C2C12-Luc: Enabling ARE-Driven Luciferase Reporter Assays for Nrf2 Activation Screening in Muscle Health Research
Introduction
Chronic oxidative distress drives cellular damage in skeletal muscle, contributing to sarcopenia and cancer cachexia. The transcription factor Nrf2 orchestrates antioxidant and cytoprotective gene expression through the Antioxidant Response Element (ARE), making it a therapeutic target for muscle preservation. C2C12-Luc cells—mouse myoblasts stably transduced with a SIN-lenti-ARE-luciferase construct—enable sensitive, real-time monitoring of Nrf2 transcriptional activity in both proliferating and differentiated states, bridging the gap between molecular signaling and functional muscle outcomes.
Explore our C2C12-Luc cell line for your Nrf2 signaling and muscle health research—contact us to discuss your project needs!
Order now: C2C12 Cell Line / C2C12 Luciferase Cell Line
Cell Viability Screening in Proliferating C2C12 Cells
The study first established the safety profile of four candidate Nrf2 activators—luteolin, creatine, silibinin, and L-BAIBA—in proliferating C2C12-Luc myoblasts using CellTiter-Blue viability assays after 24 h exposure. Luteolin showed no cytotoxicity up to 12.5 uM but reduced viability to 57% at 50 uM. Creatine was well-tolerated up to 12.5 mM. Silibinin was limited by DMSO vehicle toxicity at higher concentrations, while L-BAIBA showed modest viability reduction starting at 25 uM. These dose-response profiles defined the non-toxic concentration ranges for subsequent reporter gene assays.
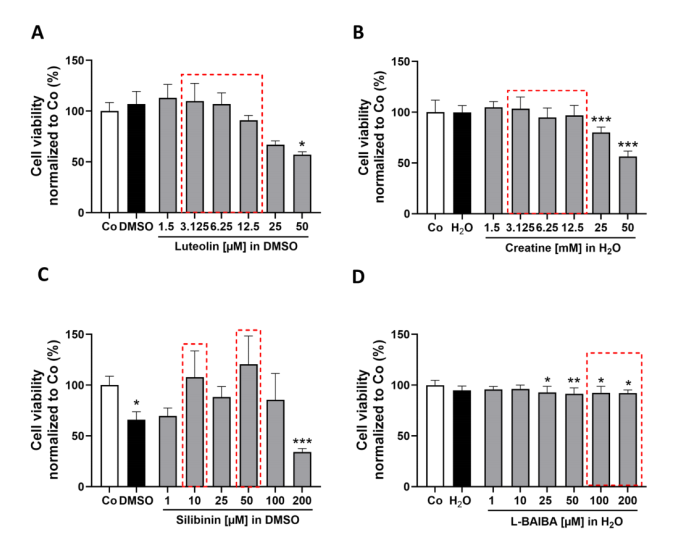
Figure 1. Analysis of cell viability in proliferating cells following 24 h exposure to Nrf2 activators (luteolin, creatine, silibinin, L-BAIBA).
ARE-Luciferase Reporter Activity in Proliferating Myoblasts
Using the ARE-driven firefly luciferase reporter integrated into C2C12-Luc cells, the study quantified Nrf2 transcriptional activation after 8 and 24 h of stimulation. Luteolin emerged as the most potent activator: at 24 h, 6.25 uM induced a 2.83-fold increase in ARE activity, and 12.5 uM reached 5.79-fold elevation—far surpassing all other compounds. Silibinin showed modest activation (1.4-1.45-fold at 10 and 50 uM), while creatine and L-BAIBA produced negligible effects. The temporal analysis revealed that luteolin's action requires sustained exposure, as 8 h stimulation yielded weaker responses than the 24 h timepoint, indicating a transcriptional amplification mechanism.
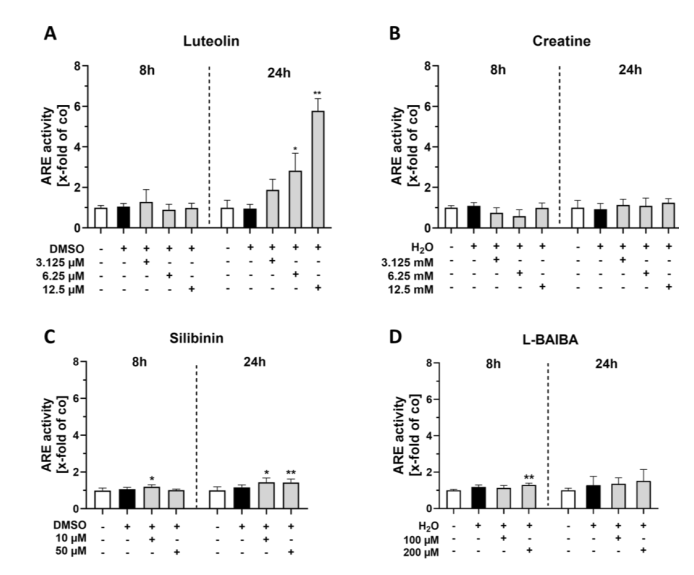
Figure 2. ARE luciferase reporter gene assays of proliferating C2C12 cells after 8 or 24 h of Nrf2 activator stimulation.
Nrf2 Target Gene Expression Confirms Transcriptional Activation
RT-qPCR validation of two canonical Nrf2 target genes—Nqo1 and Hmox1—corroborated the luciferase reporter findings in proliferating C2C12-Luc cells. Luteolin at 3.125 uM increased Nqo1 expression 1.74-fold and induced dose-dependent Hmox1 upregulation (1.43-fold at 6.25 uM; 2.98-fold at 12.5 uM). Silibinin at 50 uM modestly increased Nqo1 (1.38-fold). Creatine and L-BAIBA showed no significant effects on either gene, confirming their inability to activate Nrf2-driven transcription in myoblasts. The concordance between luciferase reporter data and endogenous gene expression validates the C2C12-Luc reporter system as a reliable screening tool.
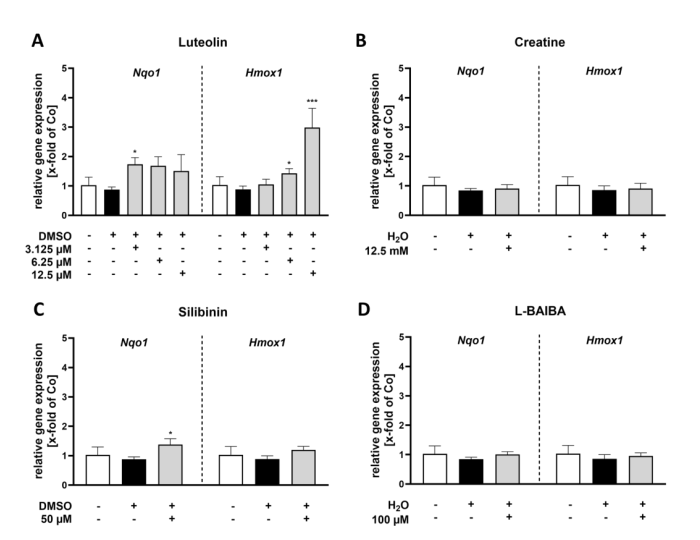
Figure 3. RT-qPCR analysis of Nrf2 activity markers (Nqo1, Hmox1) in proliferating C2C12 cells following 24 h stimulation with Nrf2 activators.
Request a quote for C2C12-Luc cells and accelerate your Nrf2 signaling and antioxidant pathway research today!
Order now: C2C12 Cell Line / C2C12 Luciferase Cell Line
Viability Profiling in Differentiated Myotubes
Differentiated C2C12-Luc myotubes—formed after 4 days of serum withdrawal—represent a more physiologically relevant model for mature muscle tissue. Viability assessment revealed distinct sensitivity profiles compared to proliferating cells: luteolin showed increased sensitivity with significant viability reduction starting at 3.125 uM (78-87% range). Silibinin exhibited no cytotoxicity even at 200 uM, while L-BAIBA at 1 and 100 uM paradoxically increased viability to 112-116%. These differential responses between proliferating and differentiated states underscore the importance of testing compounds in both cellular contexts when evaluating muscle-targeted therapeutics.
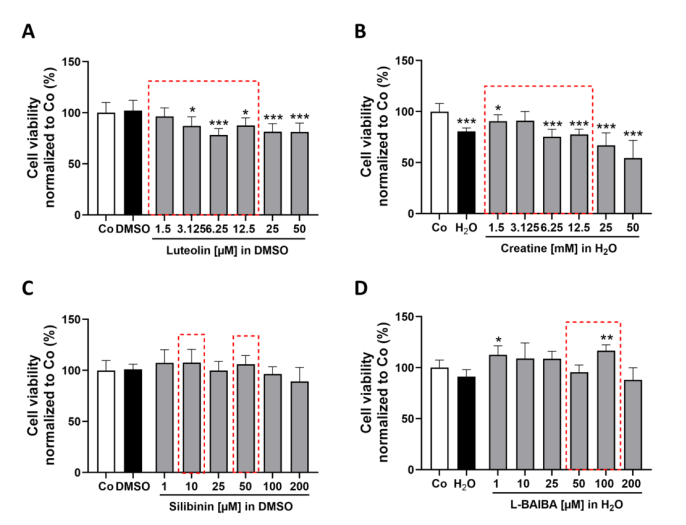
Figure 4. Assessment of cell viability in differentiated C2C12 cells following 24 h stimulation with Nrf2 activators.
Nrf2 Activation in Differentiated Myotubes via ARE-Luciferase
The ARE-luciferase reporter in differentiated C2C12-Luc myotubes confirmed luteolin as the sole effective Nrf2 activator in this mature muscle context. At 24 h, 6.25 uM luteolin induced a 1.98-fold increase in ARE activity, and 12.5 uM reached 3.5-fold—demonstrating robust Nrf2 activation despite the lower absolute magnitude compared to proliferating cells. Creatine, silibinin, and L-BAIBA all failed to stimulate ARE-driven transcription in differentiated cells at any tested concentration or timepoint. This finding highlights the cell-state-dependent nature of Nrf2 pathway modulation and validates luteolin as a candidate therapeutic for preserving muscle integrity under oxidative stress in both immature and mature myofiber contexts.
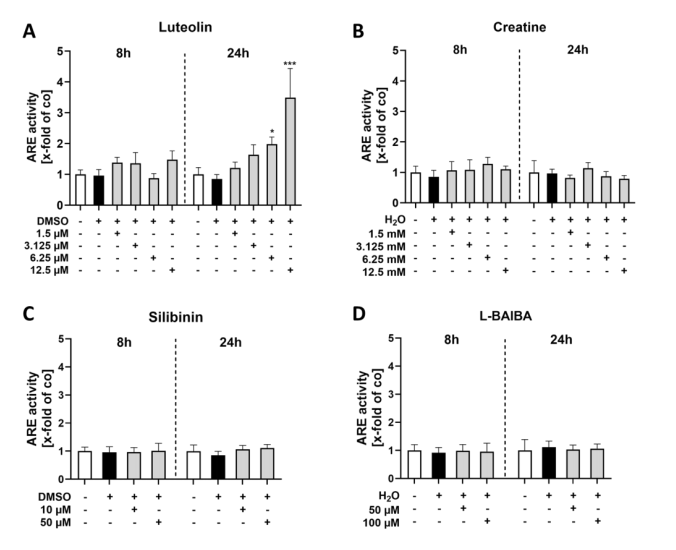
Figure 5. ARE luciferase reporter gene assays of differentiated C2C12 cells after 8 or 24 h of Nrf2 activator stimulation.
Gene Expression Validation in Differentiated Myotubes
In differentiated C2C12-Luc myotubes, RT-qPCR revealed a divergence from the proliferating cell profile: luteolin, creatine, and L-BAIBA did not significantly alter Nqo1 or Hmox1 expression, while silibinin at 50 uM upregulated both genes (Nqo1: 1.62-fold; Hmox1: 1.59-fold). This dissociation between luciferase reporter activity (where luteolin dominated) and endogenous gene expression suggests context-dependent post-transcriptional regulation. The C2C12-Luc ARE-reporter captures real-time transcriptional dynamics that may precede or differ from steady-state mRNA accumulation, underscoring the value of luciferase-based screening as a complementary approach to conventional gene expression analysis.
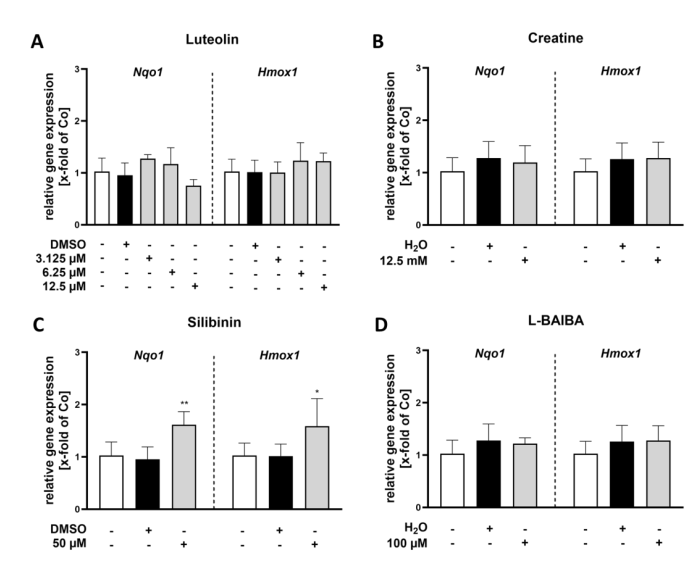
Figure 6. RT-qPCR analysis of Nrf2 activity markers (Nqo1, Hmox1) in differentiated C2C12 cells following 24 h stimulation with Nrf2 activators.
Discussion
This study establishes luteolin as a potent Nrf2 activator in skeletal muscle cells, with C2C12-Luc ARE-reporter cells serving as the central screening platform. The dual-state approach—testing both proliferating myoblasts and differentiated myotubes—reveals cell-state-specific pharmacological profiles that would be missed in single-context assays. The 5.79-fold ARE activation by luteolin in proliferating cells and 3.5-fold in differentiated myotubes demonstrates significant therapeutic potential for conditions characterized by oxidative muscle damage, including sarcopenia and cancer cachexia.
The concordance between luciferase reporter output and endogenous gene expression in proliferating cells validates the C2C12-Luc system as a reliable screening tool. The divergence observed in differentiated cells—where luteolin activated the ARE-luciferase reporter but did not increase steady-state Nqo1/Hmox1 mRNA—may reflect post-transcriptional regulation mechanisms unique to mature myofibers, warranting further investigation into ARE-dependent translational control.
Conclusion
The C2C12-Luc ARE-reporter cell line provides a sensitive, real-time platform for screening Nrf2-activating compounds in both myoblast and myotube contexts. Luteolin stands out as the most effective Nrf2 activator, with dose-dependent ARE-luciferase induction in both cellular states. This work positions C2C12-Luc as an essential tool for muscle health drug discovery, enabling rapid identification of compounds that counteract oxidative damage in skeletal muscle.
Browse our catalog and order C2C12-Luc cells today—engineered for reliable ARE-driven luciferase reporting in muscle research!
Order now: C2C12 Cell Line / C2C12 Luciferase Cell Line
References
Bottcher N, Suhr F, Pufe T, Wruck CJ, Fragoulis A. Luteolin Induces Nrf2 Activity in C2C12 Cells: Implications for Muscle Health. Int J Mol Sci 2025;26(9):4092. DOI: 10.3390/ijms26094092

