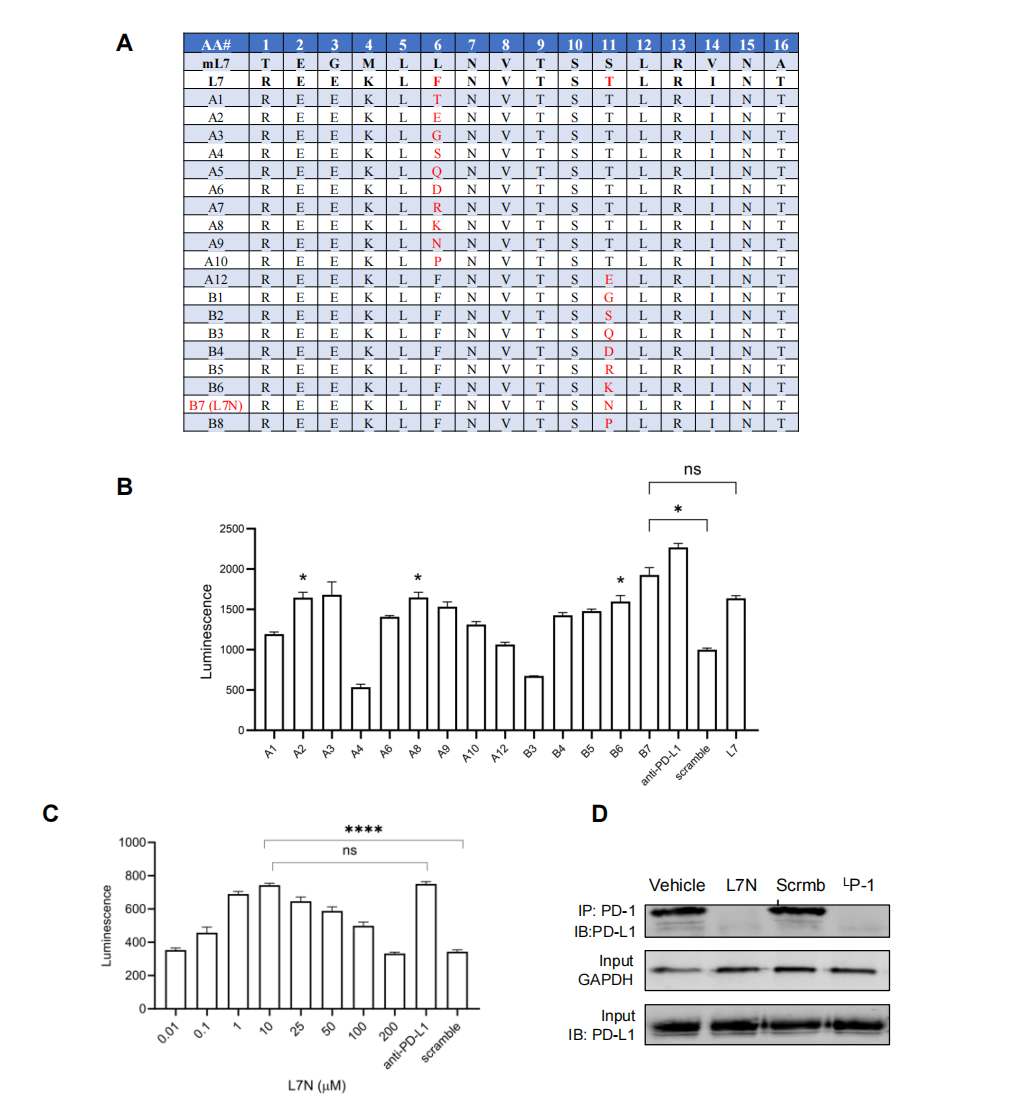
Figure 2. Optimization of the L7 peptide hit. (A) Design of L7 analogues via hydrophobic-to-hydrophilic residue substitution. (B) Comparative NFAT-luciferase activation by L7 analogues in the JT-PD1/CHO-PDL1 co-culture. (C) Dose-response curve for L7N demonstrating concentration-dependent PD-1/PD-L1 blockade.
Our Jurkat-Luc cell lines are validated for PD-1/PD-L1 checkpoint inhibitor screening, delivering reproducible dose-response data. Request a quote now to get started.
Order now: Jurkat Cell Line / Jurkat Luciferase Cell Line
Stabilizing the Peptide: The PA-L7N Albumin-Binding Strategy
A practical limitation of peptide therapeutics is their short plasma half-life due to rapid renal clearance and enzymatic degradation. To address this, the researchers conjugated the N-terminus of L7N to a palmitic acid (PA) tag, generating PA-L7N. The PA moiety binds albumin in circulation, extending the peptide’s effective half-life by leveraging albumin’s long serum residence time (approximately 19 days in humans).
In vitro pull-down assays confirmed that biotinylated PA-L7N efficiently precipitated albumin, validating the albumin-binding functionality of the palmitic acid conjugate. This modification did not compromise the peptide’s PD-1/PD-L1 blocking activity in the Jurkat-Luc reporter assay, demonstrating that stabilization and function can be achieved simultaneously.
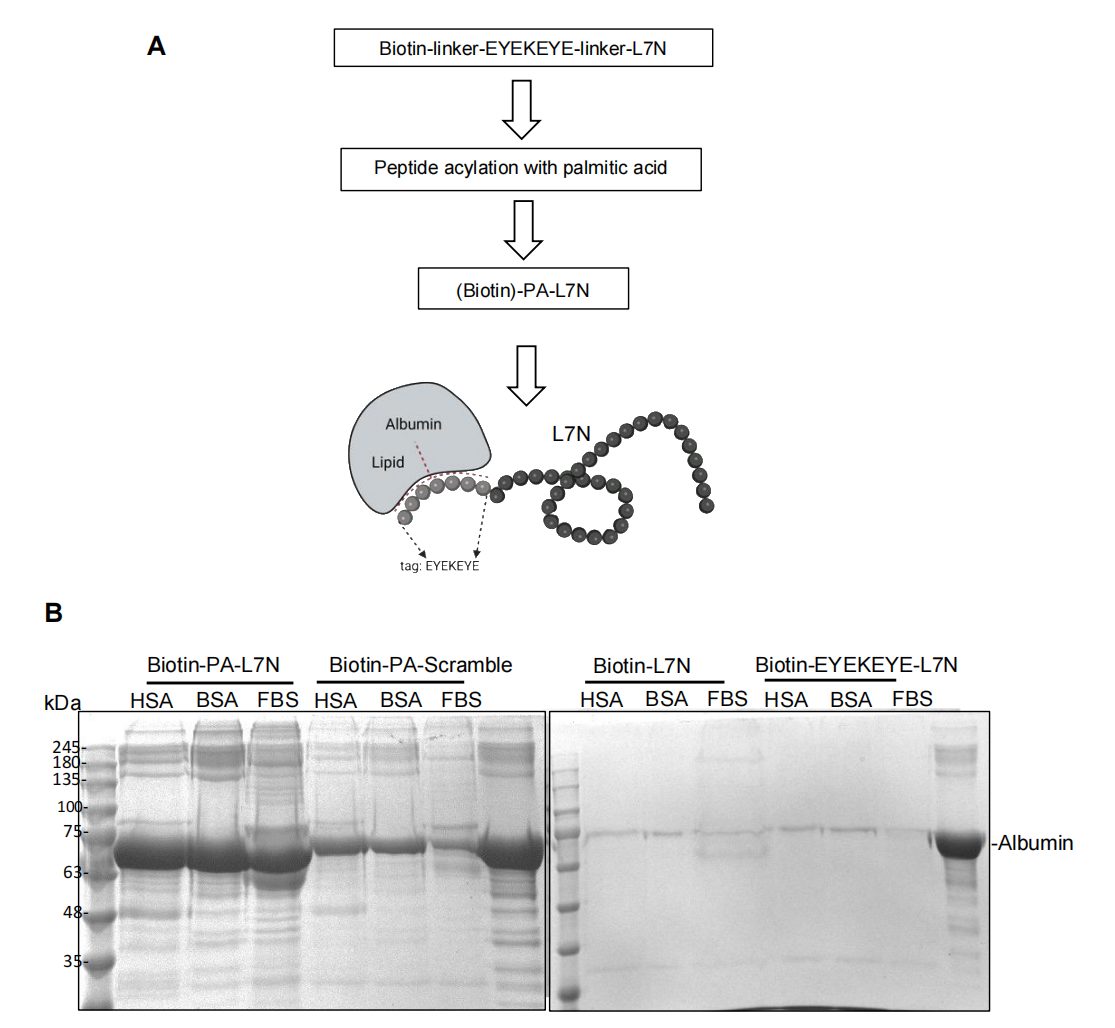
Figure 3. Stabilization of L7N via albumin-binding conjugation. (A) Schematic of PA-L7N synthesis: palmitic acid tag conjugated to the peptide N-terminus. (B) Biotinylated PA-L7N efficiently precipitates albumin in vitro, confirming albumin-binding capacity.
Validating mL7N Across Cellular Systems and Functional Readouts
To confirm that the peptide’s activity was not an artifact of a single assay format, the researchers evaluated L7N and its analogues across three independent cellular systems:
(1) JT-PD1/CHO-PDL1 co-culture — NFAT-luciferase readout confirmed that PA-mL7N (the mouse-optimized analogue) effectively reactivated PD-1-suppressed T cells; (2) JT-PD1/Raji-PDL1 co-culture — IL-2 secretion measured by ELISA demonstrated restored cytokine production; and (3) Human PBMCs co-cultured with MDA-MB-231 breast cancer cells — PA-mL7N significantly enhanced PBMC-mediated cancer cell killing. Concordant results across these mechanistically distinct readouts validate the peptide’s functional potency and the reliability of the Jurkat-Luc reporter as the primary screening tool.
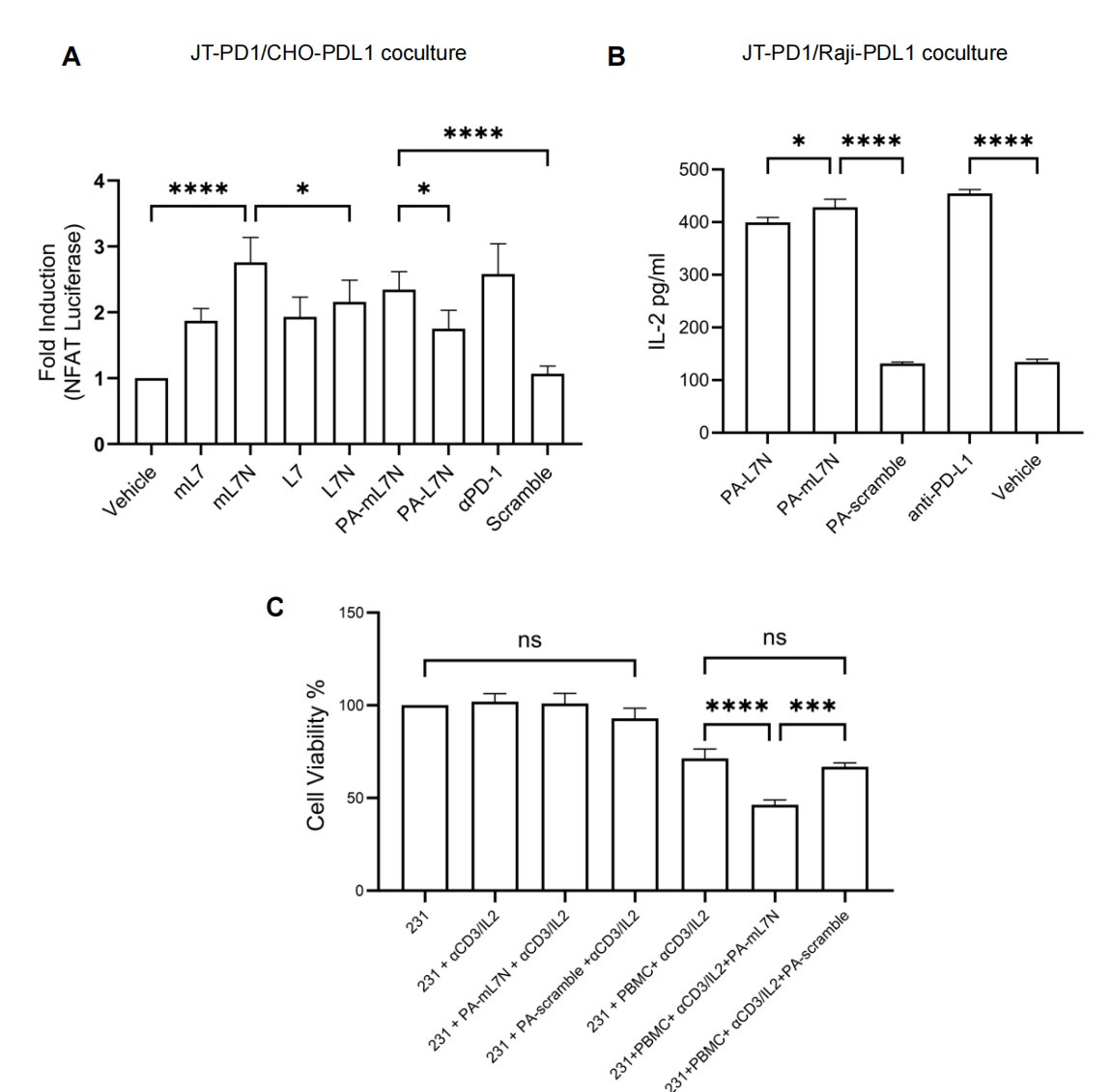
Figure 4. Multi-system validation of mL7N. (A) PA-mL7N activates PD-1-suppressed T cells in the JT-PD1/CHO-PDL1 co-culture (NFAT-luciferase). (B) PA-L7N and PA-mL7N restore IL-2 secretion in the JT-PD1/Raji-PDL1 co-culture. (C) PA-mL7N promotes PBMC-mediated killing of MDA-MB-231 breast cancer cells.
Computational Modeling of Peptide–Target Interactions
Docking simulations placed the peptide blockers at the PD-1/PD-L1 interface, with peptide #26 occupying the cleft between the IgV domains of PD-1 and PD-L1. The L7N and mL7N peptides were predicted to bind both PD-1 and PD-L1 ectodomains, potentially disrupting the protein–protein interaction through direct steric blockade. An alternative mechanism — interference with PD-L1 N-glycosylation — was also proposed, though it requires experimental validation.
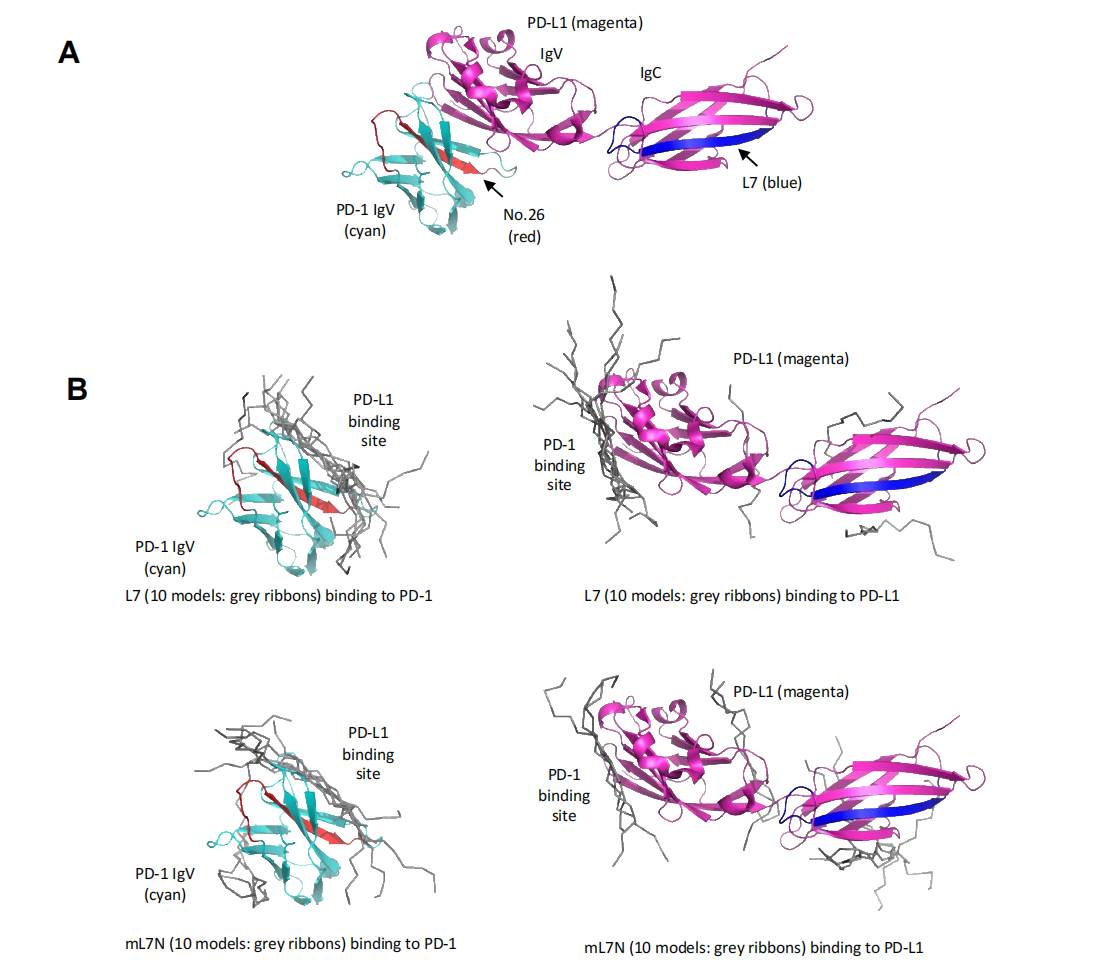
Figure 5. Computational docking models of peptide blockers. (A) Peptide #26 (red) positioned at the PD-1/PD-L1 IgV domain interface. (B) Docking of L7N/mL7N peptides to PD-1 and PD-L1 ectodomains, suggesting direct blockade of the interaction surface.
In Vivo Validation: PA-mL7N Inhibits Tumor Growth in Mice
The ultimate test of therapeutic potential is in vivo efficacy. In the 4T1 syngeneic mouse breast cancer model, daily intraperitoneal injection of PA-mL7N (starting day 7 post-tumor inoculation) produced a dose-dependent reduction in tumor volume and weight. Western blot analysis of excised tumors confirmed that PA-mL7N disrupted PD-1/PD-L1 binding in tumor tissue in a dose-dependent manner. Flow cytometry revealed a significant increase in tumor-infiltrating CD8+ T cells, consistent with checkpoint blockade-mediated immune reactivation.
These results demonstrate that a peptide identified through Jurkat-Luc-based functional screening can translate into measurable anti-tumor activity in vivo, validating the reporter system as a predictive screening platform for checkpoint inhibitor discovery.
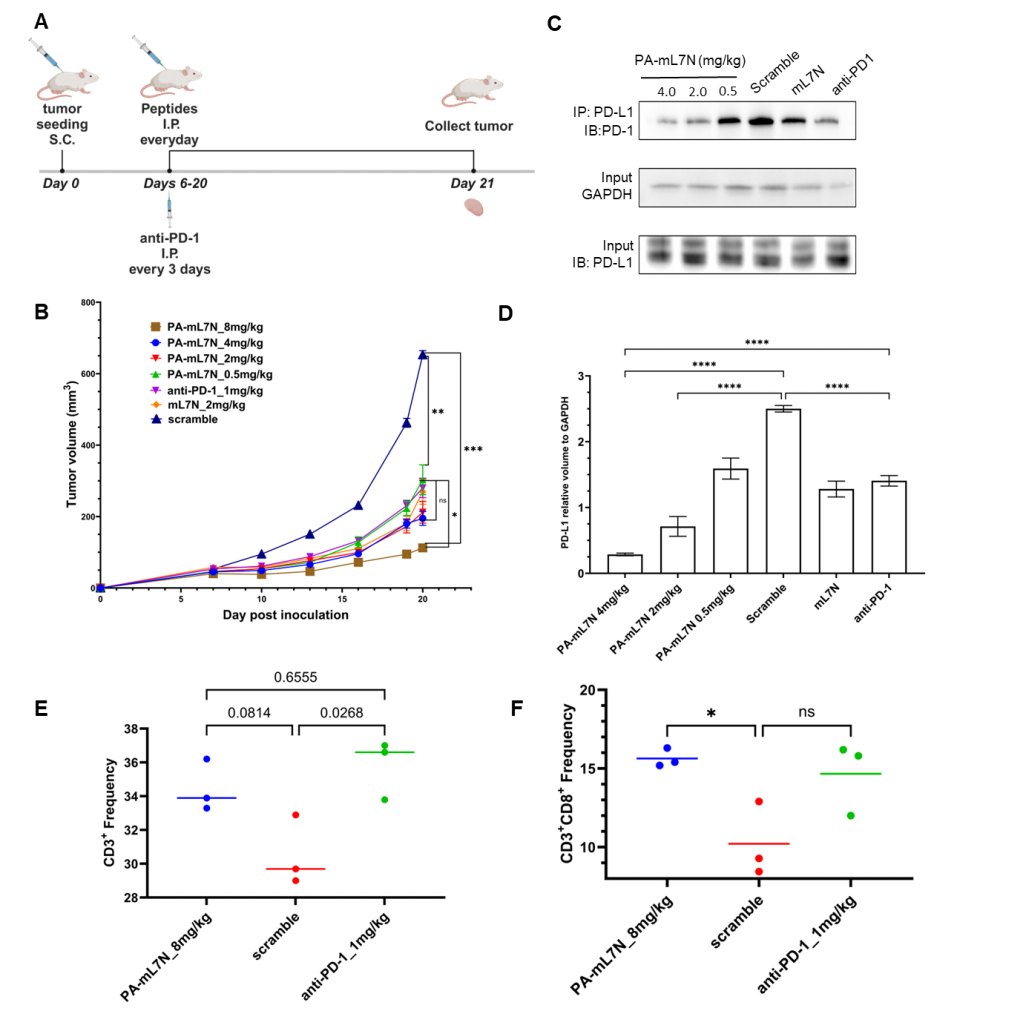
Figure 6. In vivo anti-tumor efficacy of PA-mL7N in the 4T1 syngeneic breast cancer model. (A) Experimental design. (B) Tumor growth curves across treatment groups. (C–D) Western blot showing dose-dependent reduction of PD-1/PD-L1 binding in tumor tissue. (E–F) Flow cytometry data showing increased tumor-infiltrating total T cells and CD8+ T cells following PA-mL7N treatment.
Explore our catalog of validated Jurkat-Luc reporter cell lines and find the perfect screening model for your immunotherapy program. Browse our catalog and find the perfect cell model for your study.
Order now: Jurkat Cell Line / Jurkat Luciferase Cell Line
Discussion: Jurkat-Luc as a Predictive Engine for Checkpoint Inhibitor Discovery
This study illustrates a complete discovery pipeline: from peptide array functional screening in Jurkat-Luc reporter cells, through hit optimization and stabilization, to multi-system validation and in vivo efficacy. The JT-PD1 co-culture system provided the critical quantitative bridge between structural predictions (which peptides could bind PD-1/PD-L1) and functional outcomes (which peptides actually restored T cell activation). Without this mechanism-relevant, high-throughput-capable assay, peptide hit identification would have relied solely on binding assays that cannot predict functional blockade.
The modularity of the Jurkat-Luc platform extends beyond PD-1/PD-L1. By swapping the surface receptor expression profile — PD-1 for CTLA-4, TIGIT, LAG-3, or other checkpoints — the same NFAT-luciferase backbone can be deployed for any immune checkpoint target. This modularity, combined with the assay’s demonstrated predictive value (in vitro luciferase activity correlated with in vivo tumor suppression), positions Jurkat-Luc as a universal screening chassis for next-generation checkpoint inhibitor discovery.
Conclusion
The discovery of mL7N as a functional peptide blocker of PD-1/PD-L1 underscores the value of mechanism-based reporter cell screening. The Jurkat-Luc (PD-1/NFAT) system enabled rapid, quantitative, and functionally relevant evaluation of peptide candidates, directly linking NFAT-driven bioluminescence to T cell reactivation. The translation of in vitro luciferase signals into in vivo anti-tumor efficacy validates this reporter platform as a predictive screening tool for checkpoint inhibitor development, extending its utility beyond antibody-based formats to include peptides, small molecules, and other therapeutic modalities.
References
Zhong SJ, Liu X, Kaneko T, et al. Peptide Blockers of PD-1-PD-L1 Interaction Reinvigorate PD-1-Suppressed T Cells and Curb Tumor Growth in Mice. Cells. 2024;13(14):1193. DOI: 10.3390/cells13141193.

