SK-OV-3 Luc: OR7A10 GPCR Engineering Boosts CAR-NK Therapy Against Ovarian Cancer
Introduction
Ovarian cancer remains the most lethal gynecologic malignancy, with most patients presenting at advanced stages characterized by peritoneal dissemination. While CAR-NK cell therapy has shown promise in hematological malignancies, its application to solid tumors including ovarian cancer has been limited by three fundamental challenges: poor tumor infiltration, inadequate persistence within the immunosuppressive tumor microenvironment (TME), and rapid functional exhaustion.
Researchers at Yale University led by Professor Sidi Chen and Dr. Lei Peng addressed these barriers through an innovative in vivo functional gain-of-function screening approach. Instead of targeting individual suppressive pathways, they performed an unbiased genome-wide CRISPR activation (CRISPRa) screen in tumor-infiltrating CAR-NK cells to identify genes that could broadly enhance anti-tumor activity when overexpressed. Their work culminated in the identification of OR7A10, an orphan G protein-coupled receptor (GPCR) originally classified as an olfactory receptor, as the most potent enhancer of CAR-NK cell function across multiple solid tumor types.
Contact us to discuss your ovarian cancer research needs. Our SK-OV-3 Luc cells are rigorously validated for bioluminescence imaging and ideal for establishing intraperitoneal dissemination models. Request a quote now to get started.
Order now: SK-OV-3 Luciferase Cell Line / SK-OV-3-Luc-GFP Cell Line
In Vivo CRISPRa Screening Identifies OR7A10
The research team employed a two-stage screening pipeline. First, they performed an unbiased in vivo CRISPR activation screen using the SAM (synergistic activation mediator) sgRNA library in HER2-CAR-NK92 cells, which were then infused into HT29 colon cancer-bearing immunodeficient mice. By sequencing sgRNAs from tumor-infiltrating NK cells versus input populations, they identified genes whose activation conferred a selective advantage for NK cell persistence and expansion within tumors. Among the top hits, OR7A10, SGSM2, and APLN showed the strongest enrichment signals.
To validate these findings in primary human NK cells with clinical relevance, the team conducted a second barcoded targeted open reading frame (ORF) screen using peripheral blood-derived primary human CAR-NK cells. Each ORF was linked to a DNA barcode, enabling quantitative tracking via next-generation sequencing. This secondary screen confirmed OR7A10 as the single most significantly enriched gene, establishing it as the lead candidate for further development.
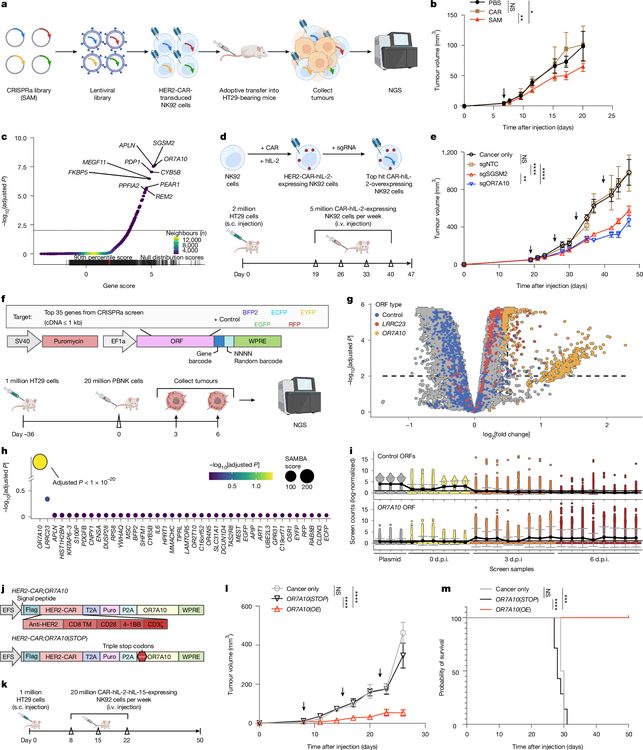
Figure 1. In vivo CRISPRa and barcoded ORF screening confirm OR7A10 as the top CAR-NK enhancer. (a) Schematic of the SAM-based in vivo CRISPRa screen in HER2-CAR-NK92 cells using HT29 tumor-bearing mice. (b) Volcano plot showing gene-level enrichment scores from tumor-infiltrating NK cells, with OR7A10, SGSM2, and APLN among top hits. (c) Barcoded ORF library validation in primary human NK cells confirming OR7A10 dominance. (d) "All-in-one" vector design co-expressing OR7A10 cDNA with HER2-CAR. (e) Tumor growth curves demonstrating near-complete regression in the OR7A10-engineered group versus controls.
Multi-dimensional Functional Enhancement of Primary CAR-NK Cells
The team validated OR7A10's effects using primary human NK cells isolated from seven independent healthy donors, transducing them with lentiviral vectors co-expressing OR7A10 and a mesothelin-targeting CAR construct. Across all donors, OR7A10-expressing CAR-NK cells exhibited markedly enhanced cytotoxicity against HT29 colon carcinoma cells compared to control CAR-NK cells. This enhancement was consistent across multiple effector-to-target ratios and extended to diverse target cell lines including H1299 lung carcinoma, MCF7 breast adenocarcinoma, and K562 leukemia cells, indicating broad applicability.
Mechanistic analyses revealed that OR7A10 overexpression upregulated key effector molecules including granzyme B, perforin, Fas ligand (FasL), and TRAIL. Cytokine production assays showed increased IFN-gamma and TNF-alpha secretion. Surface marker profiling demonstrated elevated expression of activation markers CD69, CD25, and 4-1BB alongside increased CXCR2 chemokine receptor levels. Critically, exhaustion markers TIM-3, LAG-3, PD-1, and NKG2A were all significantly reduced, suggesting that OR7A10 not only enhances effector function but also protects against the dysfunctional state that commonly limits cellular immunotherapies in solid TMEs.
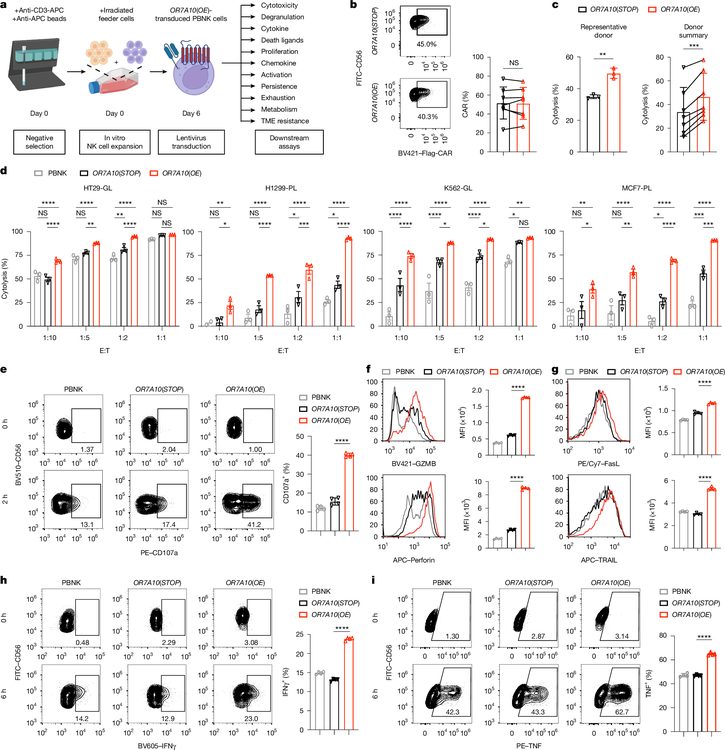
Figure 2. OR7A10 ORF engineering enhances CAR-NK anti-tumor function across multiple dimensions. (a) Workflow schematic: primary NK isolation, expansion, transduction, and functional assessment. (b) CAR expression efficiency confirmed by flow cytometry across seven donors. (c) Cytotoxicity assay showing enhanced killing of HT29 cells by OR7A10-CAR-NK from all seven donors. (d) Broad-spectrum cytotoxicity against H1299, K562, and MCF7 targets. (e) Increased degranulation (CD107a). (f-h) Elevated granzyme B, perforin, and death ligand expression. (i-k) Enhanced cytokine production (IFN-gamma, TNF-alpha), proliferation, and chemokine receptor (CXCR2) upregulation. (l) Reduced exhaustion markers (TIM-3, LAG-3, PD-1, NKG2A).
Metabolic Adaptation and Tumor Microenvironment Resistance
One of the most remarkable aspects of OR7A10 engineering is its ability to confer metabolic resilience. Seahorse extracellular flux analysis revealed that OR7A10-overexpressing CAR-NK cells exhibit elevated basal oxygen consumption rate, maximal respiratory capacity, and spare respiratory capacity compared to controls. Mitochondrial staining and electron microscopy confirmed increased mitochondrial mass with enlarged individual mitochondria, indicating enhanced mitochondrial biogenesis rather than fusion-mediated remodeling.
The team systematically evaluated OR7A10-engineered CAR-NK cells under six distinct TME stress conditions mimicking the hostile ovarian cancer microenvironment: lactic acid-induced acidity (pH ~6.5), TGF-beta-mediated suppression, IL-2 deprivation, immunosuppressive drugs (cyclosporine A, tacrolimus), adenosine signaling via A2A receptor agonism, and hypoxia induced by cobalt chloride treatment. Under every challenge, OR7A10-CAR-NK cells maintained robust cytotoxic activity while control cells showed severe functional impairment. This comprehensive resistance profile positions OR7A10 engineering as particularly relevant for ovarian cancer, where the peritoneal TME is characterized by hypoxia, adenosine accumulation, and abundant TGF-beta.
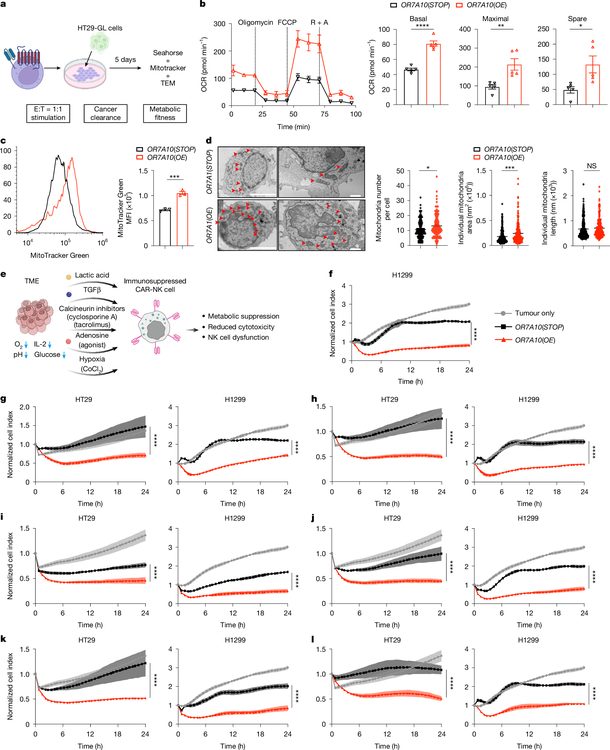
Figure 3. OR7A10 confers metabolic adaptability and multi-faceted TME resistance on engineered CAR-NK cells. (a-c) Seahorse analysis showing elevated basal OCR, maximal respiration, and spare capacity in OR7A10-CAR-NK cells. (d-e) MitoTracker staining and EM images revealing increased mitochondrial mass and enlarged mitochondrial area. (f) Systematic TME challenge scheme covering six inhibitory conditions. (g-l) Cytotoxicity maintained by OR7A10-CAR-NK under acidosis, TGF-beta, IL-2 deprivation, immunosuppressants, adenosine, and hypoxia, while control cells fail under each condition.
In Vivo Efficacy: From Subcutaneous Models to Intraperitoneal Ovarian Cancer
The therapeutic potential of OR7A10 engineering was validated across three clinically relevant solid tumor models. In the HT29 subcutaneous colorectal cancer model, weekly intravenous infusion of IL-15-secreting OR7A10-CAR-NK cells achieved significant tumor growth inhibition. More impressively, in the orthotopic MCF7 mammary fat pad model representing locally invasive breast cancer, OR7A10-CAR-NK therapy resulted in 100% complete remission with long-term, tumor-free survival in all treated mice — a result not achieved by any prior CAR-NK monotherapy approach in this setting.
For ovarian cancer specifically, the team established an intraperitoneal disseminated model using luciferase-expressing SK-OV-3 Luc cells. This model faithfully recapitulates the clinical pattern of ovarian carcinomatosis, wherein tumor cells spread throughout the peritoneal cavity and seed onto serosal surfaces. Following systemic administration of OR7A10-engineered CAR-NK cells expressing human IL-15, serial bioluminescence imaging (BLI) demonstrated sustained reduction of abdominal tumor signal compared to progressive disease in control animals. The survival benefit was statistically significant, confirming that the in vitro advantages of OR7A10 translate into meaningful therapeutic outcomes in this challenging disease context.
Pharmacokinetic analysis revealed that OR7A10-engineered CAR-NK cells exhibited superior accumulation in both peripheral blood and tumor tissue at days 3 and 6 post-infusion, with no differences in splenic distribution. However, by day 14, NK cell numbers declined in all groups, suggesting that while OR7A10 enhances short-term trafficking and persistence, strategies to extend long-term engraftment remain an important avenue for future optimization.
Place your order for our premium SK-OV-3 Luc cells and receive expert technical support for establishing intraperitoneal ovarian cancer BLI models. Browse our catalog and find the perfect cell line for your immunotherapy research program.
Order now: SK-OV-3 Luciferase Cell Line / SK-OV-3-Luc-GFP Cell Line
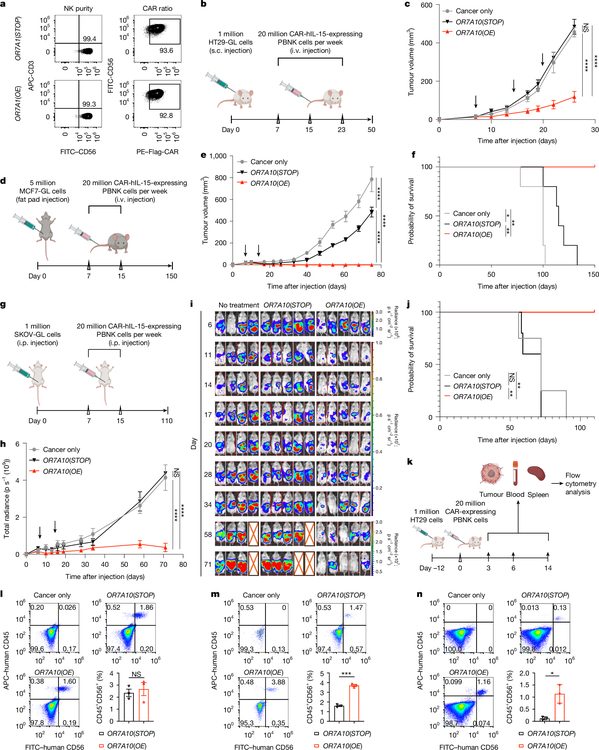
Figure 4. OR7A10-engineered primary human CAR-NK cells demonstrate potent in vivo efficacy across three solid tumor platforms. (Top row) Flow cytometry confirming NK purity and CAR expression in infused cells. (Model 1) HT29 subcutaneous colon cancer: weekly IV infusion of hIL-15+ OR7A10-CAR-NK cells yields significant tumor suppression. (Model 2) MCF7 orthotopic breast cancer (mammary fat pad): OR7A10 group achieves 100% complete response with durable, tumor-free survival. (Model 3) SK-OV-3 Luc intraperitoneal ovarian cancer: bioluminescence imaging tracks progressive abdominal disease in controls versus sustained tumor control and survival advantage in the OR7A10 group. (Bottom panels) Day 3 and day 6 biodistribution shows greater OR7A10-CAR-NK accumulation in blood and tumor; tumor weight reduced at day 6; persistence limited by day 14 across all groups.
Molecular Mechanism: GNAS-cAMP-PKA-ERK-NF-kappaB Signaling Cascade
To elucidate how OR7A10 exerts its pleiotropic enhancing effects, the team performed single-cell RNA sequencing on tumor-infiltrating CAR-NK cells isolated from HT29-GL-bearing mice. They identified eight distinct NK cell subsets ranging from immature to highly differentiated states. Notably, OR7A10 overexpression did not substantially alter subset composition, instead producing a coordinated transcriptional remodeling across all clusters characterized by upregulation of effector molecules (GZMB, IFNG, LTA), anti-apoptotic genes (BCL2, BCL2L1), and downregulation of pro-apoptotic BAX.
Pathway enrichment analysis pointed to activation of NFAT, JNK, ATF2, RB-1, and IL-23 signaling axes. Biochemical studies ultimately defined the core cascade: OR7A10 signals through GNAS (Gs alpha subunit), elevating intracellular cyclic AMP, activating protein kinase A (PKA), which then stimulates ERK phosphorylation and nuclear translocation of NF-kappaB. This pathway explains the simultaneous enhancement of metabolism (via cAMP-PKA-ERK-driven mitochondrial biogenesis), cytotoxicity (via NF-kappaB-dependent transcription of effector genes), and survival (via BCL2 family induction). The causal inference framework applied to the scRNA-seq data further identified a TCR-like calcium-NFAT axis as the primary driver linking OR7A10 expression to IFNG, TNF, and LTA upregulation, while GPCR1 signaling downregulation correlated inversely with immunosuppressive gene programs involving FGR and TGFB1.
Conclusion and Clinical Outlook
This Nature study establishes OR7A10 GPCR engineering as a transformative strategy for overcoming the historical limitations of CAR-NK cell therapy against solid tumors. By employing cDNA overexpression within a single vector alongside the CAR construct, the approach avoids the manufacturing complexity associated with multiplexed gene editing while delivering multi-dimensional functional benefits: enhanced cytotoxicity, improved metabolic fitness, broad TME resistance, and reduced exhaustion across multiple donor-derived primary NK cell products.
The demonstration of sustained tumor control in the SK-OV-3 Luc intraperitoneal ovarian cancer model is particularly noteworthy given the unmet need for effective immunotherapies in advanced-stage disease. Bioluminescence-enabled longitudinal tracking provided clear visual evidence of therapeutic benefit, underscoring the value of well-characterized luciferase-tagged cell lines like SK-OV-3 Luc for preclinical evaluation of novel cellular therapies. As the field moves toward clinical translation, OR7A10-engineered CAR-NK cells represent a promising off-the-shelf candidate for treating ovarian cancer and other solid malignancies where conventional CAR approaches have struggled.
References
Bernard PL et al. OR7A10 GPCR engineering boosts CAR-NK therapy against solid tumours. Nature. 2026; DOI: 10.1038/s41586-026-10149-8

