HeLa luc in Cervical Cancer Immunotherapy Research: Decoding NAD⁺-Mediated PD-L1 Regulation and Immune Evasion
Introduction
Cervical cancer is one of the most prevalent gynecologic malignancies worldwide, with approximately 660,000 new cases and 350,000 deaths reported annually. High-risk human papillomavirus (HPV) infection, particularly HPV18 — the strain carried by the historic HeLa cell line — is the primary etiological driver. Despite progress in vaccination and early detection, advanced and recurrent cervical cancer continues to carry a poor prognosis.
PD-1/PD-L1 immune checkpoint therapy has emerged as a promising treatment strategy for cervical cancer. Pembrolizumab, the first FDA-approved anti-PD-1 antibody for PD-L1-positive cervical cancer, demonstrated meaningful clinical benefit in the KEYNOTE-158 trial. However, a substantial proportion of patients — including those with high PD-L1 tumor expression — fail to respond, highlighting the existence of complex resistance mechanisms that remain poorly understood.
The HeLa LUC cell line (stably expressing firefly luciferase) is a cornerstone tool for cervical cancer in vivo research, enabling non-invasive real-time bioluminescence imaging (BLI) of tumor growth, drug biodistribution, and therapeutic efficacy in xenograft mouse models. Understanding the molecular mechanisms underlying immunotherapy resistance in HeLa-based models is therefore directly translatable to the clinic.
HeLa luciferase cells enable non-invasive BLI tracking of tumor growth and immunotherapy responses in living animals — an indispensable tool for PD-L1 checkpoint biology research. Contact us today to discuss how HeLa LUC can accelerate your cervical cancer immunotherapy studies.
Order now: Hela Cell Line / Hela Luciferase Cell Line / Hela-Luc-GFP Cell Line
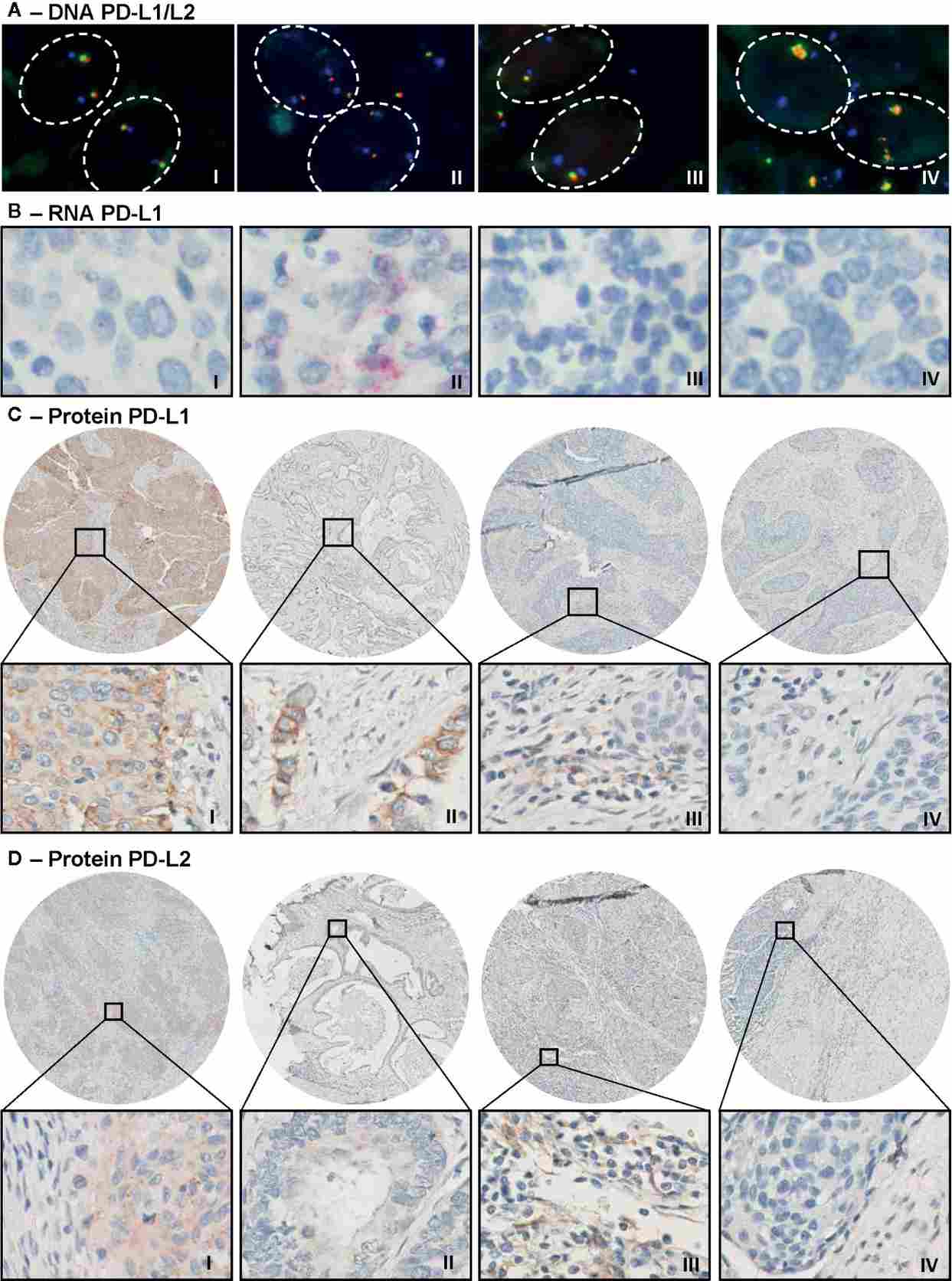
Figure 1. Representative immunohistochemistry and FISH analysis of PD-L1/PD-L2 expression in cervical cancer tissues. PD-L1 is widely expressed at both transcript and protein levels in cervical squamous cell carcinoma and adenocarcinoma, providing a rationale for checkpoint immunotherapy targeting this axis.
Key Findings: The NAMPT/SIRT1/PD-L1 Metabolic Axis
A landmark 2025 study published in Advanced Science (IF 14.1) by Lu et al. from Jiangnan University unveiled a previously unrecognized layer of PD-L1 regulation in cervical cancer. The research team identified that nuclear localization of PD-L1 — rather than simply surface expression — is a critical determinant of immunotherapy resistance in advanced cervical cancer patients.
Using multiple cervical cancer cell lines including HeLa, SiHa, and C33A, the investigators demonstrated that NAD⁺ metabolism plays a pivotal role in governing PD-L1 subcellular localization. Specifically, the salvage pathway enzyme NAMPT (nicotinamide phosphoribosyltransferase) generates NAD⁺ that fuels SIRT1, a class III histone deacetylase. SIRT1 in turn deacetylates histone H3 at lysine 27 (H3K27), reducing acetylation levels at the PD-L1 gene promoter and triggering PD-L1 nuclear translocation.
Acetylome proteomic profiling confirmed that SIRT1 is the central regulator of H3K27 deacetylation in this context. When NAMPT activity was inhibited using FK866 — a clinical-stage NAMPT inhibitor — NAD⁺ levels dropped, SIRT1 activity was suppressed, H3K27 acetylation was restored, and PD-L1 redistributed from the nucleus back to the cell surface, thereby becoming accessible to immune effectors and checkpoint antibodies.
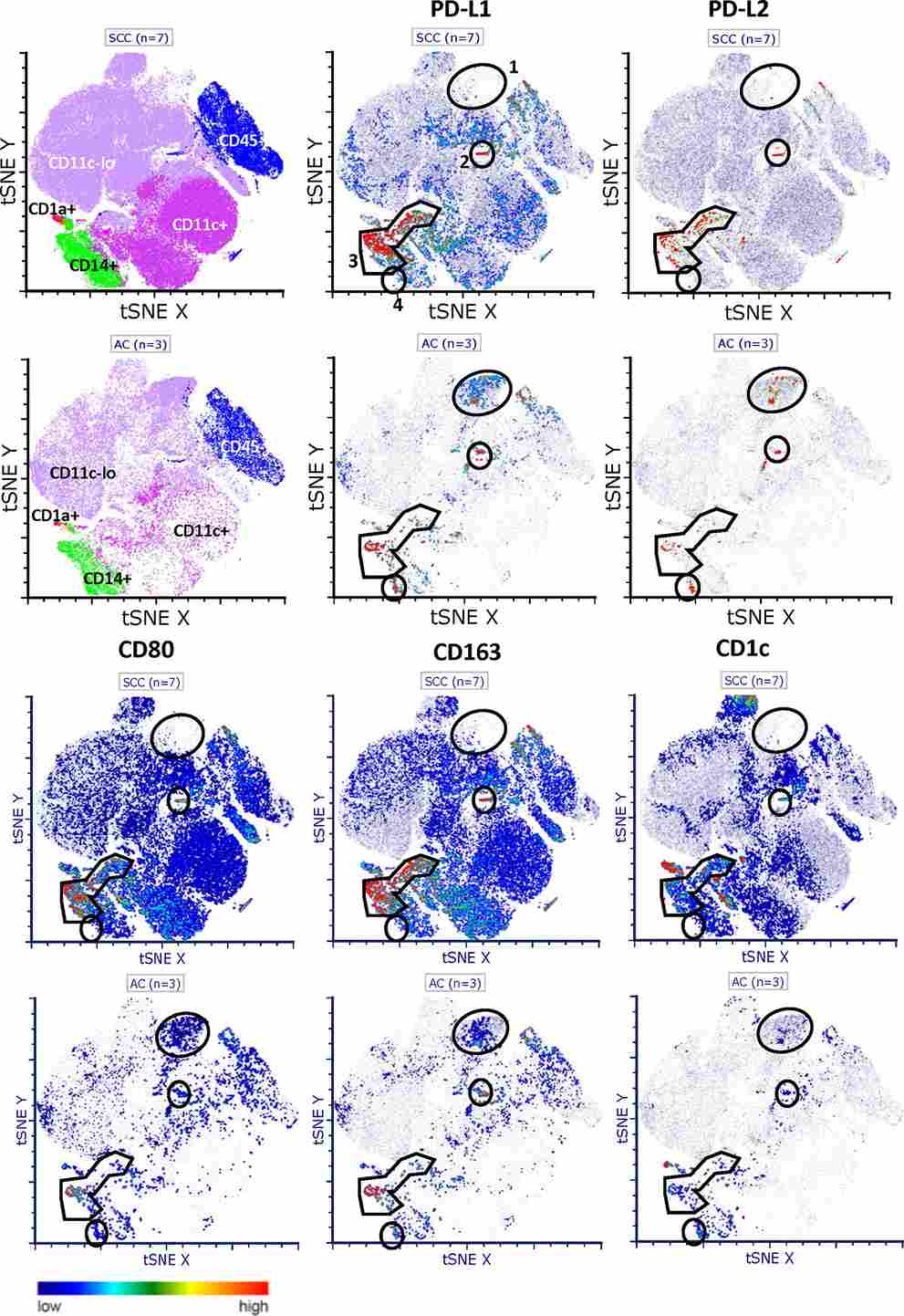
Figure 2. High-dimensional flow cytometric analysis (t-SNE visualization) of PD-L1 and PD-L2 co-expression across distinct macrophage populations in cervical cancer tumor microenvironments. NAD⁺-driven immune suppression operates at multiple levels, including both tumor cell-intrinsic PD-L1 nuclear relocation and extrinsic myeloid compartment regulation.
Synergistic Antitumor Efficacy: FK866 + Anti-PD-L1 Combination
The translational significance of these findings was validated in vivo. In xenograft mouse models, the combination of FK866 (NAMPT inhibitor) and anti-PD-L1 antibody demonstrated synergistic antitumor effects compared to either agent alone. This finding provides a compelling mechanistic rationale for combining metabolic inhibitors with immune checkpoint blockade in cervical cancer — a strategy now being explored in clinical trials.
Critically, the authors established that PD-L1 nuclear localization score could serve as a novel stratification biomarker for patient selection. Advanced-stage cervical cancer biopsies showed significantly elevated nuclear PD-L1, correlating with reduced response to anti-PD-1/PD-L1 monotherapy. Re-sensitization to immune checkpoint therapy by targeting the NAMPT/SIRT1 axis thus represents a promising new therapeutic direction.
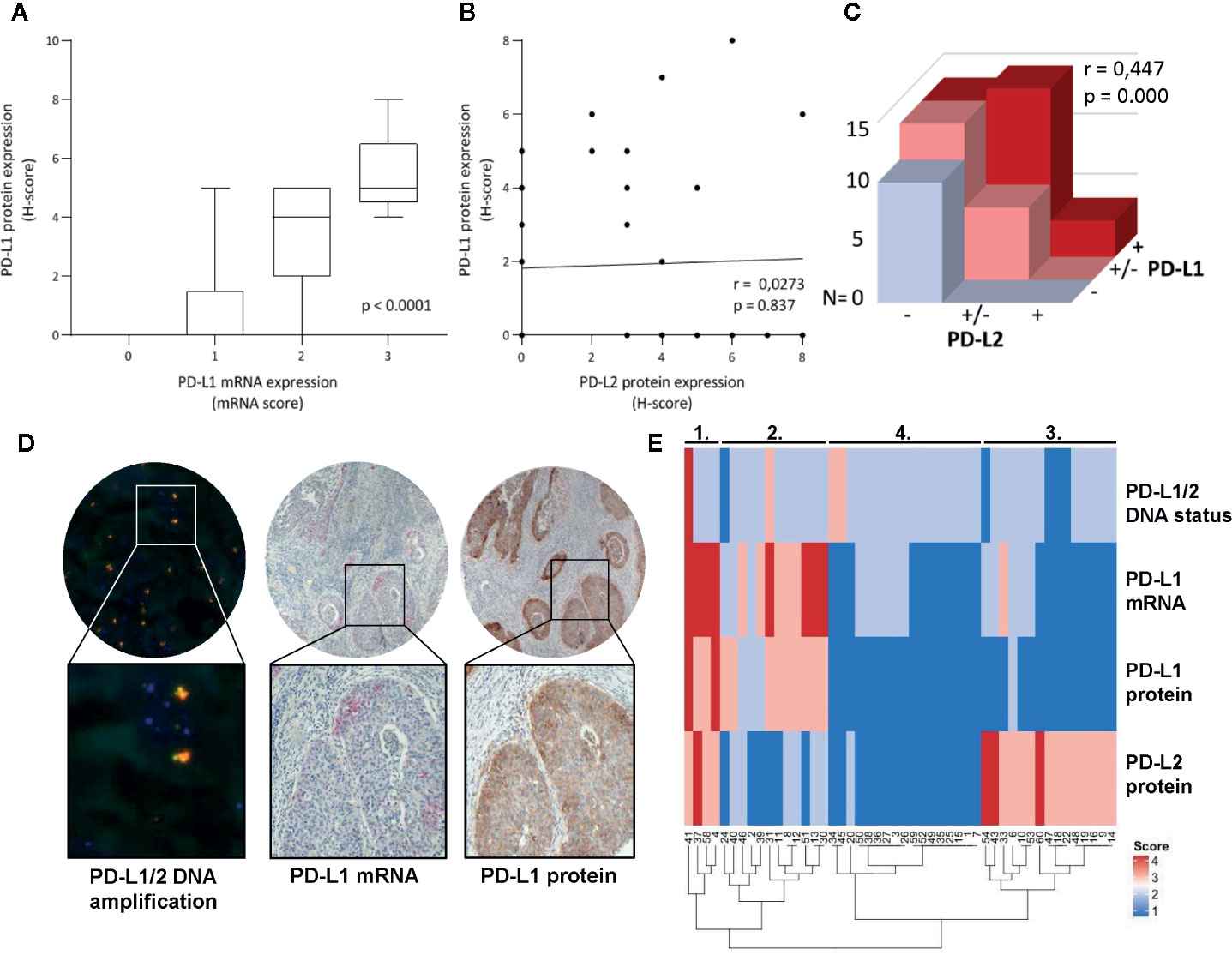
Figure 3. Correlation analysis of PD-L1/PD-L2 DNA copy number status, mRNA expression, and protein levels in cervical cancer. The data underscore the complexity of PD-L1 regulation at multiple tiers — from genomic amplification to post-translational modifications — relevant to the NAD⁺/SIRT1-mediated deacetylation mechanism described in the Adv Sci 2025 study.
Elevate your cervical cancer checkpoint research with HeLa LUC — the gold-standard bioluminescent model for non-invasive monitoring of tumor immunotherapy responses in vivo. Request a quote now and receive expert technical support for your xenograft BLI experiments.
Order now: Hela Cell Line / Hela Luciferase Cell Line / Hela-Luc-GFP Cell Line
Discussion: Implications for HeLa LUC-Based Preclinical Research
The discovery of NAD⁺-driven PD-L1 nuclear localization fundamentally reframes how we evaluate immunotherapy responses in cervical cancer preclinical models. Traditional assays that measure only surface PD-L1 expression may substantially underestimate the immunosuppressive state of tumors harboring high NAMPT activity. Incorporating nuclear PD-L1 quantification alongside conventional endpoints is therefore essential for accurate efficacy readouts.
HeLa LUC cells, derived from the HPV18-positive parent HeLa line, are particularly relevant for this research area given that HPV18 infection is mechanistically linked to dysregulated NAD⁺ metabolism and aberrant immune checkpoint expression. The stable luciferase reporter enables longitudinal, real-time BLI tracking of tumor dynamics in response to FK866, anti-PD-L1 antibodies, or combination regimens — without the need for tumor excision or endpoint sacrifice at each time point.
Subcutaneous and orthotopic HeLa LUC xenograft models in nude or NSG mice offer complementary advantages: subcutaneous models provide convenient caliper measurements alongside BLI quantification, while orthotopic cervical tumor models more faithfully recapitulate the immune microenvironment, lymph node architecture, and metastatic behavior relevant to clinical disease. Both applications benefit from the high sensitivity and dynamic range of luciferase-based bioluminescence.
Beyond the NAMPT/SIRT1/PD-L1 axis, HeLa LUC cells have been used extensively to evaluate gene therapies, nanoparticle-based drug delivery systems, oncolytic viral vectors, CAR-T cell therapies, and NK cell-mediated cytotoxicity — all of which require non-invasive, quantitative in vivo readouts that BLI uniquely provides.
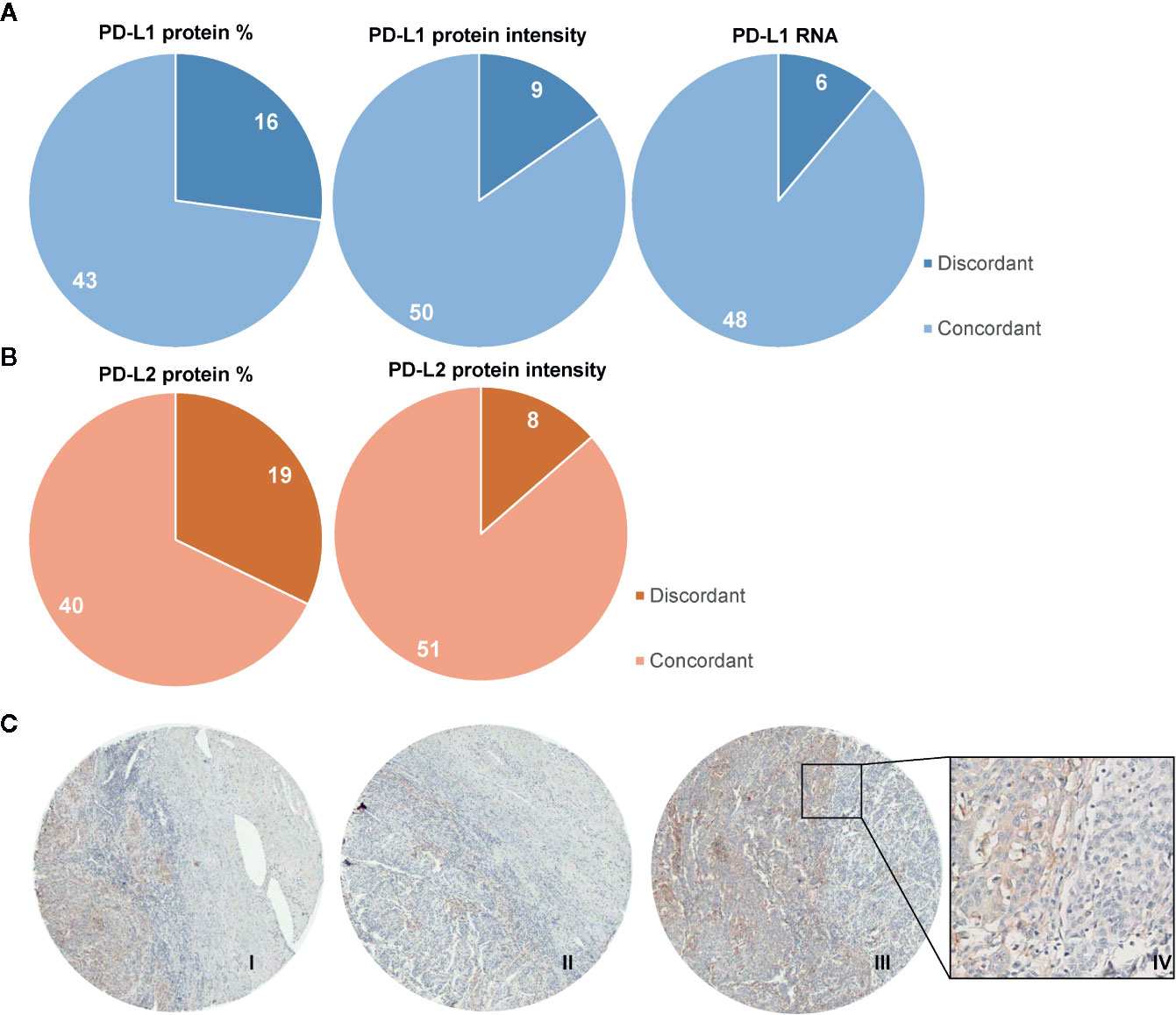
Figure 4. Heterogeneity of PD-L1 protein expression within individual tumor microarray cores from cervical cancer patients. Spatial and temporal heterogeneity in PD-L1 expression — including nuclear versus membrane distribution — has direct implications for the design and interpretation of HeLa LUC xenograft immunotherapy experiments.
Conclusion
This study provides compelling evidence that NAD⁺ metabolic reprogramming through NAMPT/SIRT1 drives PD-L1 nuclear localization and immune evasion in cervical cancer, including in the widely-used HeLa cell line. The identification of FK866 + anti-PD-L1 combination synergy opens a new therapeutic avenue and highlights the inadequacy of surface-only PD-L1 assessment. HeLa LUC remains an indispensable in vivo model for cervical cancer immunotherapy research, enabling real-time BLI tracking of tumor immune responses, drug penetration, and combination treatment outcomes.
Advance your cervical cancer research with confidence — browse our catalog of HeLa LUC cells and companion luciferase substrate reagents. Place your order today and receive expert technical support from cell line receipt through in vivo imaging protocol optimization.
Order now: Hela Cell Line / Hela Luciferase Cell Line / Hela-Luc-GFP Cell Line
References
1. Lu X, Jin P, Tang Q, et al. NAD+ Metabolism Reprogramming Drives SIRT1-Dependent Deacetylation Inducing PD-L1 Nuclear Localization in Cervical Cancer. Adv Sci (Weinh). 2025 Feb 23:e2412109. DOI: 10.1002/advs.202412109. PMID: 39988985.

