DU145-Luc: Enabling In Vivo Ratiometric Bioluminescence Imaging of Metal Ions in Prostate Cancer Models via Semisynthetic
Introduction
Zinc ions (Zn²⁺) play critical roles in numerous biological processes, including enzyme catalysis, gene transcription, immune function, and neurotransmission. Aberrant Zn²⁺ homeostasis is closely associated with prostate cancer initiation, progression, and metastasis. The prostate gland accumulates the highest concentration of zinc of any soft tissue in the human body, and dramatic zinc depletion is a well-established hallmark of malignant prostate transformation. Consequently, the ability to image Zn²⁺ dynamics in vivo—particularly within prostate tumors—represents a powerful diagnostic and research capability.
However, existing fluorescence-based DNA probes suffer from fundamental limitations when applied in vivo: tissue autofluorescence, light scattering, and the need for external laser excitation all compromise signal-to-noise ratios and imaging depth. Bioluminescence imaging (BLI) overcomes these barriers by generating light through enzymatic reactions within the target tissue, eliminating excitation light and enabling deeper tissue penetration with superior sensitivity.
A groundbreaking 2023 study published in Angewandte Chemie International Edition by a collaborative team from Hunan University and the University of Texas at Austin addressed this challenge by developing bioluminescent nucleic acid (bioLUNA) sensors—semisynthetic probes that couple DNAzyme-based metal ion recognition with an engineered Nano-luciferase (NLuc) fusion protein. Critically, the in vivo validation of this platform was achieved using DU145-Luc cells, a human prostate carcinoma line stably expressing firefly luciferase, orthotopically implanted into immunodeficient mice.
Our validated DU145-Luc cell line provides the stable, high-signal bioluminescence platform essential for advanced in vivo biosensing applications. Whether you are developing novel molecular sensors, studying metal ion biology, or evaluating prostate cancer therapeutics, DU145-Luc delivers reliable and reproducible in vivo imaging performance.
Order now: DU 145 Cell Line | DU 145 Luciferase Cell Line
Engineering the bioLUNA Sensor Platform
The core innovation of this work lies in the construction of a semisynthetic bioluminescent sensor that integrates a DNAzyme metal ion recognition element with an engineered bioluminescent protein. The research team designed a genetic fusion between a circularly permuted Nano-luciferase (cpNLuc) and HaloTag, creating a protein scaffold (cpHNLuc) that enables efficient covalent conjugation with DNA oligonucleotides through the HaloTag self-labeling reaction. Computational modeling and systematic mutagenesis were employed to optimize the fusion protein for maximal labeling efficiency and bioluminescence output.
The resulting bioLUNA sensor operates on a bioluminescence resonance energy transfer (BRET) principle. In the absence of the target metal ion, a fluorophore-labeled DNA strand remains hybridized to the DNAzyme on the protein, and BRET occurs between the luciferase donor and the fluorophore acceptor, producing a characteristic dual-wavelength emission signal. Upon Zn²⁺ recognition by the DNAzyme, a substrate cleavage reaction releases the fluorophore strand, abolishing BRET and shifting the emission spectrum. This mechanism enables ratiometric imaging—reporting metal ion concentrations as the ratio of two emission wavelengths rather than absolute intensity, which provides inherent correction for variations in probe concentration, tissue depth, and substrate delivery.
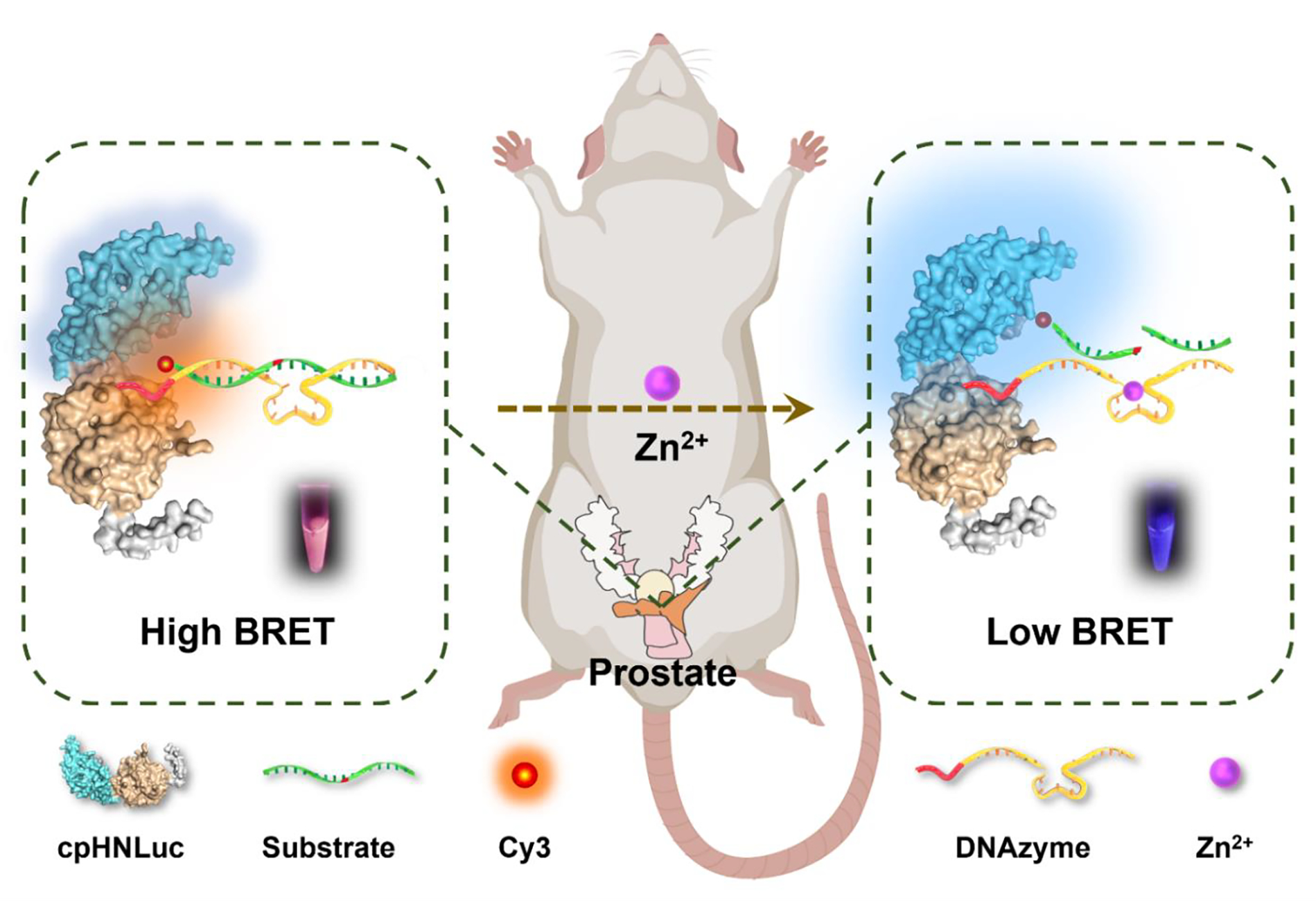
Scheme 1. Schematic illustration of the semisynthetic bioluminescent nucleic acid (bioLUNA) sensor platform. A circularly permuted Nano-luciferase (cpNLuc) is genetically fused to HaloTag, enabling covalent attachment of a fluorophore-labeled DNAzyme sensor strand. In the absence of Zn²⁺, BRET between NLuc and the fluorophore generates a characteristic dual-wavelength signal. Zn²⁺-activated DNAzyme cleavage releases the fluorophore strand, eliminating BRET and enabling ratiometric detection of metal ion concentration.
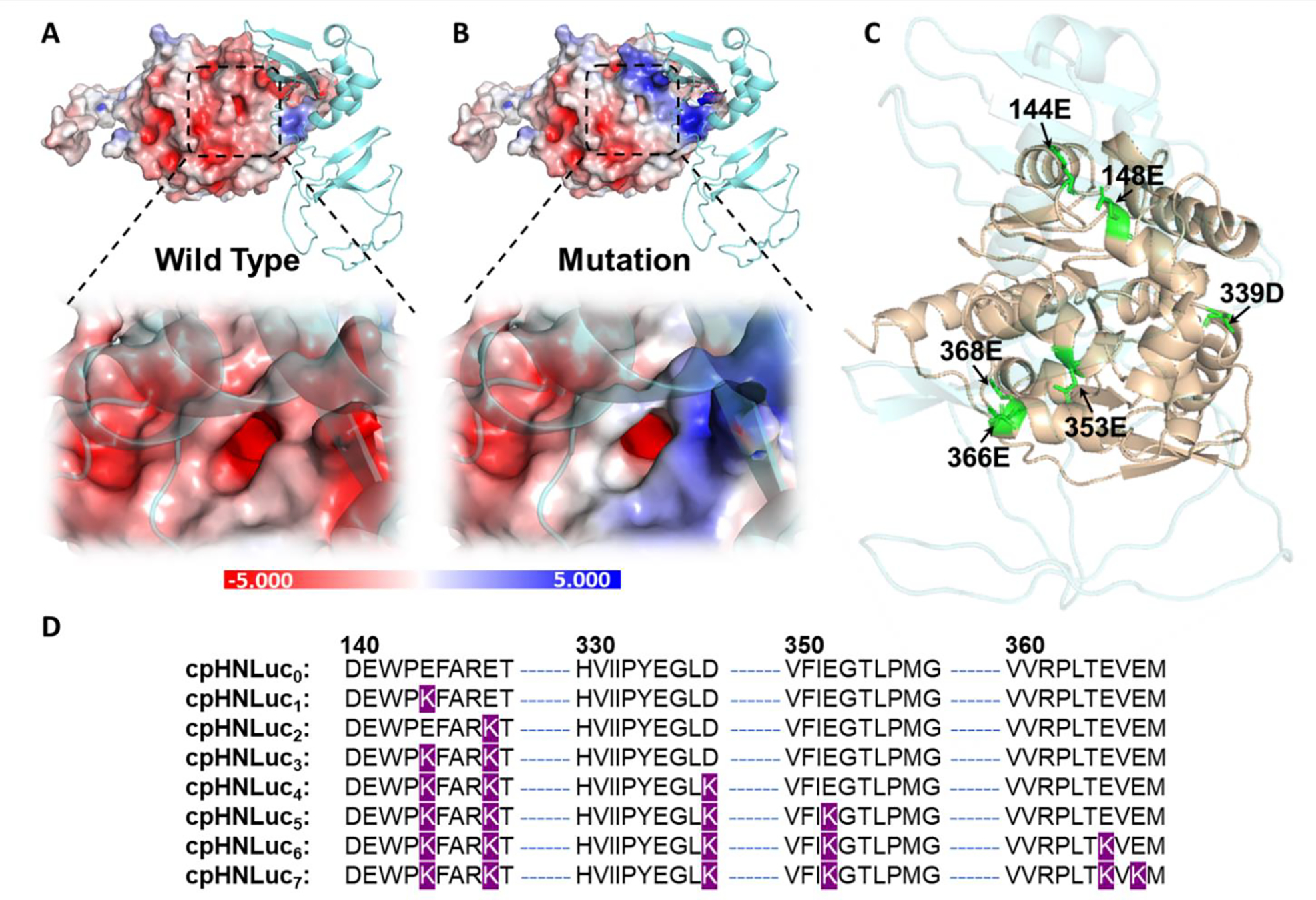
Figure 1. Engineering and characterization of cpHNLuc for efficient DNA sensor conjugation. (a) Structural model of the cpNLuc-HaloTag fusion protein showing the spatial arrangement of functional domains. (b) Labeling kinetics demonstrating rapid and efficient covalent attachment of chloroalkane-modified DNA oligonucleotides via the HaloTag reaction. (c) BRET ratio optimization across different fusion constructs. (d) Emission spectra confirming efficient energy transfer between the Nano-luciferase donor and the fluorophore acceptor in the assembled bioLUNA sensor.
In Vitro and In Vivo Sensing Performance
The Zn²⁺-sensing bioLUNA construct was rigorously validated through a series of in vitro and in vivo experiments. In cell-free solution, the sensor demonstrated a linear dose-dependent response to Zn²⁺ concentrations across a physiologically relevant range, with high selectivity against competing metal ions including Na⁺, K⁺, Ca²⁺, Mg²⁺, Fe²⁺, and Cu²⁺. The detection limit and dynamic range were suitable for monitoring biological Zn²⁺ fluctuations in tissue microenvironments.
For in vivo validation, the team adopted a sophisticated dual-luciferase strategy. DU145-Luc cells—stably expressing firefly luciferase—were orthotopically implanted into the ventral prostate of immunodeficient mice, establishing a clinically relevant prostate cancer model. The DU145-Luc cells served a dual purpose: (1) providing a constitutive bioluminescence signal for tumor localization and monitoring, and (2) creating a prostate tumor microenvironment in which to evaluate exogenous and endogenous Zn²⁺ sensing. The bioLUNA sensor was administered by intratumoral injection, and ratiometric bioluminescence images were acquired using IVIS spectrum imaging systems with appropriate bandpass filters.
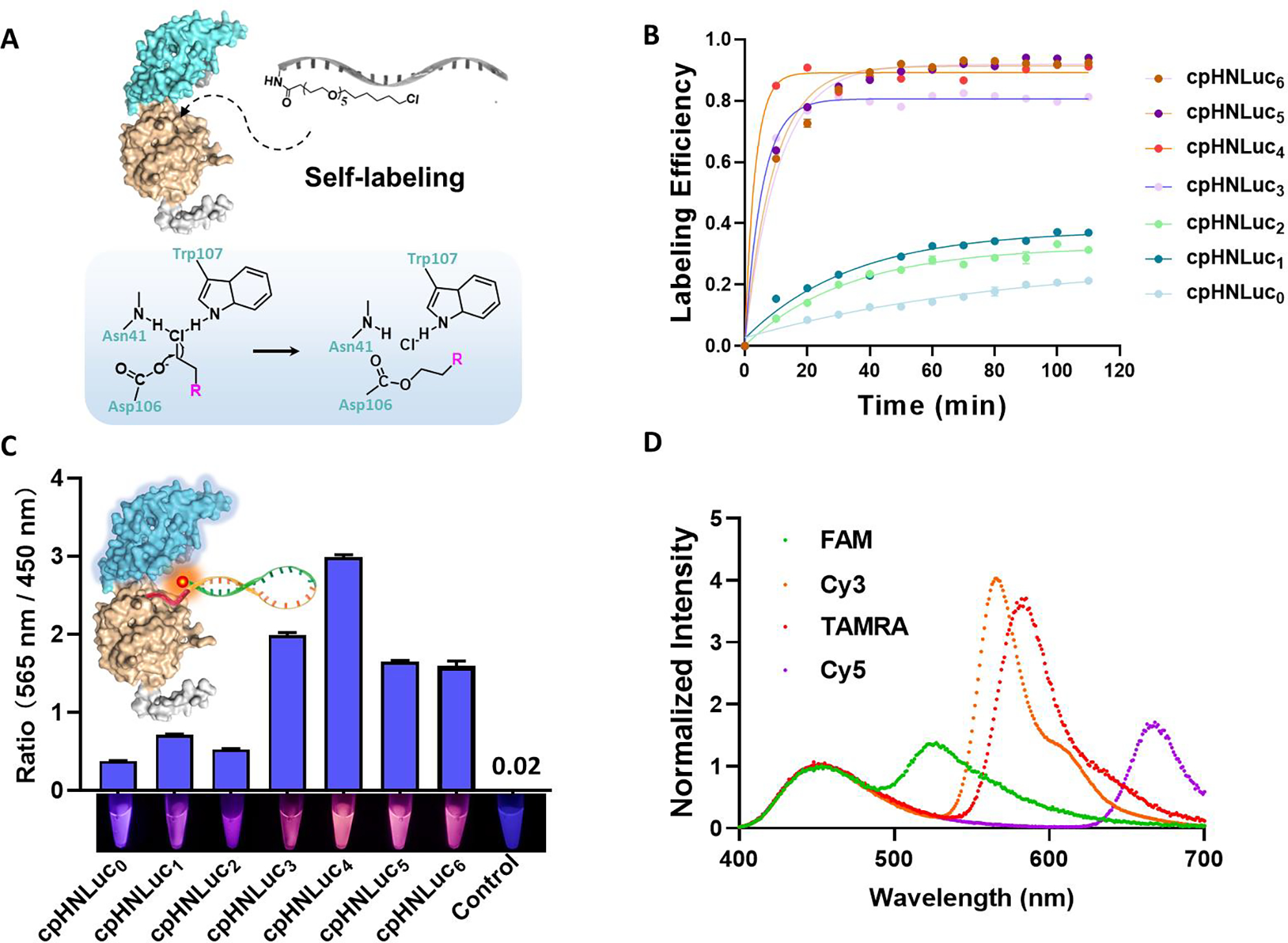
Figure 2. Characterization of the bioLUNA sensor performance. (a) Self-labeling efficiency of cpHNLuc with chloroalkane-modified DNAzyme substrate, confirmed by gel electrophoresis and fluorescence imaging. (b) Labeling kinetics showing rapid conjugation reaching completion within minutes. (c) BRET ratio (acceptor/donor emission intensity) as a function of Zn²⁺ concentration, demonstrating dose-dependent ratiometric response. (d) Emission spectra at varying Zn²⁺ levels, confirming spectral shift upon metal ion-triggered DNAzyme cleavage.
In Vivo Prostate Cancer Zinc Ion Imaging
The pivotal in vivo experiments demonstrated the bioLUNA sensor’s capability to image Zn²⁺ dynamics in live tumor-bearing mice. Whole-body bioluminescence imaging clearly visualized the orthotopic DU145-Luc prostate tumors, confirming successful engraftment and providing an anatomical reference for sensor readout. Upon intratumoral injection of the bioLUNA sensor followed by systemic delivery of the luciferase substrate, ratiometric BLI images revealed distinct Zn²⁺ signal patterns within the tumor region.
The researchers further demonstrated the ability to image endogenous Zn²⁺ efflux events in both normal prostate epithelium and prostate tumors. By comparing ratiometric signals between tumor-bearing and control mice, and between treated and untreated conditions, they established that the bioLUNA platform could reliably report physiological Zn²⁺ dynamics in deep tissue without the artifacts that plague fluorescence-based approaches. The use of DU145-Luc cells as the tumor model was essential—their stable luciferase expression provided a consistent reference signal for image registration and tumor delineation, while the orthotopic implantation ensured that the Zn²⁺ sensing occurred within the physiologically relevant prostate tissue microenvironment.
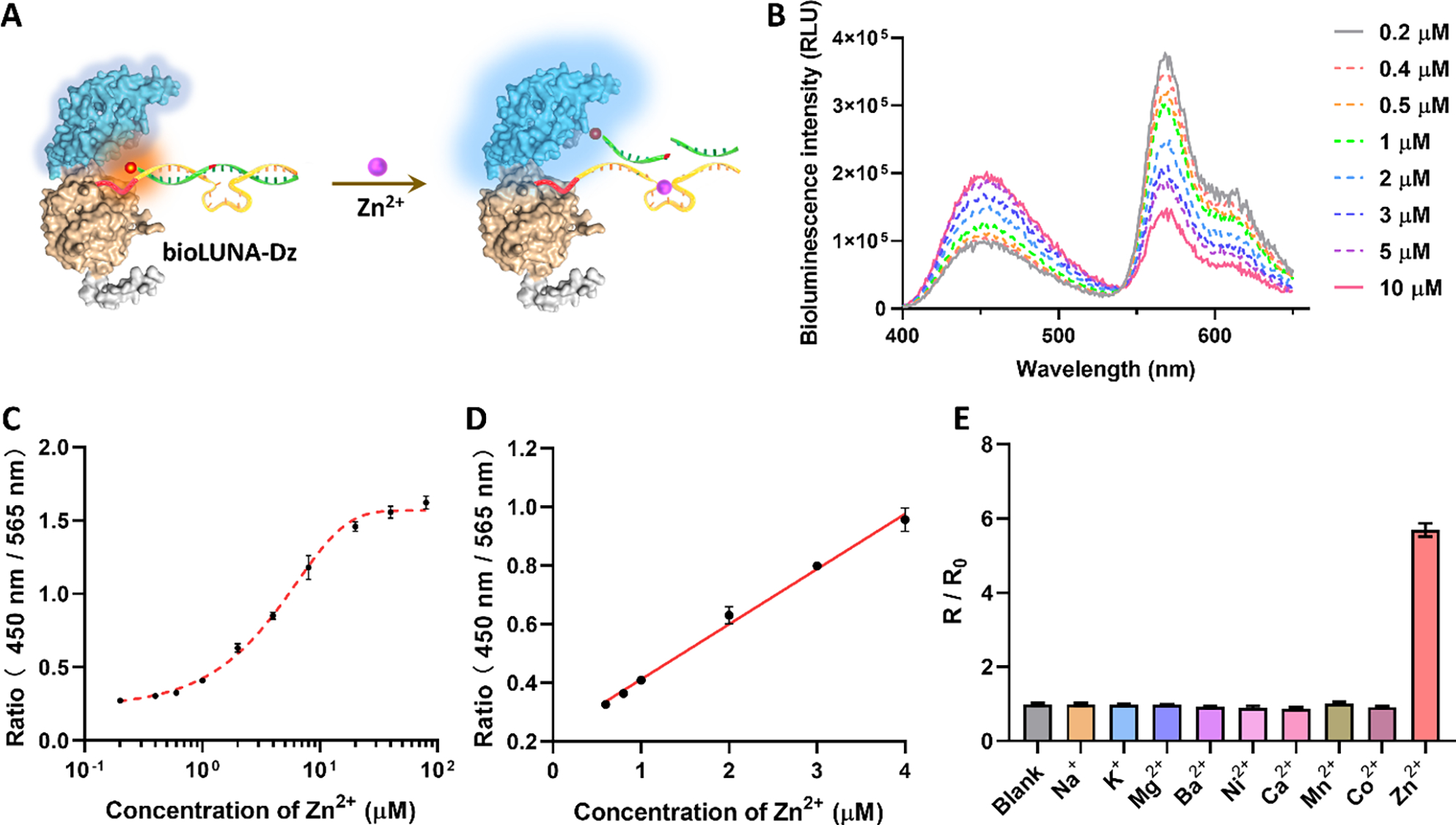
Figure 3. In vitro Zn²⁺ detection performance of the bioLUNA sensor. (a) Schematic of the BRET-based DNAzyme sensor mechanism: Zn²⁺-activated cleavage separates the fluorophore from the luciferase, modulating the BRET signal. (b–e) Dose-response curves, selectivity profiles against competing metal ions, kinetic analysis of the sensing reaction, and pH-dependent performance, confirming the sensor’s robustness under physiological conditions.
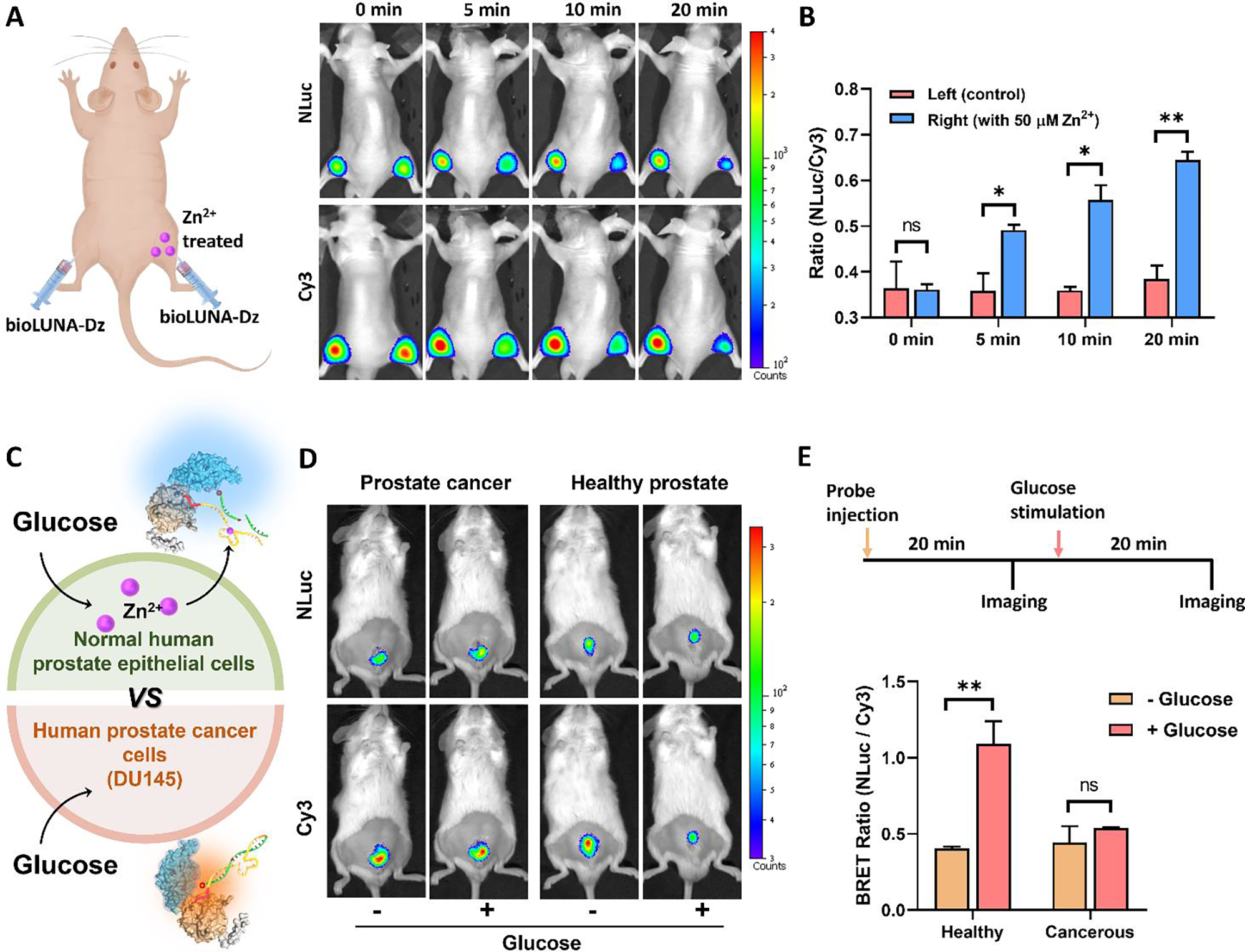
Figure 4. In vivo ratiometric bioluminescence imaging of Zn²⁺ in DU145-Luc prostate tumor-bearing mice. (a) Representative whole-body BLI images showing DU145-Luc tumor localization (firefly luciferase signal) and bioLUNA sensor ratiometric readout within the prostate tumor region. (b) Quantitative analysis of ratiometric BRET signals in tumor versus normal tissue, demonstrating differential Zn²⁺ dynamics. (c) Time-course imaging of endogenous Zn²⁺ efflux events following pharmacological stimulation. (d) Validation of sensing specificity through control experiments with mutated DNAzyme sequences.
The DU145-Luc cell line is more than a tumor model—it is a versatile bioluminescence platform for advanced molecular imaging applications. From drug screening to biosensor development, DU145-Luc delivers the stable, high-sensitivity in vivo signal that your research demands.
Order now: DU 145 Cell Line | DU 145 Luciferase Cell Line
Discussion
This study by Xiong, Lu, and colleagues represents a significant advance in both DNA-based sensing technology and in vivo molecular imaging methodology. By engineering a semisynthetic bioluminescent nucleic acid sensor that operates on a ratiometric BRET principle, the team overcame the longstanding limitations of fluorescence-based probes—autofluorescence, limited tissue penetration, and dependence on external excitation light—that have constrained in vivo metal ion imaging for decades.
The selection of DU145-Luc as the in vivo tumor model was strategically important for several reasons. First, as a human prostate carcinoma line, DU145 faithfully recapitulates key aspects of prostate cancer biology, including androgen-independent growth and metastatic potential. Second, the stable expression of firefly luciferase in DU145-Luc cells provides a constitutive, tissue-penetrating bioluminescence signal that enables precise tumor localization without the need for external contrast agents or ionizing radiation. Third, the orthotopic (intraprostatic) implantation model ensures that the Zn²⁺ sensing occurs within the native prostate tissue architecture, where zinc biology is most physiologically relevant.
For the preclinical research community, this work opens new avenues for studying metal ion biology in live animals. The bioLUNA platform’s modular design means that different DNAzyme sequences can be substituted to detect a wide variety of metal ions and small molecule targets, all read out through the same bioluminescence imaging infrastructure. The DU145-Luc cell line serves as an ideal companion model for these applications, providing both a clinically relevant tumor system and a reliable bioluminescent reference signal for image co-registration and longitudinal monitoring.
Conclusion
The DU145-Luc cell line is an essential tool for advanced in vivo bioluminescence applications, from traditional tumor growth monitoring to next-generation molecular biosensing. The Xiong et al. study demonstrates how DU145-Luc’s stable luciferase expression, clinically relevant prostate cancer biology, and proven in vivo performance make it uniquely suited for validating novel imaging technologies such as the bioLUNA semisynthetic sensor platform. Whether used for drug screening, tumor microenvironment analysis, metal ion dynamics studies, or multi-modal imaging applications, DU145-Luc provides the reliable, high-sensitivity bioluminescence signal that cutting-edge preclinical research demands.
Elevate your prostate cancer research with our validated DU145-Luc cell line. Stable luciferase expression, proven orthotopic tumor engraftment, and seamless compatibility with advanced biosensing platforms make DU145-Luc the premier choice for in vivo bioluminescence studies.
Order now: DU 145 Cell Line | DU 145 Luciferase Cell Line
References
Xiong M, Wu Y, Kong G, Lewis W, Yang Z, Zhang H, Xu L, Liu Y, Liu Q, Zhao X, Zhang X-B, Lu Y. A Semisynthetic Bioluminescence Sensor for Ratiometric Imaging of Metal Ions In Vivo Using DNAzymes Conjugated to An Engineered Nano-Luciferase. Angew Chem Int Ed. 2023;62(37):e202308086. doi:10.1002/anie.202308086

