Targeted Mucosal Immune Activation via Engineered Nanoparticles Enables Complete Eradication of MC38-LUC Lung Metastases
1. Research Background
Clearance of lung metastases is a key clinical challenge in cancer treatment. In recent years, with the development of pulmonary immunotherapy, inhaled delivery of the STING agonist cGAMP to activate mucosal immunity has become a promising anti-tumor strategy. However, existing delivery systems face three technical bottlenecks: structural instability during nebulization causes drug leakage, mucus barrier blocks formulation penetration to target cells, and insufficient cytosolic delivery efficiency limits immune activation effects. Conventional strategies fail to balance these issues—rigid lipids or high PEGylation enhance stability but impair cytosolic delivery capacity, while traditional single-layer liposomes enable effective drug loading but cannot resist shear force damage during nebulization. These limitations severely hinder the clinical application of inhaled STING agonists.
Track tumor dynamics in real time and obtain accurate experimental data for your immunotherapy studies. Order MC38-LUC Cell>>
2. Research Design
To address these multiple challenges, the research team proposed a dual-regulation strategy integrating structural reconstruction and chemical optimization. For structural design, the team introduced negatively charged hyaluronic acid (HA) to reconstruct traditional single-layer DOTAP cationic liposomes into HLHC, a nanosystem with multi-layered concentric structures. This design retained the excellent cytosolic delivery capacity of each lipid layer while significantly improving the mechanical stability of the formulation, enabling it to resist shear force damage during nebulization. The stable multi-layered structure, combined with the surface HA coating, further endowed the formulation with efficient mucus penetration capacity, promoting rapid recognition by APCs and increased cytosolic cGAMP delivery. For chemical optimization, to improve the final therapeutic efficacy of HLHC, the team systematically screened lipid chemical structures to enhance endosomal escape and cytosolic delivery efficiency—the final step of intracellular delivery. By conjugating endogenous basic amino acids (e.g., arginine) with lipid molecules, they developed Arg-HLHC with significantly improved performance.
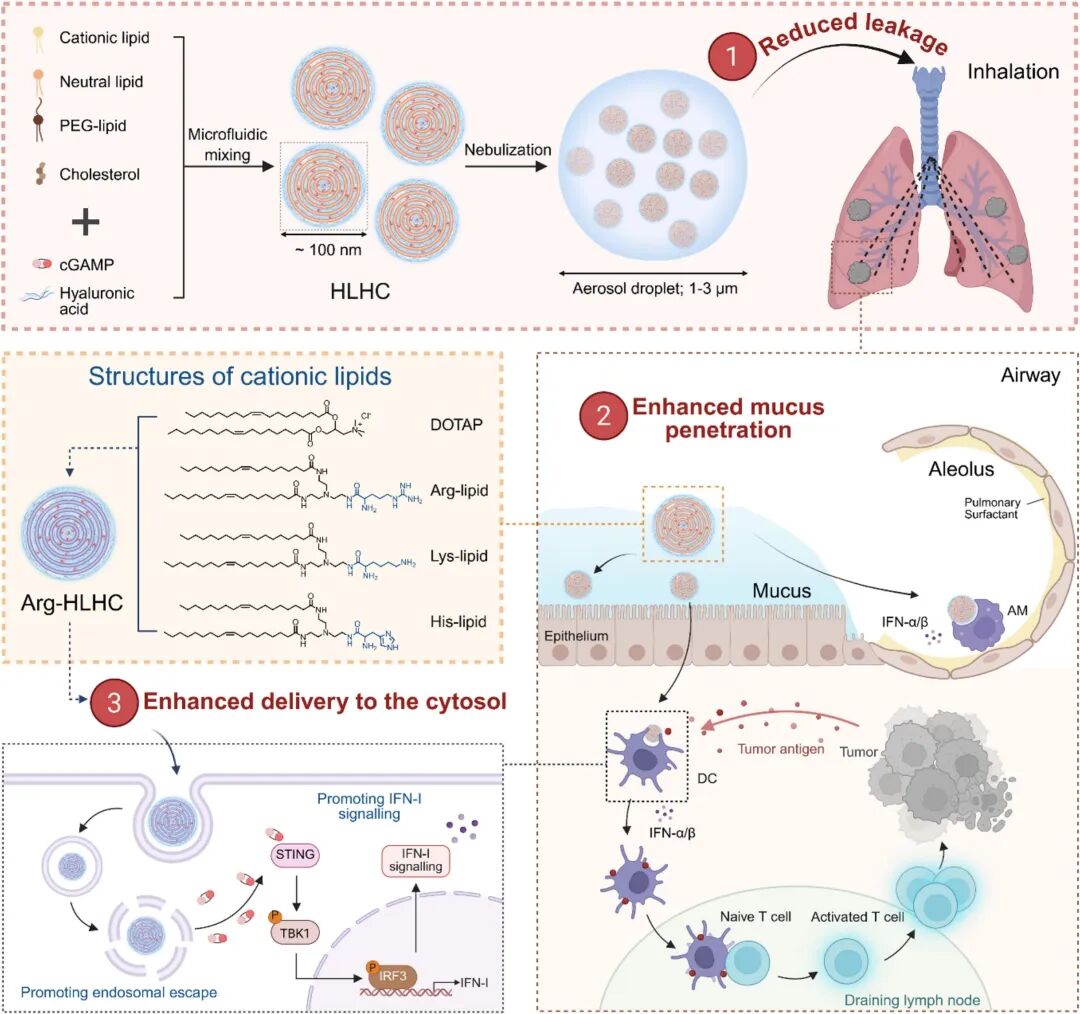
Fig 1. Schematic illustration of modulating nanostructure and chemical structure of cGAMP-loaded lipid nanoparticles for potent mucosal immunity induction
The research team prepared the multi-layered nanoparticle HLHC via microfluidic technology, which presented an onion-like multi-layered concentric structure (Fig 2a). Compared with single-layer liposomes (LC), HLHC maintained structural integrity and stable drug encapsulation during nebulization (Fig 2c-f), while LC underwent membrane fusion and drug leakage (Fig 2b, g-j).
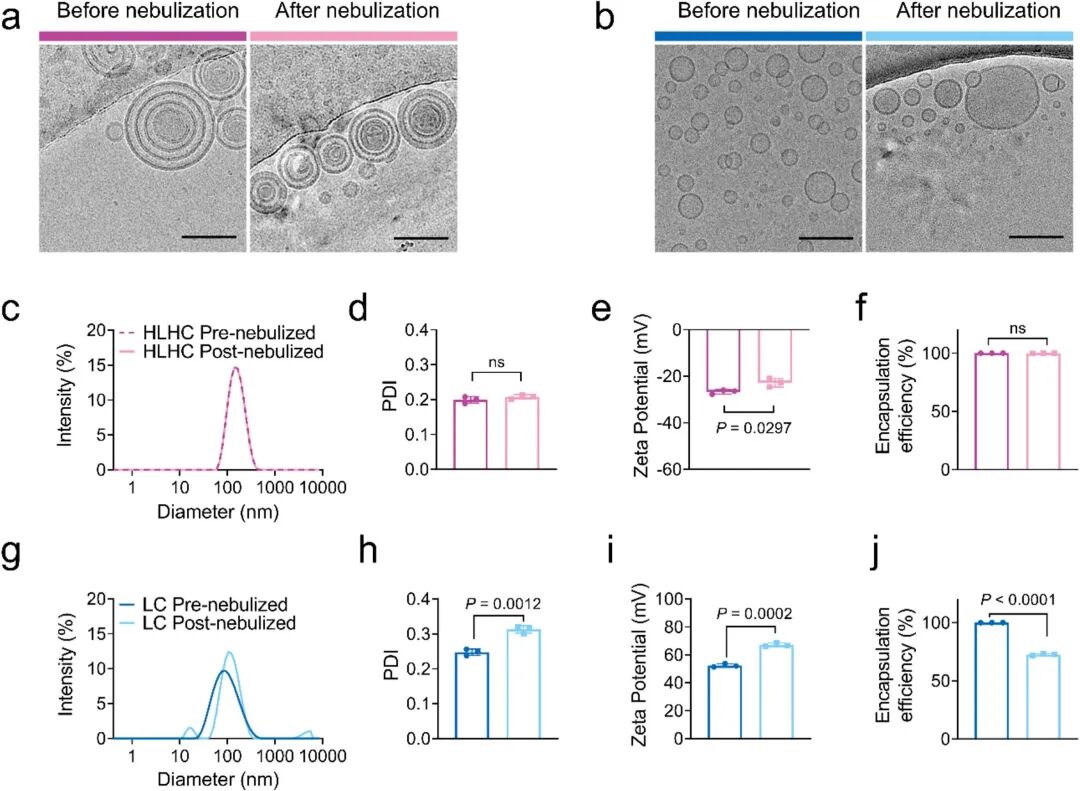
Fig 2. Effects of nebulization on HLHC and LC
The team then investigated the delivery behavior of the formulation in mucus penetration. After incubating the formulation with bronchoalveolar lavage fluid (BALF) at 37℃ for 0-6 hours, HLHC maintained particle size and PDI stability in BALF, while LC rapidly aggregated due to electrostatic interaction between positive charges and mucus (Fig 3a-d). Multi-particle tracking technology revealed that the mucus penetration capacity of HLHC was 35 times higher than that of single-layer liposomes; control experiments confirmed that the surface HA coating also contributed to improved mucus penetration efficiency (Fig 3e-g). FRET-FLIM technology further verified that the multi-layered structure effectively maintained the integrity of the surface HA coating and prevented dissociation in the mucus environment (Fig 3h-l). After inhalation, HLHC was widely distributed in lung airways and parenchyma (Fig 3m-p).
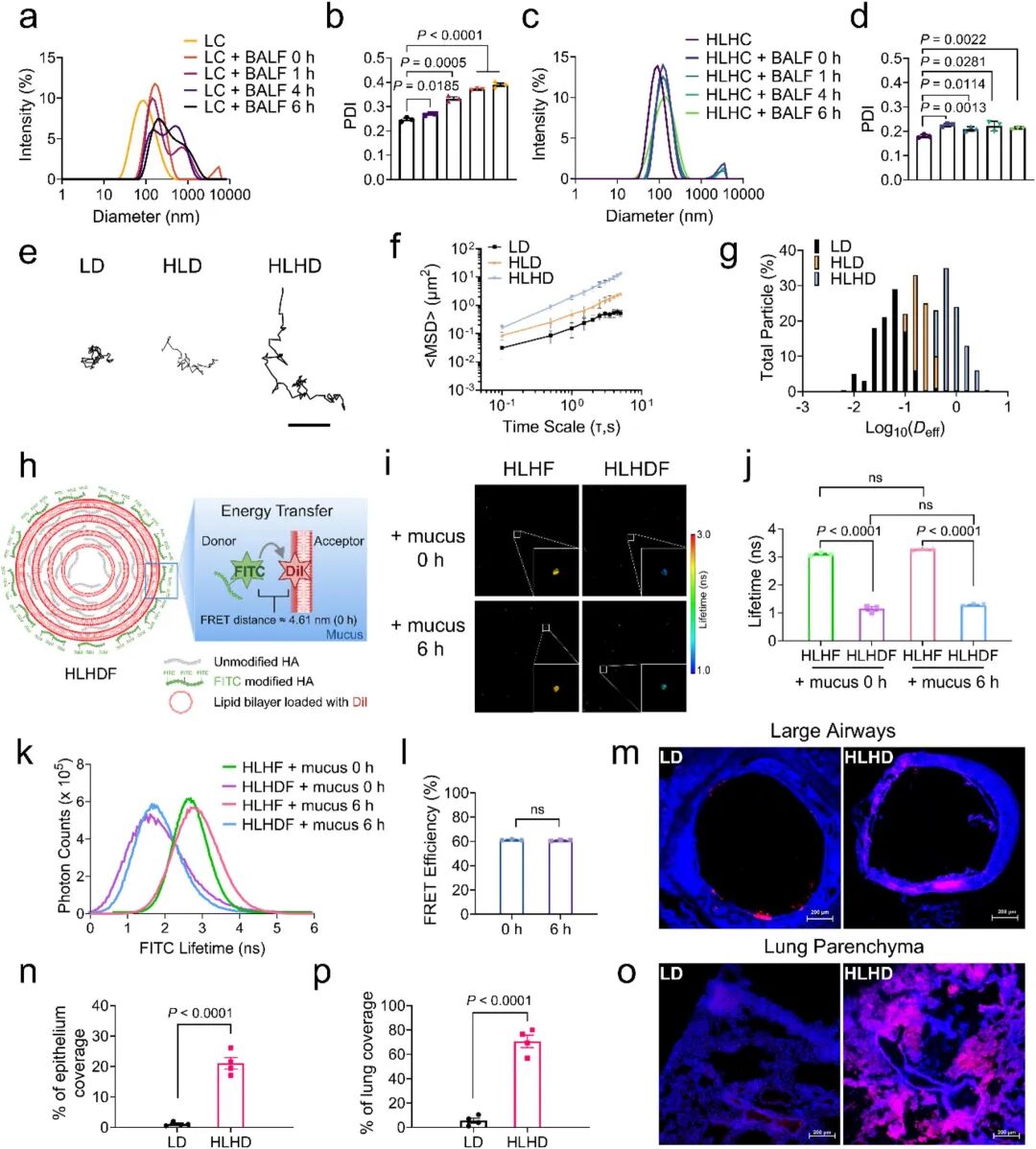
Fig 3. Evaluation of mucus penetration capacity of LC and HLHC
Subsequent in vivo experiments validated the delivery efficiency of HLHC. Dual labeling with DiR (carrier tracking) and SRB (cGAMP mimic) showed that the multi-layered structure significantly prolonged the pulmonary retention time of water-soluble drugs, reduced drug leakage into the systemic circulation (especially the liver), and promoted preferential uptake by pulmonary APCs including alveolar macrophages (AMs), interstitial macrophages (IMs), and dendritic cells (DCs) (Fig 4a-j). Immunofluorescence staining confirmed colocalization of HLHC with CD11c+ APCs but not tumor cells, verifying its immune cell targeting ability (Fig 4k). The research team then evaluated the therapeutic effects of single-layer and multi-layered formulations in the MC38-LUC lung metastasis model. Compared with free cGAMP and LC, inhaled HLHC significantly inhibited tumor progression (Fig 5b-e). Mechanistic studies confirmed that the therapeutic efficacy depended on host STING pathway activation (Fig 5g-h).
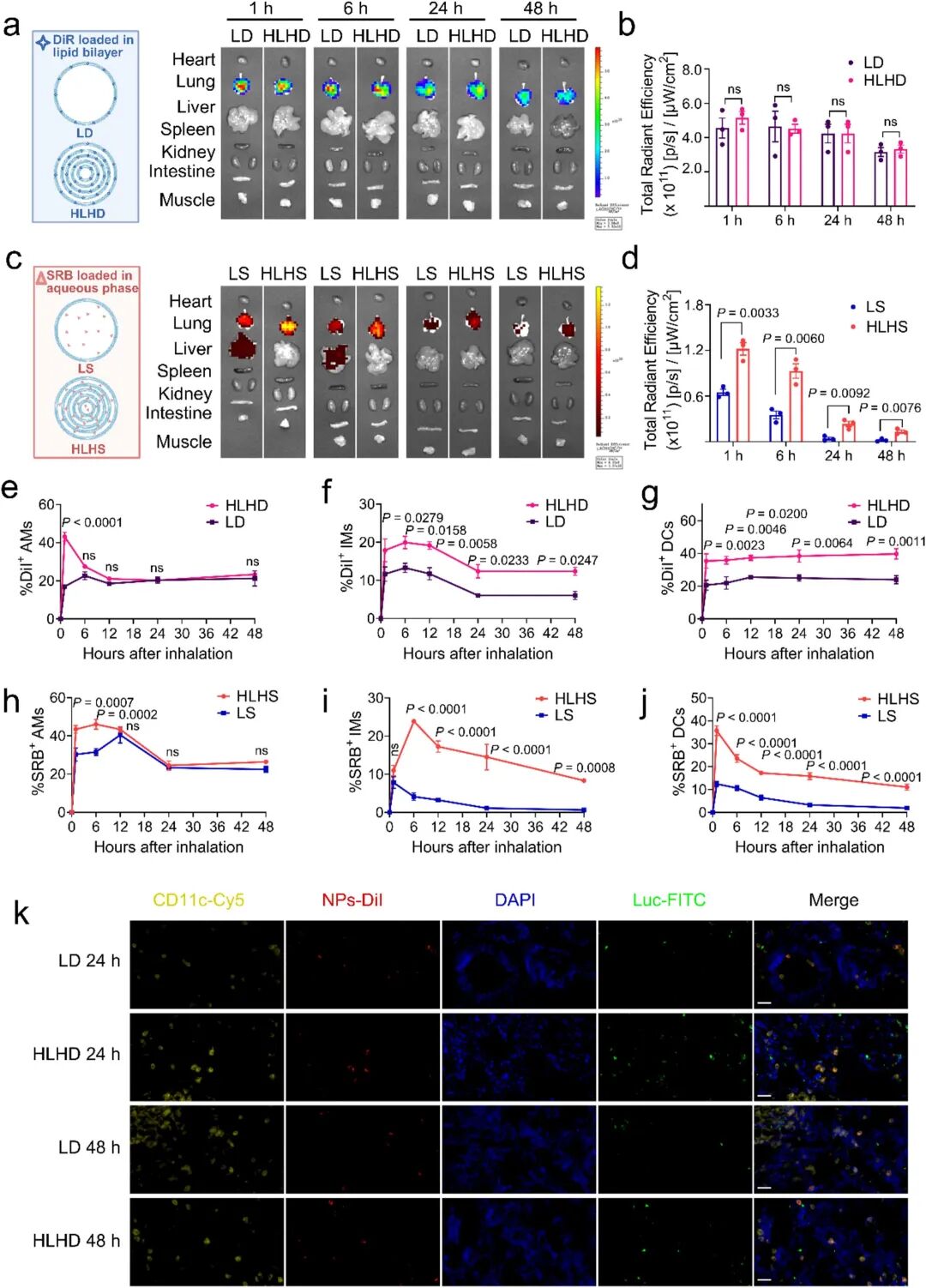
Fig 4. Multi-layered nanostructure enables sustained pulmonary retention and targeted delivery to APCs
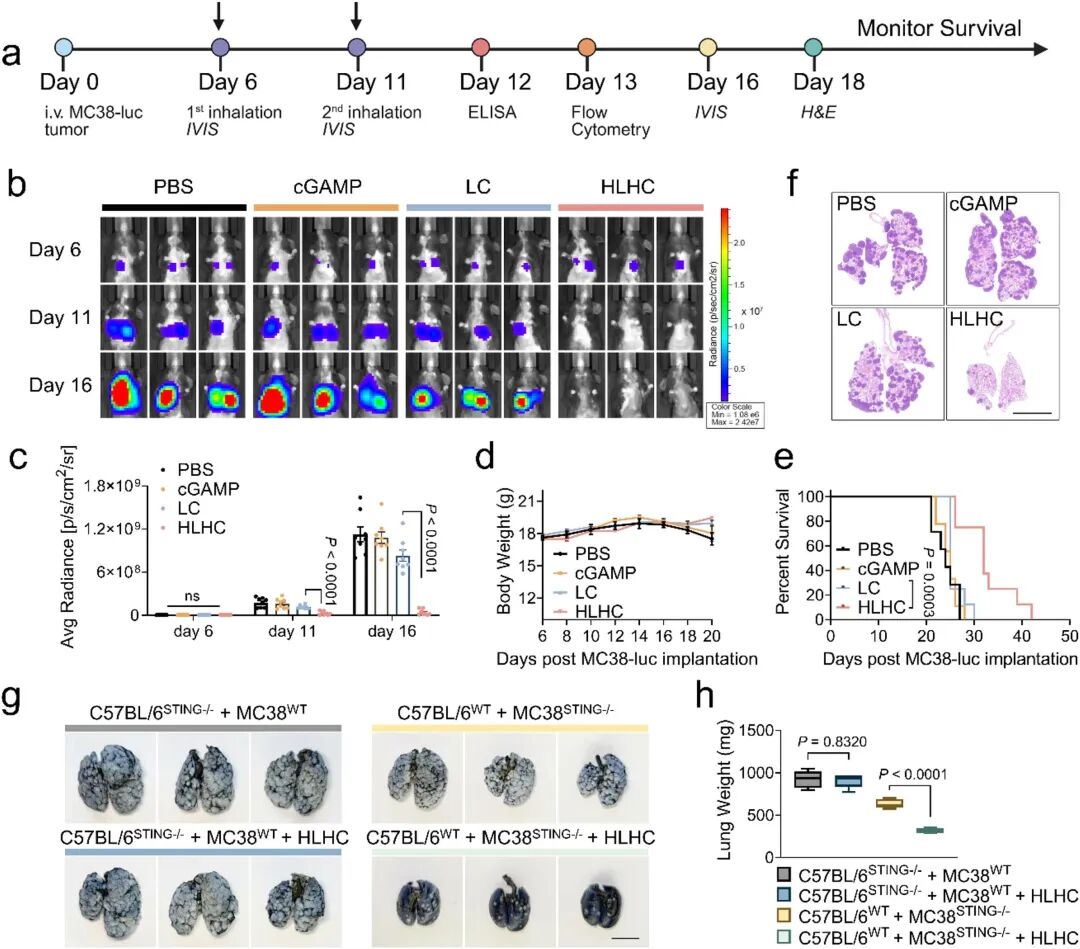
Fig 5. Multi-layered HLHC effectively inhibits MC38-LUC lung metastatic lesions
Elevate your lung metastasis research with our high-purity MC38-LUC cells—consistent luciferase activity ensures sensitive, non-invasive tumor monitoring for immunotherapy and nanodelivery efficacy assessment. View Now>>
Despite the favorable efficacy of HLHC, the research team further optimized lipid chemical structures to improve cytosolic delivery efficiency. Through five rounds of iterative screening, the team systematically evaluated key parameters including neutral phospholipid tail chain unsaturation, PEGylated lipid chain length/molecular weight/density, and cationic lipid chemical structures. Finally, arginine-modified HLHC (Arg-HLHC) was developed, which exhibited a 3.6-fold higher in vivo IFN-β induction capacity than DOTAP-HLHC (Fig 6e), demonstrating excellent cytosolic delivery efficiency. This advantage originated from the high pKa of arginine and its guanidyl group providing triple hydrogen bond donors, which synergized with cationic charges to enhance membrane disruption capacity—a mechanism consistent with studies on viral fusion peptides. The ultimate therapeutic efficacy of optimized Arg-HLHC was then assessed. Low-dose Arg-HLHC (1.5 μg cGAMP) achieved 100% complete remission in the MC38-LUC lung metastasis model (Fig 7b-f), with efficacy significantly superior to DOTAP-HLHC and Onpattro®-like formulation (MC3-LC). In C57BL/6 STING-/- mice, Arg-HLHC completely lost its therapeutic effect, confirming that its efficacy also relied on host STING pathway activation.
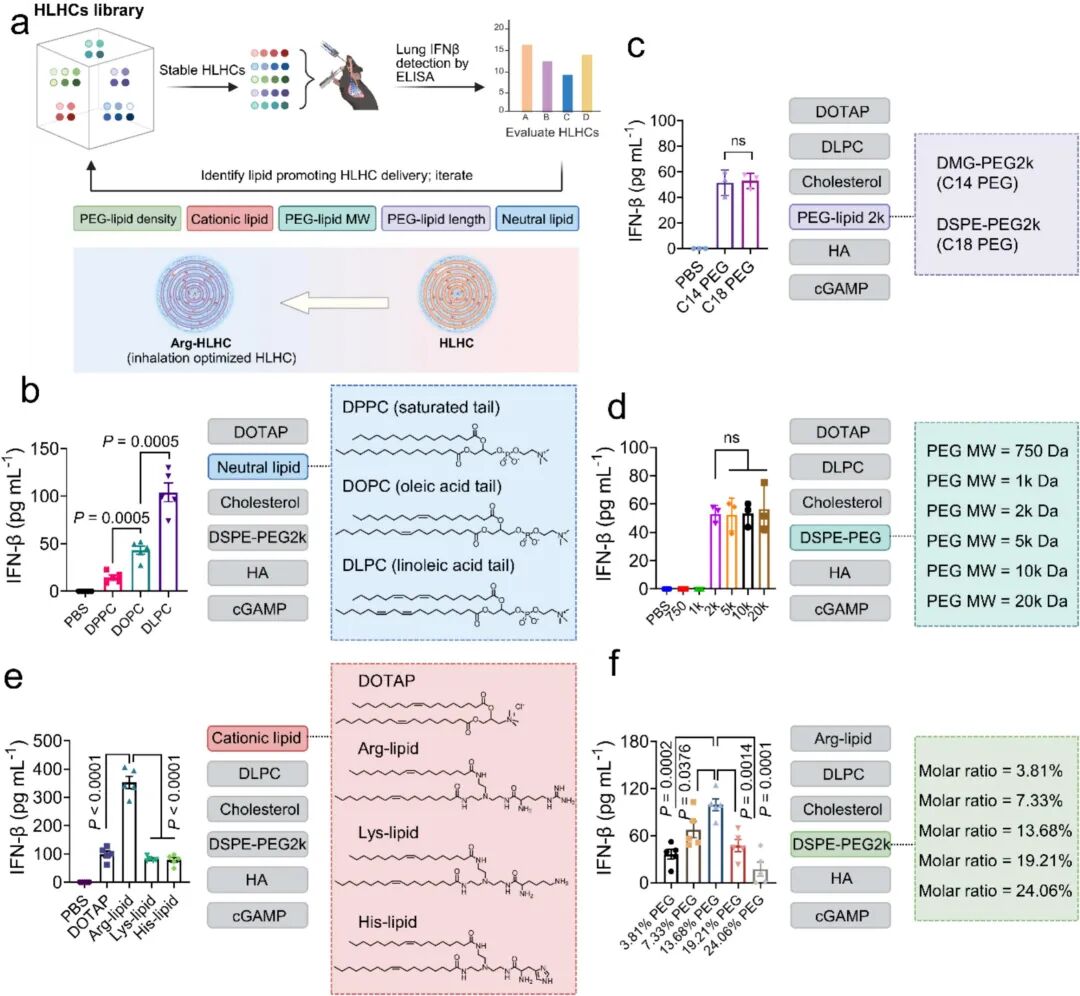
Fig 6. Iterative optimization of lipid chemical structures of HLHC
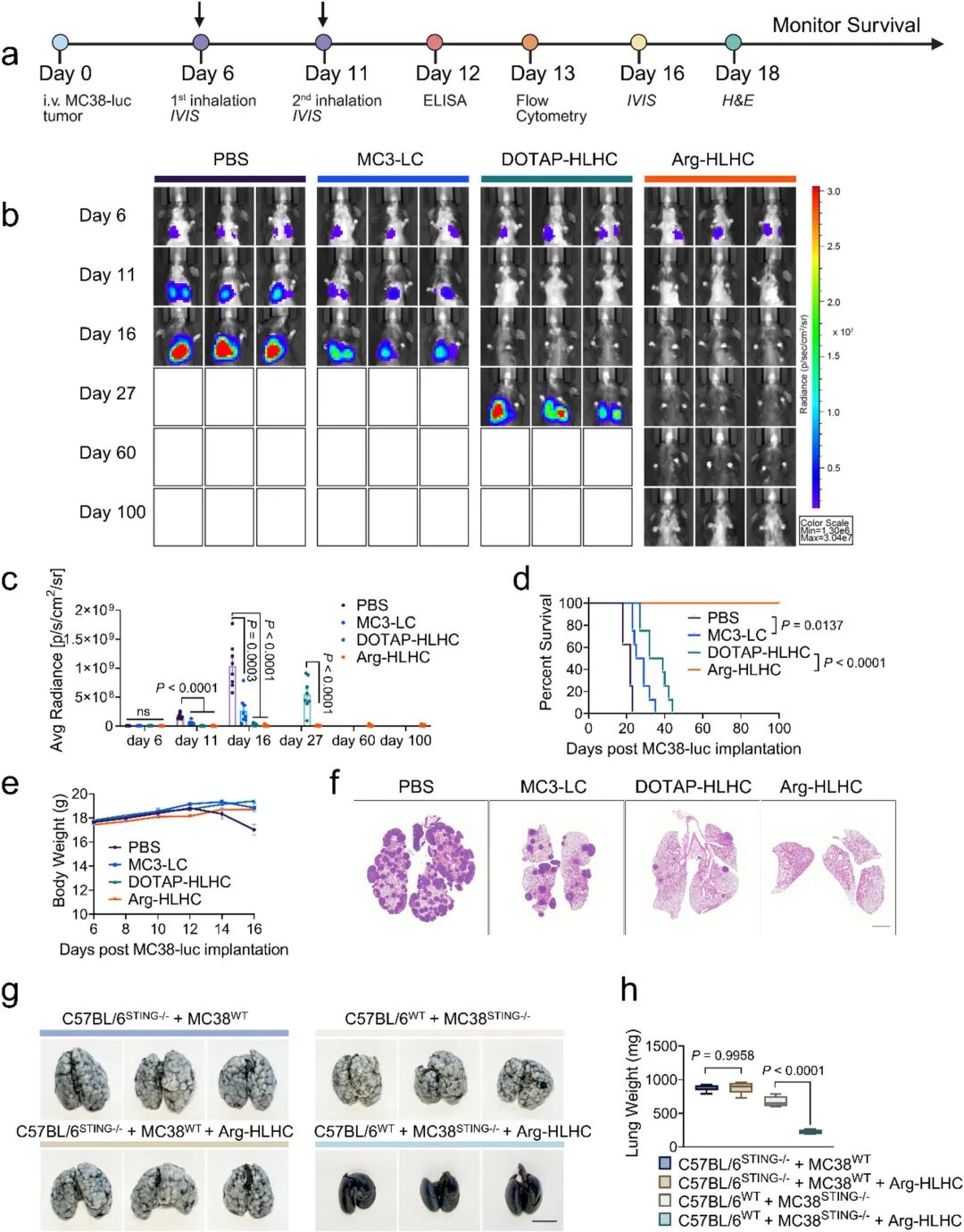
Fig 7. Multi-layered Arg-HLHC enables complete eradication of MC38-LUC lung metastatic lesions
Mice with complete tumor remission still achieved complete tumor clearance when re-challenged with the same tumor cells 100 days later (Fig 8a-c). Meanwhile, the populations of memory CD4+ and CD8+ T cells were significantly increased in the lung tissue, tumor-draining lymph nodes and spleens of these mice (Fig 8d-u). These results indicated that inhaled Arg-HLHC therapy successfully induced a robust immune memory effect, which could prevent tumor recurrence.
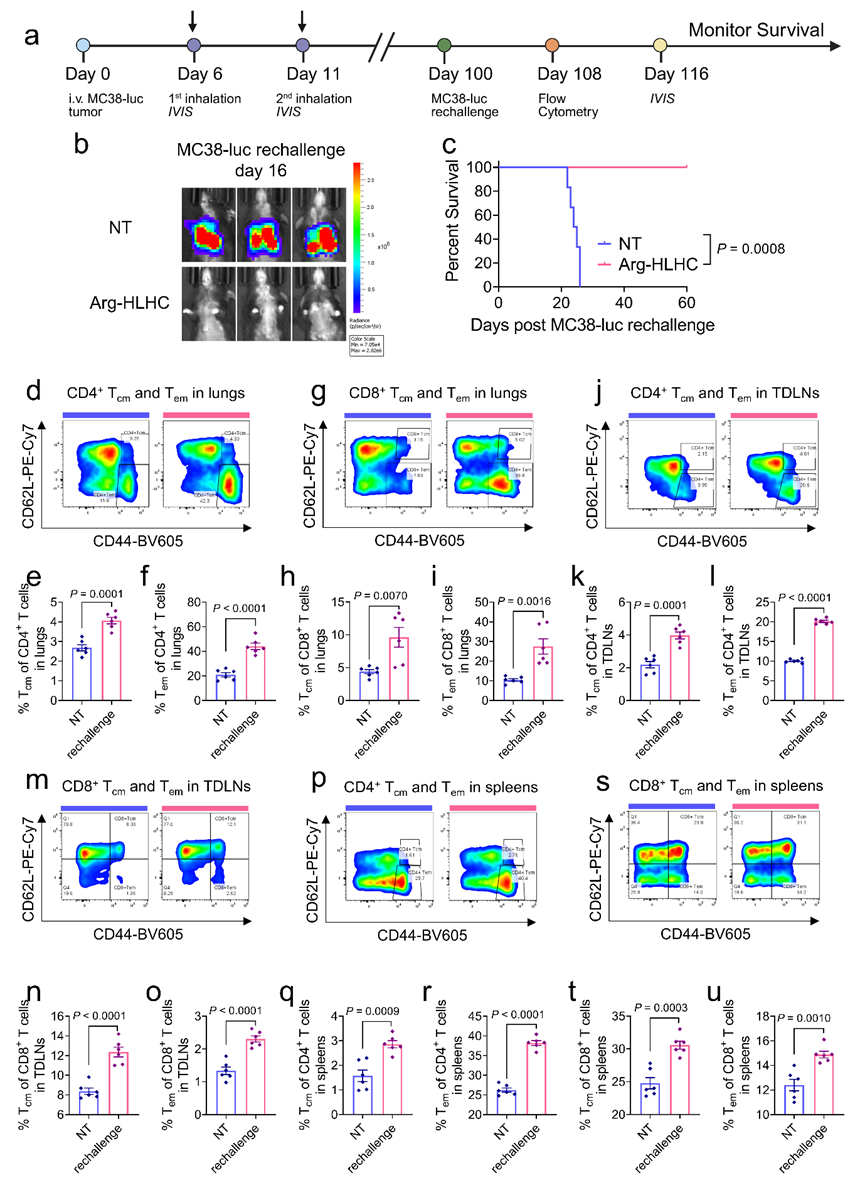
Fig 8. Tumor rechallenge study in MC38-LUC tumor-bearing mice cured by inhaled Arg-HLHC therapy
3. Key Findings
- The concentric multi-layered HLHC efficiently encapsulates cGAMP and maintains structural integrity and drug encapsulation efficiency under nebulization shear force.
- Compared with LC, HLHC has superior mucus penetration, drug sustained-release properties and APC targeting ability, enabling efficient mucosal immune activation.
- Arg-HLHC, optimized via systematic lipid screening, achieves complete regression of MC38-LUC lung metastases via low-dose pulmonary administration. At the same cGAMP dose, its efficacy is significantly superior to Onpattro®-like formulation encapsulating cGAMP with good safety.

